Abstract
Background
Although methylene blue and hydroxocobalamin are commonly used as rescue treatments for refractory shock, large-scale head-to-head trials comparing these agents are lacking. This study aimed to evaluate their effects on vasopressor doses in patients with refractory shock.
Methods
A retrospective observational study was conducted among patients who received methylene blue or hydroxocobalamin for refractory shock. The primary endpoint was the change in vasopressor dose 24 hours after administration. Secondary outcomes included changes in mean arterial pressure and vasopressor dose over 24 hours (0, 6, 12, and 24 hours), time to vasopressor discontinuation, vasopressor-free days at 28 days, ICU and hospital length of stay, and hospital mortality.
Results
A total of 303 patients who received methylene blue (n=269) or hydroxocobalamin (n=34) were included. The mean reduction in vasopressor doses over 24 hours was similar (methylene blue 0.36 mcg/kg/min vs hydroxocobalamin 0.33 mcg/kg/min, p=0.588). The mean reduction from baseline was significant in both groups at 6, 12, and 24 hours after administration (p<0.001 each). The methylene blue group had a higher in-hospital mortality rate (78% vs 59%, OR 2.4 [95% CI 1.2-5.1]) and shorter median time to death (18 hours vs 106 hours, p = 0.003). However, among patients alive at discharge, there was no statistically significant difference between methylene blue and hydroxocobalamin in time to discontinuation of vasopressors (90 hours vs 129 hours), ICU length of stay (19 days vs 33 days), hospital length of stay (33 days vs 54 days), or vasopressor-free days at 28 days (18 days vs 14 days).
Conclusion
Among patients with refractory shock, methylene blue and hydroxocobalamin were associated with significant reductions in vasopressor doses over 24 hours; however, no significant difference was observed between the two agents.
Introduction
Refractory shock is variously defined as failure to achieve a systemic arterial blood pressure goal despite vasopressor therapy, need for rescue vasopressor therapy, or need for high-dose vasopressor support, frequently greater than 0.2 to 0.5 mcg/kg/min of norepinephrine or an equivalent, in the presence of hypotension and end-organ failure.1–4 One of the best indicators of short-term mortality in critically ill patients is the amount of vasopressors needed to maintain an acceptable mean arterial pressure (MAP).5,6 Patients who continue to worsen clinically and receive vasopressors at doses greater than 1.0 mcg/kg/min of norepinephrine equivalents have reported mortality rates as high as 80–90%, which may be attributable to a more serious underlying condition, immune dysregulation, or possible side effects from vasopressor medications.7–10 Methylene blue and hydroxocobalamin have been used as rescue treatments, although there is limited evidence demonstrating differences in clinical outcomes between the two agents.11–13
Methylene blue inhibits both guanylate cyclase (GC) and nitric oxide synthase (NOS), which in turn diminishes nitric oxide’s effects on smooth muscle relaxation.14,15 Similarly, high dose intravenous hydroxocobalamin diminishes the effects of inflammatory mediators including NOS, GC, and hydrogen sulfide (H2S).15,16 Most of the data comparing hydroxocobalamin and methylene blue originates from the treatment of refractory vasoplegic syndrome, a type of sepsis-like vasodilatory shock that occurs after cardiopulmonary bypass. In these studies, methylene blue and hydroxocobalamin have been found to reduce the need for vasopressors and improve hemodynamic parameters, with one study suggesting a greater reduction in vasopressor doses with hydroxocobalamin. 17
Numerous studies individually evaluating methylene blue or hydroxocobalamin in septic shock have found reductions in time to vasopressor discontinuation and dose. Results from one randomized controlled trial demonstrated that early adjunctive methylene blue reduced vasopressor doses. 18 In another randomized controlled trial, high dose IV hydroxocobalamin was also found to lower vasopressor doses, with improved hemodynamic parameters such as MAP demonstrated in preclinical trials. 19
While hydroxocobalamin is typically administered as a 5 g dose over 10–15 min, methylene blue dosing varies more widely, with an initial dose of 1–2 mg/kg over 15 min to 6 h, which may be followed by a continuous infusion if needed. This variability in dosing is reflected across several studies. For example, one randomized controlled trial evaluated a fixed 100 mg dose over 6 h in septic shock. 18 On the other hand, in vasoplegic shock, two retrospective studies reported a median 1.2 mg/kg dose, which was followed by a median 0.25 mg/kg/hr continuous infusion in 25% of patients in one study.17,20
Although methylene blue and hydroxocobalamin have demonstrated efficacy in refractory shock across various dosing strategies, there is a lack of large-scale head-to-head trials directly comparing these agents or dosing strategies. We hypothesized that there would be no significant difference between methylene blue and hydroxocobalamin in reducing vasopressor doses. To address this knowledge gap, we aimed to compare their effects on vasopressor doses in patients with refractory shock.
Methods
This retrospective observational cohort study evaluated adult patients with refractory shock at two academic medical centers who received methylene blue or hydroxocobalamin between January 1, 2018 and June 30, 2023. Patients 18 years of age or older were included if vasopressor doses were at least 0.2 mcg/kg/min of norepinephrine equivalents (NEE) at the time of methylene blue or hydroxocobalamin administration, following the threshold used in the ATHOS-3 trial. 4 Exclusion criteria included administration of both methylene blue and hydroxocobalamin during admission, burn injury, and pregnant patients. Intraoperative administration of these agents was also excluded, as we were unable to collect all of the relevant data points.
The primary outcome of this study was the change in vasopressor dose 24 h after methylene blue or hydroxocobalamin administration. Vasopressor doses were expressed in NEE as defined by: NEE = Norepinephrine (mcg/kg/min) + Epinephrine (mcg/kg/min) + (Phenylephrine (mcg/kg/min)/10) + (Dopamine (mcg/kg/min)/100) + (Vasopressin (units/min) *2.5) + (Angiotensin II (mcg/kg/min)*10). 21 Vasopressor doses were assessed at methylene blue or hydroxocobalamin initiation (baseline) and then at hours 6, 12, and 24 post-administration to characterize early and sustained hemodynamic effects of each agent. Secondary outcomes included MAP at baseline and hours 6, 12, and 24. Clinical outcomes evaluated included vasopressor-free days at 28 days, time to vasopressor discontinuation, intensive care unit (ICU) length of stay, hospital length of stay, and hospital mortality.
Other baseline characteristics and interventions were also recorded, such as Quick Sequential Organ Failure Assessment (QSOFA) prior to agent administration and utilization of corticosteroids, positive inotropes, or mechanical circulatory support including Extracorporeal Membrane Oxygenation (ECMO), intra-aortic balloon pumps (IABPs), and transvalvular microaxial flow pumps. The QSOFA score was used to provide a measure of illness severity to compare the groups. Shock type was determined based on clinician documentation.
Following approval by the Institutional Review Board, patients were identified based on reports summarizing administrations of each agent during the study period. A designated member of the study team at each site collected data from the corresponding medical records.
Institutional practice
Both agents are available adjuncts for refractory shock at our medical centers, with no restrictions or preferences for use in specific patient populations. At one institution, an order set is available to guide clinicians in initiating these agents. This order set notes that methylene blue can be considered in patients who are refractory to three or more vasopressors. The dosing starts with a 2 mg/kg bolus, which can be followed by a 3 mg/kg infusion over six hours in patients who respond to the bolus, particularly in septic shock. The other institution primarily uses bolus doses alone for both agents, with dosing up to the clinician’s discretion.
Statistical analysis
Descriptive statistics were used for baseline characteristics. Categorical variables were summarized as discrete values and percentages, n (%), while continuous variables were described using mean with standard deviation (SD) or median with interquartile range (IQR) depending on the distribution. Normal distribution was determined based on whether kurtosis and skewness values were between −2 and +2. Categorical variables such as sex and type of shock were compared between agents using Pearson chi-square tests. Independent t-tests or Mann-Whitney U tests were used to compare continuous variables such as age and dose depending on the parametric or non-parametric distribution. Multiple imputations were used to account for two missing MAP data points. Then, a linear mixed-effects model was created to adjust for differences in baseline characteristics, including QSOFA, mechanical circulatory support, use of positive inotropes, type of shock, number of administrations, and number of vasopressors at baseline. These variables were selected based on clinical relevance and statistical significance in the univariate analysis. A subgroup analysis was also completed to identify differences in the reduction in vasopressor doses across shock types and initial methylene blue dosing strategies. A classification and regression tree analysis was conducted to evaluate vasopressor doses associated with risk of mortality at each time point. All analyses were conducted with a significance level of 0.05 using SPSS software version 29.0.2.0 (IBM Corporation, Armonk, NY, USA).
Results
Baseline characteristics
Baseline characteristics.
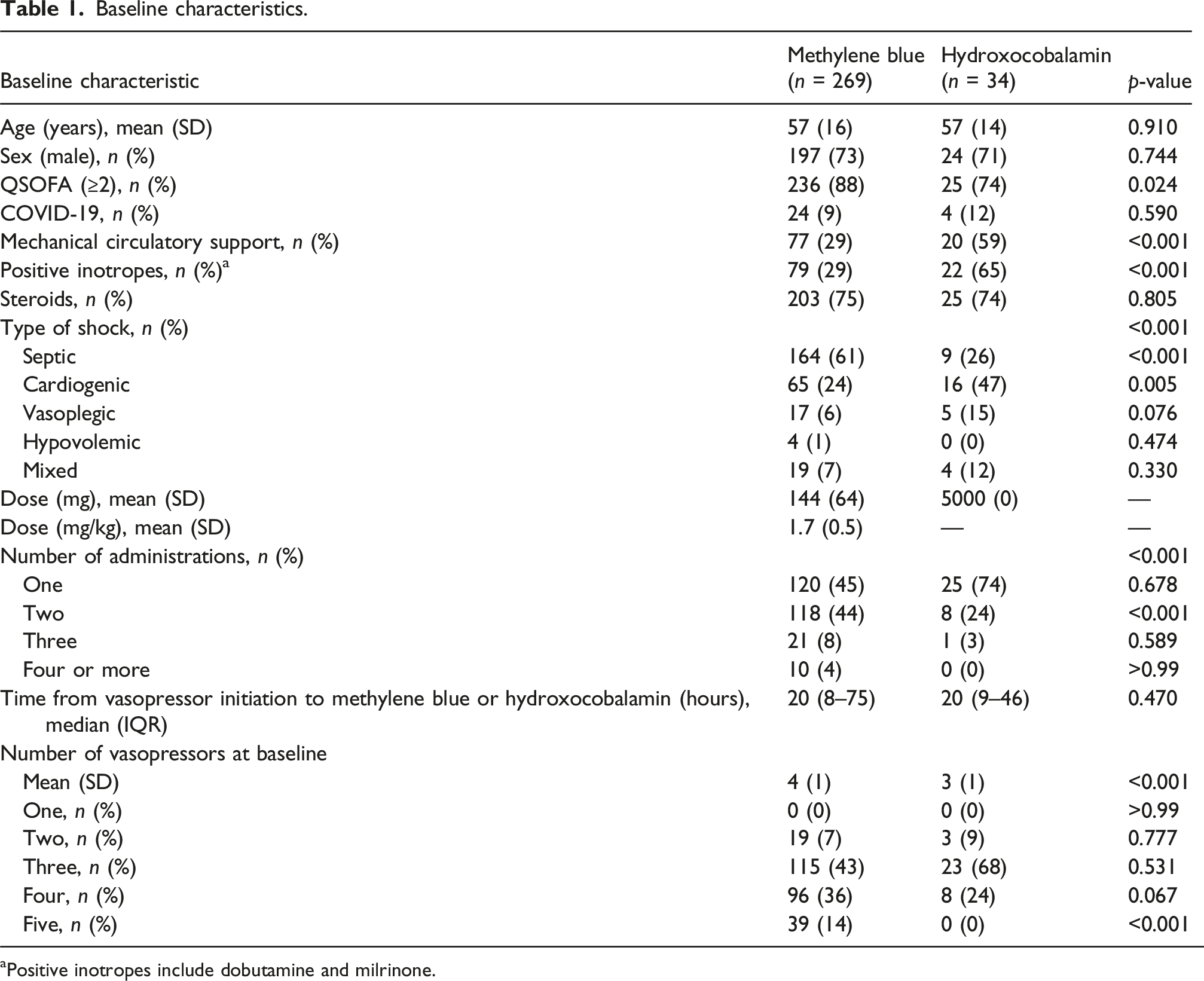
aPositive inotropes include dobutamine and milrinone.
The mean dose was 144 ± 64 mg (1.7 ± 0.5 mg/kg) in the methylene blue group and 5 grams in the hydroxocobalamin group. The median time from vasopressor initiation to administration was 20 h for both methylene blue (IQR 8–75) and hydroxocobalamin (IQR 9–46). Additionally, a higher proportion of patients in the methylene blue group received more than one dose during their hospitalization. Specifically, among those in the methylene blue group who received more than one dose (n = 149), 123 patients (83%) received a bolus dose followed by at least one 6-hour infusion, and the remaining 26 patients (17%) received several bolus doses over 30 min.
Outcomes
Vasopressor doses over time.
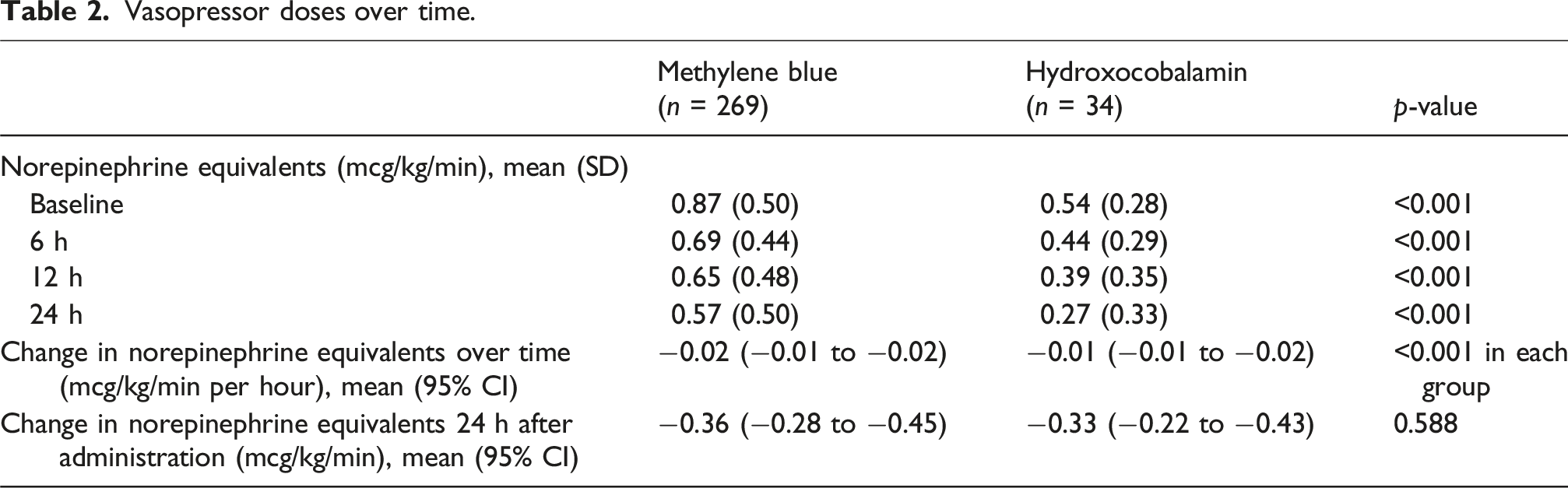
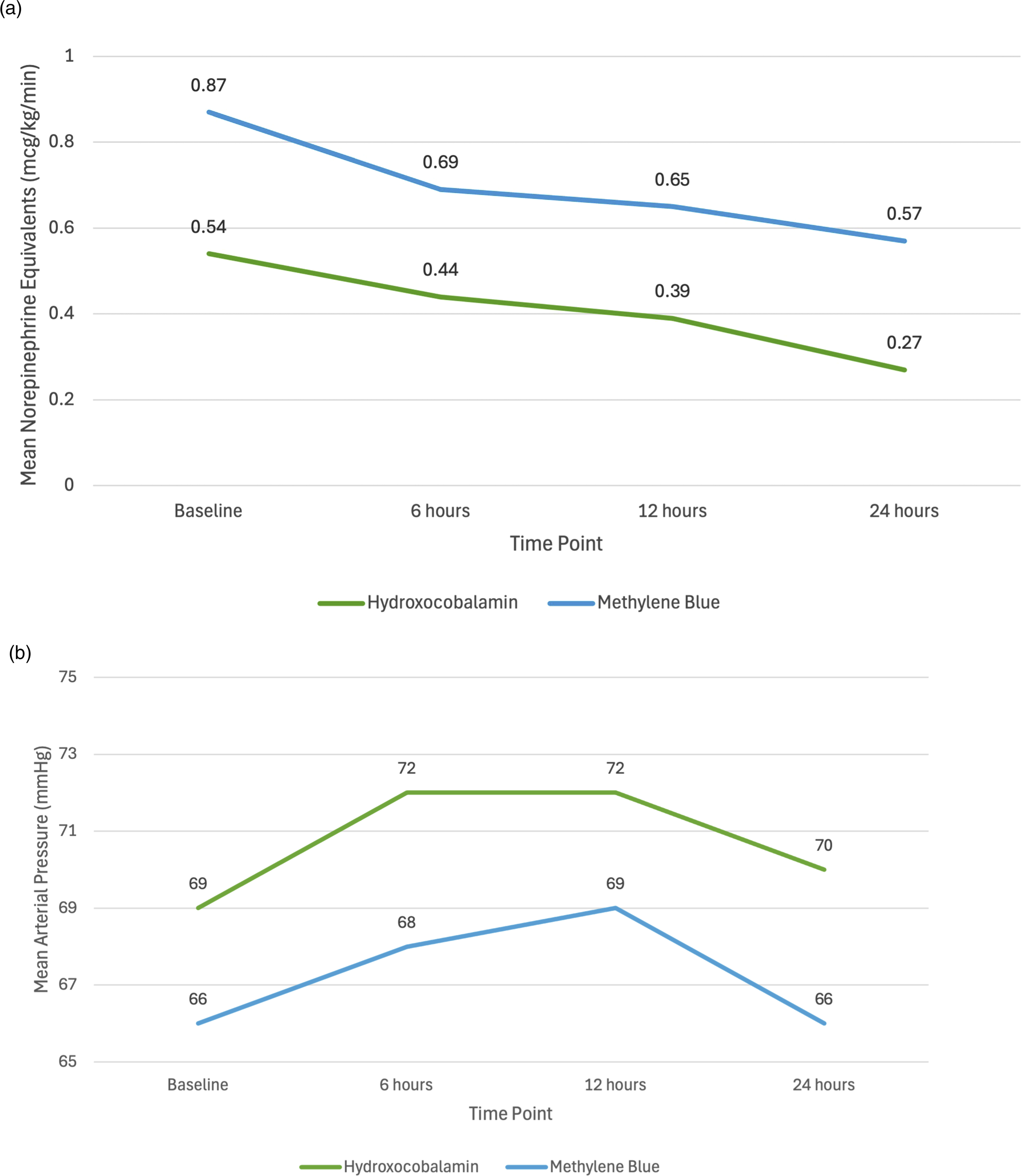
Vasopressor Doses and Mean Arterial Pressure Over Time.
A significant difference in baseline vasopressor doses was observed in a subgroup analysis by shock type (p < 0.001), with patients in septic or mixed shock receiving the highest vasopressor doses (Supplement Table 3, Supplement Figure 1). However, there was no significant difference in reduction in vasopressor doses across shock subtypes over time (p = 0.85) or between agents used in each shock type (p = 0.38). Additionally, there was no significant difference in reduction in vasopressor doses over time between agents when adjusted for differences in QSOFA (p = 0.69), use of positive inotropes (p = 0.07), multiple administrations (n = 0.74), or number of vasopressors at baseline (p = 0.77). Among patients on mechanical circulatory support, hydroxocobalamin was associated with a significantly greater reduction in vasopressor doses over 24 h (0.40 mcg/kg/min vs 0.23 mcg/kg/min, p = 0.018, Supplement Figure 2). Furthermore, patients who received initial weight-based doses of 2 mg/kg of methylene blue achieved a significantly greater reduction in vasopressor doses than those who received fixed 50–100 mg (0.34 mcg/kg/min vs 0.21 mcg/kg/min, p < 0.001) or 1 mg/kg dosing (0.34 mcg/kg/min vs 0.16 mg/kg/min, p = 0.016, Supplement Figure 3).
Secondary outcomes.
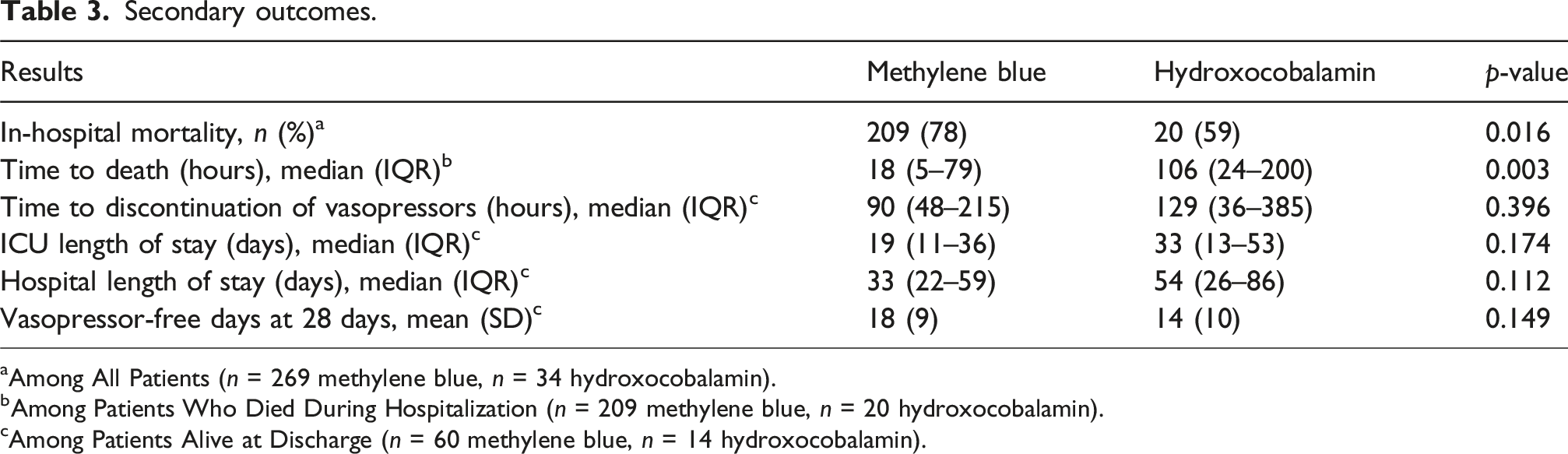
aAmong All Patients (n = 269 methylene blue, n = 34 hydroxocobalamin).
bAmong Patients Who Died During Hospitalization (n = 209 methylene blue, n = 20 hydroxocobalamin).
cAmong Patients Alive at Discharge (n = 60 methylene blue, n = 14 hydroxocobalamin).
A classification and regression tree analysis indicates that among patients with baseline vasopressor doses ≤0.81 mcg/kg/min NEE, those who are able to achieve a reduction in vasopressor doses to ≤0.31 mcg/kg/min NEE at 24 h have a higher odds of survival (51% vs 12%, odds ratio 7.6 [95% CI 3.1–18.2]). Similarly, among patients with baseline vasopressor doses greater than 0.81 mcg/kg/min NEE, those who are able to achieve a reduction in vasopressor doses to no more than 0.31 mcg/kg/min NEE at 12 h have a higher odds of survival (64% vs 4%, odds ratio 43.1 [95% CI 9.4–196.7]), which decreases to only 1% survival if vasopressor doses remain greater than 0.51 mcg/kg/min NEE at 24 h (Supplement Figure 4).
Discussion
In this large retrospective cohort study at two academic health systems, we found that both methylene blue and hydroxocobalamin were associated with similar and significant reductions in vasopressor doses 24 h after administration while maintaining a minimum MAP. Vasopressor load, which has been identified as one of the strongest predictors of mortality in refractory shock, was significantly reduced by both agents in this study at each time point, suggesting that the two agents were generally effective.5,6
In septic shock, hydroxocobalamin has demonstrated significantly greater reductions in vasopressor doses compared to placebo. 19 Similarly, methylene blue has demonstrated significantly shorter time to vasopressor discontinuation and greater vasopressor-free days at 28 days compared to placebo. 18 In vasoplegic shock, Hiruy et al. demonstrated significantly greater reductions in vasopressor doses with hydroxocobalamin compared to methylene blue, while Kram et al. found no difference between the two agents.17,20 We did not observe a difference in vasopressor reduction between shock types. However, at both centers, methylene blue is often preferred for septic shock, while hydroxocobalamin is more commonly used in cardiogenic and vasoplegic shock. The lack of difference seen overall with either agent may be due to exclusion of intraoperative administration, whereas Hiruy et al. studied vasoplegic shock associated with cardiopulmonary bypass, with primarily intraoperative administration of either agent. 17 Notably, our patients on mechanical circulatory support were able to achieve significantly greater reductions in vasopressor doses with hydroxocobalamin, which may support continued use of this agent among these patients. Previous studies have demonstrated the efficacy of methylene blue and hydroxocobalamin for vasoplegia with ECMO in particular.22,23
Furthermore, among patients who receive methylene blue, our findings suggest that higher weight-based doses may be required to achieve greater reductions in vasopressor doses. This is in line with the results of a dose-finding study in septic shock, which demonstrated that methylene blue had a dose-dependent effect on cardiac index, mean arterial, mean pulmonary artery, and pulmonary artery occlusion pressures, left ventricular function, oxygen delivery and consumption, and lactate levels. However, it is important to weigh the potential benefits of higher doses with the risk of splanchnic ischemia, although that study found that doses between 1–3 mg/kg do not impair splanchnic perfusion. 24
Four trials across patients with septic and vasoplegic shock have not demonstrated a significant mortality benefit with either agent despite benefits in vasopressor doses and hemodynamics.17–20 In vasoplegic shock, Hiruy et al. found hospital mortality rates of 21% and 12% with methylene blue and hydroxocobalamin, respectively. 17 In septic shock, Ibarra-Estrada et al. found a 33% 28-days mortality rate with methylene blue while Patel et al. found a 40% hospital mortality rate with hydroxocobalamin.18,19 Although mortality rates were higher in our study, particularly among the patients who received methylene blue, this may be explained by greater illness severity as evidenced by the higher QSOFA scores and vasopressor doses at baseline.
In fact, our entire cohort was extremely ill. Given that there is no consensus definition of refractory shock, we followed the threshold of the ATHOS-3 trial and included patients with baseline vasopressor doses of at least 0.2 mcg/kg/min NEE to evaluate the role of these agents across a wide range of shock severity and earlier in the development of refractory shock than other studies. While previous studies have included patients with baseline vasopressor doses between 0.29 and 0.59 mcg/kg/min NEE, our cohort had baseline vasopressor doses between 0.54 and 0.87 mcg/kg/min NEE, which may indicate that early initiation of these agents is a critical part of improving patient outcomes.17–20 The classification and regression tree analysis suggests that baseline vasopressor doses of ≤0.81 mcg/kg/min NEE may be the threshold where the greatest mortality benefit is demonstrated with these agents. Furthermore, a rapid response to treatment with a reduction in vasopressor doses to less than 0.31 mcg/kg/min NEE is crucial, as failure to achieve this threshold is associated with a very low chance of survival regardless of baseline vasopressor doses. To our knowledge, this is the first study that has conducted such analysis to establish optimal NEE prognostic values at major time-points. However, these thresholds are purely observational, and it is possible that patients whose vasopressor doses rapidly decline are more likely to survive, and not necessarily that a drop in those doses directly improves survival.
This study has several limitations. We evaluated a large cohort of patients with varying shock severity and subtypes, with a classification of refractory shock that differs from some studies. Given the retrospective observational design, this study may be susceptible to selection bias. There were significant differences in baseline characteristics between the two treatment groups, with a more critically ill cohort of patients in the methylene blue group, as evidenced by higher baseline vasopressor doses and QSOFA scores, although QSOFA scores may have more limited utility in comparison to SOFA scores in the ICU. Methylene blue was also used more commonly among patients with septic shock, while hydroxocobalamin was used more commonly among patients with cardiogenic and vasoplegic shock. It is possible that there were other external factors that prompted clinicians to favor one agent over another that we did not account for, such as G6PD deficiency or concomitant serotonergic agents. Additionally, since there was no placebo comparator group or evaluation of the trajectory of vasopressor doses prior to administration, it is possible that some patients would have improved naturally over time as a result of other interventions and without these rescue agents. Potential confounding factors include the implementation of other critical therapies such as antibiotics, fluid resuscitation, renal replacement therapy, and modes of mechanical circulatory support. Statistical adjustments for multiple comparisons were not applied, which may increase the risk of Type I error. It is also possible that we were unable to detect a small difference in efficacy between methylene blue and hydroxocobalamin given the limited sample size among the hydroxocobalamin group and heterogeneity of shock severity. Analyses of the vasoplegic, hypovolemic, and mixed shock subgroups were particularly susceptible to Type II error given the small sample sizes. Notably, patients with septic shock accounted for more than half of our patient population. However, our study saw no difference in vasopressor reductions when adjusted for differences in shock type, QSOFA, use of positive inotropes, multiple administrations, or number of vasopressors at baseline. Furthermore, at our centers, different protocols are used for the administration of methylene blue, including continuous infusions following the bolus in select cases of septic shock at one center, which may vary from practice at other institutions.
Randomized controlled trials are needed to validate these findings. Given the retrospective observational nature of this study, our results are largely hypothesis-generating. Future research could investigate alterations in vasopressor doses and hemodynamics following consecutive use of both agents or further investigate the importance of timing of administration of methylene blue or hydroxocobalamin in relation to the initiation of vasopressors.
Conclusion
In this retrospective comparative study evaluating methylene blue and hydroxocobalamin for the treatment of refractory shock, both agents were associated with significant reductions in vasopressor doses while maintaining a minimum mean arterial pressure. However, no significant difference was observed between the two agents.
Supplemental material
Supplemental material - A two-center comparison of the efficacy of methylene blue and hydroxocobalamin for the treatment of refractory shock
Supplemental material for A two-center comparison of the efficacy of methylene blue and hydroxocobalamin for the treatment of refractory shock by Natalie Y. Leung, Charlene Dinh, Lee Nguyen, Mira Bakas and Wan-Ting Huang in Perfusion
Footnotes
Ethical considerations
The study was approved by the Institutional Review Boards of the respective institutions (UCSD IRB 808980 and UCI IRB 3943), with the need for written informed consent waived.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
