Abstract
Acquired methemoglobinemia is a clinical condition precipitated by some offending agents, like nitrates. We are reporting a case of methemoglobinemia with hemolysis in a 51-year-old adult patient after oral nitrate use. The patient was a case of inferior wall myocardial infarction with complete heart block post-stenting to the right coronary artery. Two days later, the patient started desaturating with normal partial pressure of oxygen in arterial blood and fraction of inspired oxygen, along with signs of hemolysis. After reviewing medications, oral nitrate was thought to be the offending agent, which was withdrawn. Oxygen supplementation with a high dose of vitamin C and other supportive care was given. Due to the unavailability of a confirmatory test, serum methemoglobin was sent to an outside laboratory and came back positive after a few days. The patient responded to the treatment and was discharged after 10 days. By presenting this case, we aim to raise awareness about the potential risks of oral nitrates and the importance of early diagnosis and intervention.
Introduction
Methemoglobinemia is a life-threatening clinical condition that results from the oxidation of hemoglobin to methemoglobin, as the iron atom of haem is in the ferrous rather than the ferric state. 1 This causes impaired hemoglobin oxygen-carrying capacity, which leads to tissue hypoxia. Many drugs containing nitrates trigger the oxidation of hemoglobin, leading to the pathological disorder named methemoglobinemia. 2 We are reporting a case of a 51-year-old gentleman with inferior wall myocardial infarction (MI) who developed methemoglobinemia following oral nitrate ingestion. This study aims to report a rare case of oral nitrate-induced methemoglobinemia and hemolysis, emphasizing its clinical presentation, diagnosis, and management.
Case Report
A 51-year-old gentleman who is diabetic, hypertensive, and a known case of chronic kidney disease was admitted to our institute with a diagnosis of inferior wall MI with complete heart block. Primary angioplasty to the right coronary artery (RCA) was performed immediately under temporary pacemaker insertion (TPI). Post-procedure, the patient was treated and observed in the cardiac intensive care unit (ICU). Dual antiplatelet therapy and oral nitrates were continued as per the routine post-percutaneous transluminal coronary angioplasty (PTCA) protocol.
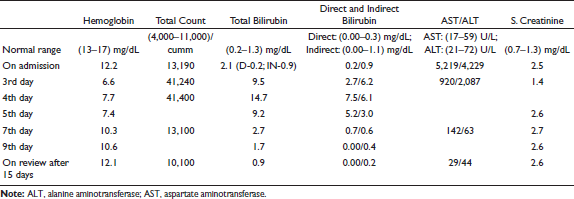
Initial blood investigations revealed deranged liver and renal function tests, which were considered secondary to the MI and complications of complete heart block.
Initially, the patient’s oxygenation was normal, with no signs of heart failure. However, on the third day, his peripheral oxygen saturation (SpO2) dropped to 80% on room air, but arterial blood gas (ABG) showed a partial pressure of oxygen in arterial blood (PaO2) of 131 mmHg and an SpO2 of 99%. Both electrocardiography (ECG) and chest X-ray were within normal limits.
Oxygen therapy was started, and repeat blood investigations showed low hemoglobin, marked leukocytosis, and a sudden rise in bilirubin, without significant changes in baseline liver enzymes. The patient was evaluated for autoimmune hemolytic anemia, glucose-6-phosphate-dehydrogenase (G6PD) deficiency, viral hepatitis, and pneumonia.
Results showed:
Indirect and direct Coombs tests: Negative Hepatitis A and E virus IgM antibodies: Negative Serum protein electrophoresis (SPEP) for M-band: Negative Antinuclear antibody reflex panel: Negative Hemoglobin typing: Normal chromatogram Serum lactate dehydrogenase (LDH) and reticulocyte count: Elevated Qualitative G6PD test: 5.7 U/g Hb (0-2: deficient; 2-6: intermediate; >6: normal)
Peripheral blood smear showed normocytic normochromic anemia with neutrophilic leukocytosis, a left shift with metamyelocytes and myelocytes, and no toxic granules.
On the fourth day, the patient became more symptomatic, developing respiratory distress and cyanosis. His urine appeared dark brown (Figure 1), and oxygen requirements increased. Despite this, ABG analysis showed normal PaO2 and SaO2. The disparity between pulse oximeter readings and arterial oxygen saturation, along with dark-colored urine, low hemoglobin, and elevated bilirubin, raised suspicion of methemoglobinemia in the setting of hemolytic anemia.
The Urine in the Container is of Dark Brown Color.

A serum methemoglobin level was sent to an external laboratory. On reviewing the patient’s medications, oral nitrate was suspected as the offending agent and was discontinued. High-flow oxygen and high-dose vitamin C were administered. Due to the unavailability of methylene blue and pending confirmation of the diagnosis, specific treatment could not be initiated. The patient was transfused with three units of packed red blood cells.
Treatment Progress
Initially, the patient’s condition worsened. However, after discontinuation of oral nitrates and the initiation of high-flow oxygen and vitamin C, he gradually responded to treatment. By the seventh day, his clinical condition improved with continued supportive care. On the eighth day, we received a positive methemoglobin report with a value of 3.40 (ref. range <1.5%).
Treatment Outcome
Finally, the patient was discharged after 10 days of hospital stay, with advice to review and avoid certain medications.
Discussion
Methemoglobinemia is of two types. Congenital form of methemoglobinemia is a rare genetic disorder, whereas acquired methemoglobinemia is often associated with exposure to certain medications (dapsone, lidocaine, benzocaine) and compounds containing nitrate, chlorate, and aniline groups. 3 One intriguing subset of acquired methemoglobinemia arises from the ingestion of oral nitrate. Nitrates are metabolized in the liver, and the metabolites oxidize hemoglobin to methemoglobin. Three clinical entities—(a) refractory hypoxia, (b) “cyanosis-saturation gap,” and (c) dark brown blood—suspect methemoglobinemia. Diagnosis of methemoglobinemia was confirmed by co-oximetry. 4
The simultaneous occurrence of both methemoglobinemia and non-immune hemolytic anemia has been documented in patients with an inherited G6PD defect and/or following an overdose of oxidative drugs. This is thought to result from increased production of reactive oxygen species (ROS), which contributes to both iron oxidation and red blood cell destruction. 5
The agents producing methemoglobinemia may also produce oxidant-induced hemolysis, and hence, a combination of methemoglobinemia and hemolytic anemia may occur. Hemolysis in our patient can be related to G6PD deficiency, but as the G6PD enzyme was in the intermediate range, we are unable to specify the reason here.
Methylene blue is indicated as the first-line antidote therapy for patients with severe methemoglobinemia. Although successful treatment with plasma exchange therapy, hyperbaric oxygen therapy, and high dose of ascorbic acid has also been reported, these therapies should be considered as second-line treatments for patients unresponsive to methylene blue.
In a resource-limited setting like ours, timely identification of methemoglobinemia, evaluation of the etiology, and supportive management without definitive treatment may cure the condition.
Footnotes
Acknowledgment
The authors would like to express their sincere gratitude to all nursing and technician staff of Cardiac ICU, Nemcare Superspeciality Hospital, for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
As this is a case report, ethical committee approval was not required for this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Since the authors are not disclosing the patient’s identity, confidentiality has been maintained. Therefore, consent from the patient was not taken.
