Abstract
Background:
Late-onset Pompe disease (LOPD) is caused by a deficiency of the acid α-glucosidase enzyme. In LOPD treatment, enzyme replacement therapy is delivered via intravenous infusion, typically in clinical settings. Cipaglucosidase alfa is delivered with the oral enzyme stabilizer miglustat (cipa + mig).
Objectives:
Evaluate the safety of cipa + mig home infusions.
Design:
Post hoc analysis of pooled safety data from three clinical trials in adults with LOPD (NCT02675465, NCT03729362, NCT04138277).
Methods:
The frequency and severity of infusion-associated reactions (IARs) during cipa + mig home and clinic administration were analyzed.
Results:
In total, 65/151 patients (43.0%) received ⩾1 cipa + mig home treatment. Of 9185 treatments, 2024 (22.0%) were administered at home. IAR frequency was similar for home (1.3%, 26/2024) and clinic (1.8%, 129/7161). The most frequent IAR following home infusion was pyrexia (6.2% of patients). Two patients with ⩾1 home-based treatment experienced serious IARs.
Conclusion:
Analyses support the safety of home cipa + mig treatment in eligible adults with LOPD.
Plain language summary
Introduction
Late-onset Pompe disease (LOPD) is a rare, multisystemic, heterogeneous disorder characterized by progressive loss of muscle and respiratory function, with patients often requiring wheelchair use and/or ventilatory support.1,2 LOPD is caused by a deficiency of acid α-glucosidase (GAA), an enzyme responsible for metabolizing lysosomal glycogen.1,3
Cipaglucosidase alfa plus miglustat (cipa + mig) is a two-component therapy comprising cipaglucosidase alfa, a novel bis-mannose-6-phosphate-enriched recombinant human GAA, administered in conjunction with miglustat. 4 Miglustat is an orally administered enzyme stabilizer that minimizes inactivation of cipaglucosidase alfa in the blood (where lysosomal enzymes are susceptible to the unfavorable, near-neutral pH) and increases bioavailability by 29% versus cipaglucosidase alfa alone. 5 Cipaglucosidase alfa and other enzyme replacement therapies (ERTs) for Pompe disease are administered via an intravenous (IV) infusion, typically over 4 h, every 2 weeks, for life.6–8 Patients’ quality of life may be negatively affected by the time, travel, and costs involved with the long and frequent clinic visits to administer therapies.9–11
Studies in patients with lysosomal storage disorders, including Pompe disease, have shown improvement in treatment satisfaction and quality of life after ERT infusions transitioned to the home environment.11–13 Patients with Pompe disease reported few, mostly mild symptoms during or after home infusion with the ERT alglucosidase alfa. 14
We assessed the safety of home administration of cipa + mig in a post hoc analysis of three clinical trials in LOPD (ATB200-02, PROPEL, and the PROPEL open-label extension (OLE)).15–17
Methods
Patients
Safety data were pooled from three clinical trials in adults with LOPD. The ongoing open-label, phase I/II ATB200-02 study (NCT02675465) assessed the long-term safety and efficacy of cipaglucosidase alfa (20 mg/kg) plus miglustat (260 mg) for up to 48 months in 29 ERT-experienced and ERT-naïve patients. 16 The double-blind, randomized, phase III PROPEL study (ATB200‑03; NCT03729362) assessed the safety and efficacy of cipaglucosidase alfa (20 mg/kg) plus miglustat (195 mg for patients with a body weight of 40 to <50 kg or 260 mg for patients with a body weight of ⩾50 kg) versus alglucosidase alfa (20 mg/kg) plus placebo for 52 weeks in 123 ERT-experienced or ERT-naïve patients (of whom 85 patients received cipa + mig). 15 The ongoing PROPEL OLE (ATB200-07; NCT04138277) assessed the long-term efficacy and safety of cipaglucosidase alfa (20 mg/kg) plus miglustat (195 or 260 mg, as described for PROPEL) in patients from either treatment arm of the PROPEL study. 17 In the PROPEL OLE, patients continued treatment with cipa + mig or switched from treatment with alglucosidase alfa plus placebo. 17 At the time of analysis, these were the only large-scale clinical trials of cipa + mig that included the option for home infusions. The data were pooled to provide the greatest quantity of safety data possible for these analyses.
All three studies were performed in accordance with the ethical principles of the Good Clinical Practice guidelines and the Declaration of Helsinki. Clinical study protocols were reviewed and approved by the appropriate independent ethics committee or institutional review board at each study site, and all participants provided written informed consent.15–17 In all three studies, cipaglucosidase alfa was administered as an IV infusion every 2 weeks, over approximately 4 h, 1 h after miglustat was taken orally.
Patients were potentially eligible to receive home-based treatment from a trained nurse if they tolerated infusions well and local laws and regulations permitted administration outside of a hospital setting. Inclusion criteria for home treatment were that ERT was feasible in the home setting (including availability of a nurse, and preparation and disposal requirements could be met), the patient had not experienced a moderate or severe infusion-associated reaction (IAR) or a mild IAR that could not be controlled by medication during the study, and the patient did not have a history of a recurrent or life-threatening IAR, including anaphylaxis, at any time.
Statistical analysis
We undertook a post hoc analysis of the frequency, nature, and severity of IARs and any medication errors recorded during home and clinic administration of cipa + mig in patients who received treatment in both settings. We also analyzed the frequency, nature, and severity of IARs in patients who received no home infusions.
An IAR was defined as a disorder characterized by one or more adverse events (AEs) related to infusion of the study treatment, as assessed by the investigator. A serious AE or IAR was defined as life-threatening or resulting in death, requiring inpatient hospitalization, resulting in persistent or significant incapacity or substantial disruption to the ability to conduct normal life functions, or an important medical event that, based on appropriate medical judgment, may jeopardize the patient and require medical or surgical intervention to prevent one of the serious outcomes above.
Results
Patients
Across the three clinical trials, 151 patients from 22 countries were treated with cipa + mig. Patients received a total of 9185 IV infusions of cipaglucosidase alfa plus oral miglustat, 2024 of which were administered at home and 7161 were administered in the clinic. The baseline characteristics of patients assigned cipa + mig in the three clinical trials have been previously described.15–17
In total, 43.0% (65/151) of patients who received cipa + mig received treatment both at home (⩾1 home-based infusion) and in the clinic across 59.1% (13/22) of countries. Eighty-six patients (57.0%) received no home infusions. Baseline characteristics were similar in patients with and without home-based infusions (Table 1). At least one home-based infusion of cipaglucosidase alfa was received by 44.8% (13/29) of patients in study ATB200-02, 28.2% (24/85) of patients in PROPEL (cipa + mig arm), and 42.4% (50/118) of patients in the PROPEL OLE.
Demographics and baseline characteristics of patients included in the current analysis.
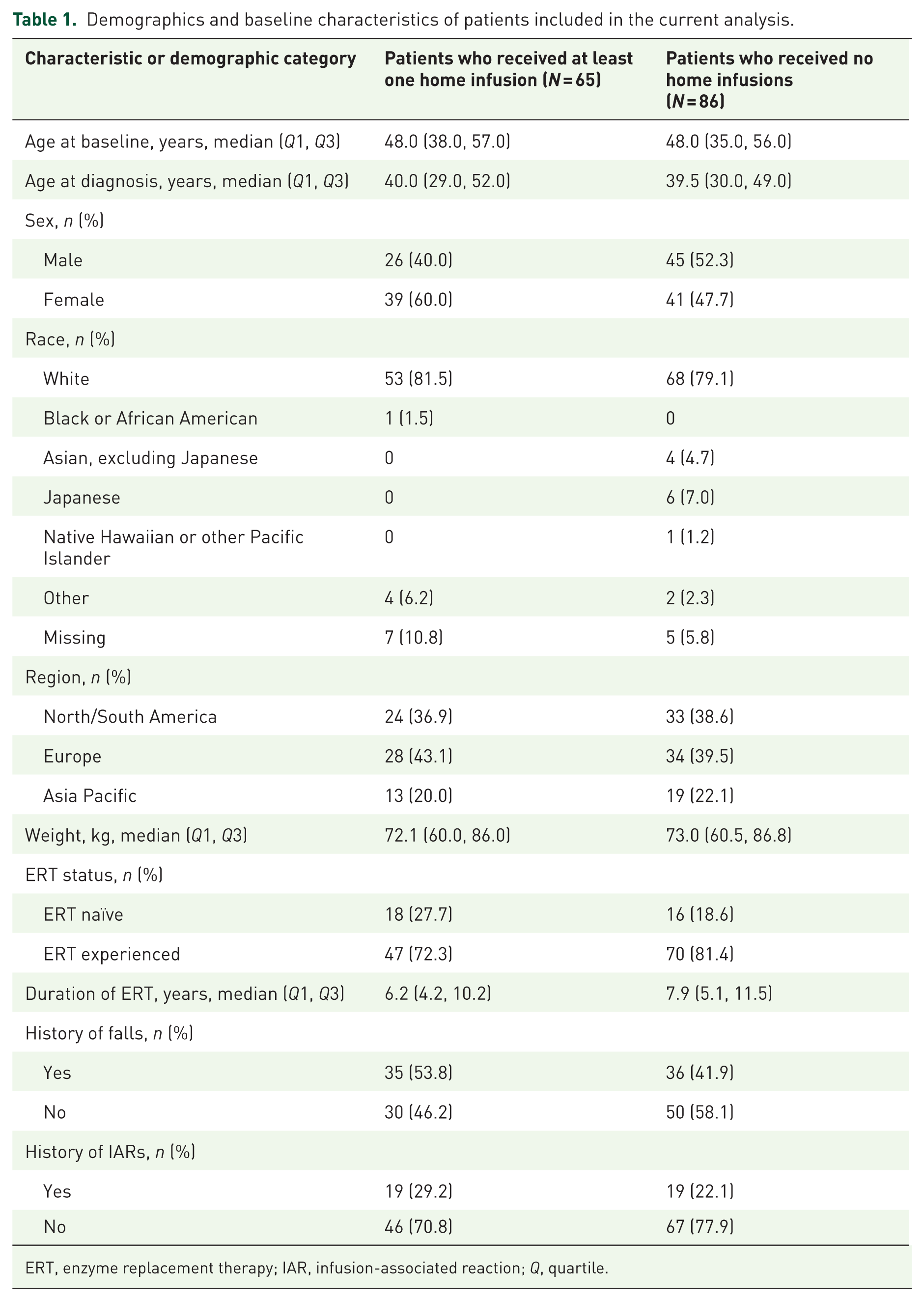
ERT, enzyme replacement therapy; IAR, infusion-associated reaction; Q, quartile.
Of the 61 patients continuing in the ongoing ATB200-02 or PROPEL OLE studies who received home infusions, 86.9% (53/61) were still receiving treatment at home at the latest data cut-off dates. For these 53 patients, the median (range) duration of home infusions was 466 (11–1273) days; only two patients (3.8%) had a duration of home infusions <50 days.
Safety of home infusions
The proportion of infusions associated with IARs was similar for those administered at home (1.3% of all home-based infusions (26/2024)) and those administered in the clinic (1.8% of all clinic-based infusions (129/7161)).
Among the 65 patients who received cipa + mig both at home and in the clinic, 20.0% (13/65) had ⩾1 IAR at home, and 30.8% (20/65) had ⩾1 IAR in the clinic (Table 2; Table S1). Of ERT-experienced patients who received home and in-clinic infusions, 14.9% (7/47) and 23.4% (11/47) had ⩾1 IAR at home and in the clinic, respectively (Table S2). Of ERT-naïve patients who received home and in-clinic infusions, 33.3% (6/18) and 50.0% (9/18) had ⩾1 IAR at home and in the clinic, respectively (Table S3).
IARs in patients who received at least one home treatment of cipa + mig (IARs with ⩾2 total events).

A patient experiencing the same TEAE multiple times is counted once for the corresponding System Organ Class/Preferred Term. TEAEs are sorted by total number of events and then alphabetically.
cipa + mig, cipaglucosidase alfa plus miglustat; IAR, infusion-associated reaction; TEAE, treatment-emergent adverse event.
The most frequent IAR (>5% of patients) experienced at home was pyrexia (6.2%; 4/65), and the most frequent IARs experienced in the clinic were headache (10.8%; 7/65) and pyrexia (6.2%; 4/65; Table 2).
Among patients who had never had treatment at home, 31.4% (27/86) experienced ⩾1 IAR, most commonly headache (9.3%; 8/86; Table S1). Of ERT-experienced and ERT-naïve patients who never had home infusions, 34.3% (24/70) and 18.8% (3/16) experienced ⩾1 IAR, respectively (Tables S2 and S3).
Among patients who had ⩾1 treatment at home, two patients (both of whom were ERT naïve upon enrollment) experienced IARs deemed serious by the investigator. One patient experienced a serious IAR of pyrexia at home, but continued to receive home-based ERT; one patient experienced a serious IAR of presyncope in the clinic before starting home infusion.
There were two medication errors: one at home (accidental overdose of miglustat) and one in the clinic (accidental overdose of blinded study treatment). Both overdose events were reported as AEs; however, neither patient experienced any adverse outcomes associated with the overdose.
Discussion
Overall, the results of this post hoc analysis of safety data pooled from three clinical trials support the safety of home-based administration of cipa + mig in ERT-experienced and ERT-naïve adults with LOPD, with a similar proportion of infusions associated with IARs when treatment was administered at home and in the clinic.
Our findings concur with other studies that provide evidence of the safety profile of home-based administration of ERT in Pompe disease and other lysosomal disorders.14,18,19 In a report evaluating the safety of alglucosidase alfa home infusions (N = 116, including n = 82 adult patients), 10 adult patients reported 13 instances of symptoms within 48 h of an infusion, with only three patients reporting IARs. 14 While the incidence of IARs for home infusions was very low in this study, there was no comparison with in-clinic infusions. Moreover, data were collected through patient questionnaires, which can yield varied results determined by the specific phrasing of the questions and the patients’ understanding. For example, while only three adults indicated IARs on one or more questions, six also indicated that their health complaint during or within 48 h of the last infusion was related to the infusion. 14 A report on the safety of home infusions for patients with Pompe disease treated with avalglucosidase alfa, another next-generation ERT, included 15 patients with LOPD and two with infantile-onset Pompe disease across three clinical studies (NEO-EXT, COMET, and Mini-COMET) who had been considered appropriate candidates for home infusion by investigators. 19 Similar rates of IARs were reported when patients with LOPD received treatment at home versus in the clinic, with no serious events occurring; however, it is possible that diagnosis and reporting of AEs may have differed between the home and clinic environments. These results are consistent with those reported here. However, the population for patients receiving avalglucosidase alfa via home infusion was much smaller, making direct comparisons between the two treatments challenging.
In the current study, each patient had access to a trained nurse and was able to meet the requirements for preparation and disposal. In real-world treatment, the possibility of transitioning to home infusions may be influenced by the availability of appropriately trained nurses or healthcare professionals and a suitable home environment, which are mandatory requirements for home infusion administration. 20 Safety could be influenced by several other factors, including whether the patient resides in a rural or urban setting and proximity to emergency care facilities. Moreover, there may be regional variability in the feasibility of home infusions. For example, different countries may have varying opinions about home care or local laws and regulations, which can determine whether home infusions are possible.
Owing to the inherent bias introduced by the inclusion criteria for home infusions (patients must have had no moderate or severe IARs, or a mild IAR that could not be controlled, during the study), it was not appropriate to compare the frequency of IARs experienced between patients who had at least one home infusion with those who had no home-based infusions. Generally, IARs can occur at any time throughout treatment, and data from PROPEL indicate that most IARs occur within 1−4 months of initiating treatment. 15 However, the incidence of early IARs does not predict nor preclude the potential occurrence of subsequent IARs with continued treatment. This analysis has several limitations, including the post hoc nature of the analysis and the potential for differential reporting of AEs during home-based and clinic-based administration. Selecting only patients who have a minimal history of IARs will impact the generalizability of these data to all patients with LOPD. However, this eligibility criterion reflects how patient suitability for home infusion would be determined in real-world clinics; therefore, these results are reflective of the population likely to transition to home infusions. Furthermore, while the original studies included various patient-reported outcomes, they did not include assessments of the impact of home infusions on patients’ satisfaction with treatment or on quality of life. To address this, future studies that include home infusions as an option should consider incorporating patient-reported outcomes to assess the impact of transitioning to home infusion on quality of life.
Conclusion
In summary, this analysis shows that cipa + mig can be safely administered in a home setting, provided patients are assessed for their eligibility (no history of moderate/severe IARs) and have access to sufficient resources. Future studies that investigate the impact of transitioning to home infusions on patients’ quality of life should incorporate patient-reported outcomes.
Supplemental Material
sj-docx-1-trd-10.1177_26330040261416943 – Supplemental material for Safety of home administration of cipaglucosidase alfa plus miglustat in late-onset Pompe disease: results from multiple clinical trials
Supplemental material, sj-docx-1-trd-10.1177_26330040261416943 for Safety of home administration of cipaglucosidase alfa plus miglustat in late-onset Pompe disease: results from multiple clinical trials by Henning Andersen, Jordi Díaz-Manera, Ozlem Goker-Alpan, Tahseen Mozaffar, Sheela Sitaraman Das, Brian Fox, Farah Amon, Kinesha O’Brien-Prince, Mitchell Goldman, Fred Holdbrook, Vipul Jain and Barry J. Byrne; in Therapeutic Advances in Rare Disease
Supplemental Material
sj-docx-2-trd-10.1177_26330040261416943 – Supplemental material for Safety of home administration of cipaglucosidase alfa plus miglustat in late-onset Pompe disease: results from multiple clinical trials
Supplemental material, sj-docx-2-trd-10.1177_26330040261416943 for Safety of home administration of cipaglucosidase alfa plus miglustat in late-onset Pompe disease: results from multiple clinical trials by Henning Andersen, Jordi Díaz-Manera, Ozlem Goker-Alpan, Tahseen Mozaffar, Sheela Sitaraman Das, Brian Fox, Farah Amon, Kinesha O’Brien-Prince, Mitchell Goldman, Fred Holdbrook, Vipul Jain and Barry J. Byrne; in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors thank the patients, their families, Pompe disease patient organizations, and the ATB200-02, 03, and 07 study investigators. The authors also thank Yasmine Wasfi, formerly of Amicus Therapeutics, Inc., for her valuable contributions to this study. Editorial assistance was provided by Kara Filbey, PhD, at Amiculum, and was funded by Amicus Therapeutics, Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
