Abstract
Cardiovascular disease remains the leading global cause of death, with its burden exacerbated by rising rates of diabetes mellitus and obesity. Recent therapeutic advances have revealed the dual potential of novel antidiabetic agents—not only to improve glycemic control but also to significantly reduce cardiovascular and renal morbidity.
This report explores the emerging roles of sodium–glucose cotransporter 2 inhibitors and glucagon-like peptide-1 receptor agonists in cardiovascular disease management. Evidence from major clinical trials highlights their efficacy in reducing heart failure hospitalizations, atherosclerotic events, and in slowing the progression of chronic kidney disease. The discussion emphasizes the mechanisms beyond glycemic control that underpin their cardio-renal benefits, and reflects on the paradigm shift towards integrated, multi-system disease management. These developments signify a promising future where metabolic therapies play a central role in cardiovascular care.
Keywords
Introduction
With global rates of obesity growing and sedentary lifestyles becoming increasingly common, the burden of cardiovascular disease (CVD) is increasing as the number one cause of mortality and morbidity worldwide. 1 Perhaps what has been given more attention in recent years is the multifactorial nature of CVD. Even with guideline-directed medical therapy, current medications are not sufficient to manage this public health issue.
Often, various comorbidities are present in combination. A common cause of CVD is the systemic disease—diabetes mellitus (DM). Frequently, there is an element of chronic kidney disease (CKD) in these patients. Due to its microvascular and macrovascular complications, DM can affect several physiological systems. 2 Optimal glycemic control of DM can have the desired effects for the heart, kidneys, and vascular systems. Considering that people with diabetes are at a higher risk for CVD, it is desirable to find a shared therapeutic solution. 3
Multiple randomized controlled trials of antidiabetic medications have been carried out with positive results. One aspect causing significant surprise was the substantial cardiovascular benefits. Two drugs in particular have shown considerable promise, namely, sodium–glucose cotransporter 2 (SGLT2) inhibitors and glucagon-like peptide-1 (GLP-1) agonists.
Existing Evidence: Sodium–Glucose Cotransporter 2 Inhibitors
Sodium–glucose cotransporter 2 inhibitors have a wide range of clinical uses. These include DM, CKD, gout, and, more recently, heart failure (HF). They target the proximal convoluted tubule (PCT) within nephrons to prevent absorption of sodium and glucose. 4 This mechanism works by the selective inhibition of the SGLT2 channel protein, which promotes the reuptake of these substances. A compound known as “Phlorizin” was the first natural substance to act as a competitive antagonist of both the SGLT1/2 proteins. 5
Inhibiting the SGLT2 transporter results in glucosuria, thereby highlighting its role in treating diabetes by increasing glucose excretion. 6 In addition, sodium reuptake is reduced equally, meaning that natriuresis occurs along with diuresis due to osmotic gradients. It is via this mechanism that SGLT2 inhibitors have shown drastic reductions in major adverse cardiovascular events (MACE), coupled with cardiorenal protection.7, 8
Besides the diuretic and glycosuric effects, there is evidence to suggest that SGLT2 inhibitors have anti-hypertrophic and anti-apoptotic effects, contributing to the reduction in cardiovascular morbidity. 9 As well as this, they may also promote the production of erythropoietin (EPO) shortly after the initiation of treatment. 10 In the context of HF, dapagliflozin and empagliflozin are commercially produced. Previously, in large clinical trials, SGLT2 inhibitors have performed significantly well in reducing plasma glucose levels in comparison to other therapies. 11
Whilst the initial indication was for diabetes, the slowing of cardiac and renal dysfunction cannot be attributed to this glucose-lowering action alone. 12 Indications of an additional advantage can be seen when compared to other antidiabetic drugs. In fact, stronger antihyperglycemic agents do not exhibit similar cardiorenal benefits. At the lowest ranges of estimated glomerular filtration rate (eGFR: 0-15—Stage 5 CKD), observed in patients on maintenance hemodialysis (MHD), the glycosuric effect would be considered negligible. However, benefits are still seen in dialysis patients with supposedly no intrinsic kidney function, even when the local action of SGLT2 inhibitors is hindered.
This transformative use of SGLT2 inhibitors has revolutionized the pharmacological management of HF. Now, dapagliflozin and empagliflozin are Food and Drug Administration (FDA)-approved drugs (Table 1) that are incorporated into the existing first-line regimen for both HF with reduced ejection fraction (HFrEF) and HF with preserved ejection fraction (HFpEF). In stark contrast to the other nephrotoxic drugs utilized to manage HF, the SGLT2 inhibitors have shown promising results in the latest trials. 13 Sodium–glucose cotransporter 2 inhibitors have demonstrated cardiovascular outcomes comparable to other standard HF medications, such as angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and mineralocorticoid-receptor antagonists.
Overall Characteristics of Sodium–Glucose Cotransporter 2 (SGLT2) Inhibitors and Eligibility in Heart Failure (HF).
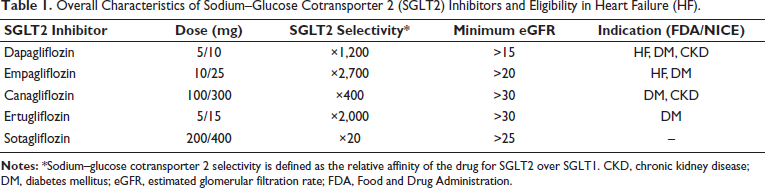
The higher the value, the more specific it is for SGLT2. A value of 1 indicates no difference. For reference, the original inhibitor, “Phlorizin,” has an SGLT2 selectivity factor of roughly 1.5.
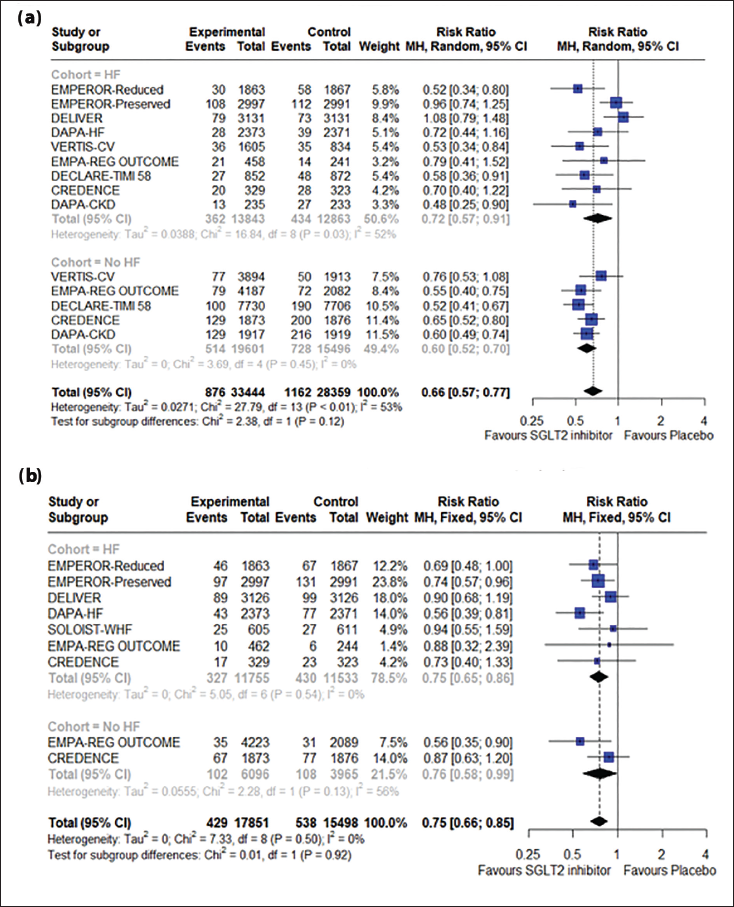
Not only does this class of drugs prevent cardiovascular death and heart failure hospitalization (HHF), but progression to CKD is also slowed. Here we can see the interplay of the cardiac and renal systems, and how SGLT2 inhibitors can slow down the impairment that would otherwise culminate in cardiorenal syndrome. The renin-angiotensin-aldosterone system (RAAS) is activated when the body detects reduced blood pressure or renal blood flow. 14 It is also sensitive to levels of sodium chloride (NaCl) at the distal convoluted tubule in the nephron. 15 Excessive stimulation of the RAAS leads to fluid and sodium retention, and the preload is increased. Moreover, increased pressures have a detrimental effect on afterload. Natriuresis via SGLT2 inhibitors causes afferent arteriole constriction via tubuloglomerular feedback, dampening the effect of high pressures in the glomerulus.16, 17 Vascular function and hemodynamics are preserved. This is one of the mechanisms linked to the protection of renal function—one of the features studied in this review. 18 Similarly, it could be argued that conventional diuretics also have the same effect. However, SGLT2 inhibitors prevent the excess depletion of intravascular volume and promote tissue fluid excretion.19, 20 Lowering blood pressure limits the afterload pressure on the heart. These changes do not show a significant difference depending on the HF status, highlighting the utility of these drugs (Figure 1A and 1B).
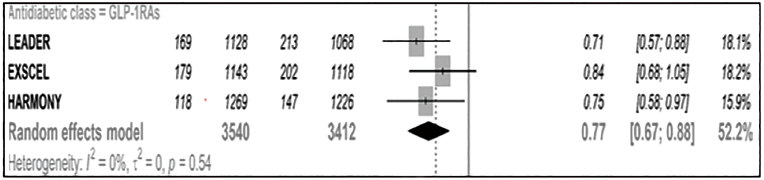
Furthermore, there have been various off-target effects postulated relating to SGLT2 inhibitors (Figure 2). As mentioned previously, the site of action of SGLT2 inhibitors is in the PCT. Nevertheless, animal studies have theorized possible mechanisms within the heart itself. Examples of this are anti-inflammatory and anti-fibrotic effects. As well as this, reductions in adipose tissue cause weight loss, which in turn can reduce epicardial fat, thereby reducing the prevalence of atherosclerotic heart disease.
Forest Plot—Major Adverse Cardiovascular Events (MACE). 15
Summary of Described Mechanisms of Cardiorenal Protection by Sodium–Glucose Cotransporter 2 (SGLT2) Inhibitors.
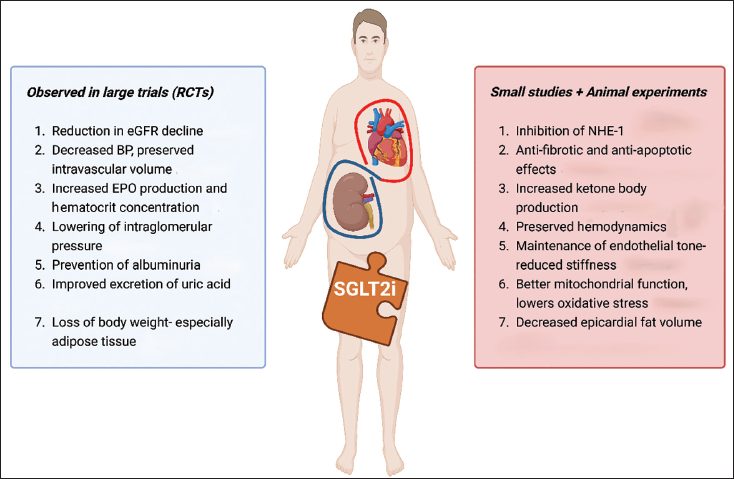
Conflicting results have been published by different groups, with perhaps the most compelling evidence coming from Zuurbier et al. 21 , demonstrating inhibition of the sodium-hydrogen antiporter 1 (NHE1). Sodium-hydrogen antiporter 1 is responsible for the exchange of hydrogen ions out and sodium ions into cells to maintain intracellular pH. When this process is active, the nearby sodium-calcium exchanger (NCX) re-releases this sodium and takes in calcium. 22 Inhibition of NHE1 prevents cytosolic build-up of calcium, which can cause impaired cardiac contraction in HF.
A novel association in this fat differentiation pathway has demonstrated that SGLT2 inhibitors could potentially reduce epicardial fat (Figure 3). 23 This reduction is likely to be a result of improved glycemic control combined with increased insulin sensitivity after exposure to SGLT2 inhibitors. Moreover, β-hydroxybutyrate (β-OHB) gives more energy than the oxidation of glucose or non-esterified fatty acids (NEFAs). β-Hydroxybutyrate levels are increased with SGLT2 inhibitors, ameliorating pump function and energy usage. 24 Infusion of β-OHB in patients with chronic HF has been shown to enhance ejection fraction and stroke volume in a small study. 25
Recent Developments: Glucagon-like Peptide-1 Receptor Agonists
On a similar topic of glycemic control, glucagon-like peptide-1 receptor agonist (GLP-1RA) agonists have also shown benefits extending beyond their use in diabetes. Glucagon-like peptide-1 is a hormone that has multiple effects. It increases insulin secretion during the post-prandial state. In addition, glucagon levels and appetite are suppressed. 26 Commonly, GLP-1RAs are prescribed for weight loss in individuals with class II obesity (BMI ≥35 kg/m2) or obesity-related comorbidities. Semaglutide, Exenatide, and Liraglutide are some of the GLP-1RAs that are licensed for use in the United Kingdom.
In the same fashion as SGLT2 inhibitors, cardiovascular and renal benefits have been observed to a significant level in the LEADER (Liraglutide) and SUSTAIN-6 (Semaglutide) trials. Overall, the LEADER trial showed the most convincing results across cardiovascular endpoints (Figure 2). 27 SUSTAIN-6 demonstrated that Semaglutide caused a significant 26% reduction in the risk of non-fatal stroke. In the REWIND study (Dulaglutide), renal outcomes were also reduced. Rates of macroalbuminuria and declines in glomerular filtration rate occurred less with therapy compared to placebo. Definitions for these endpoints were similar to those used for the renal-composite outcomes in SGLT2 inhibitor trials.
Twenty years ago, patients meeting the indication for this type of medication would be few and far between. However, with obesity and DM rates rising, there is now likely to be a larger pool of patients where GLP-1RA can be used. Subsequently, utilizing GLP-1RA can also bring about consistency by reducing the need for multiple adjunctive therapies.
Again, the exact pathophysiology by which GLP-1RA shows their efficacy in CVDs is yet to be understood fully. A proposed mechanism is credited to blood pressure reduction (preserving vascular/endothelial tone). Nonetheless, the pattern is the same as illustrated by patients receiving SGLT2 inhibitors.
Conclusion
To conclude, in an era of polypharmacy and personalized medicine, the phrase “to kill two birds with one stone” is an apt description of the characteristics required of new treatments. In fact, one could argue that SGLT2 inhibitors and GLP-1RA do much more than this, with the evidence for them demonstrating a myriad of ways in which they can improve cardiac, renal, and vascular health. Compared to other drugs, the evidence base for these two drug classes has been tested across large populations in multicenter studies.
Sodium–glucose cotransporter 2 inhibitors now form part of a four-pronged combination of medications for HF treatment. Additionally, it is the only one that confers benefits across all HF phenotypes in contrast to ACE inhibitors or angiotensin receptor blockers/neprilysin inhibitors (ARNIs). With GLP-1RA also demonstrating similar benefits, it could only be a matter of time before they are approved for the management of CVD in overweight patients.
These developments underpin the importance of integrated care and managing multiple conditions using a multidisciplinary approach. Whilst novel drugs are expensive, the hope is that overall, they will vastly reduce stress on healthcare systems and improve patient quality of life.
Footnotes
Declaration of Conflicting Interest
The author declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
The participant has provided informed consent for the submission of the article to the journal.
