Abstract
Acute coronary syndrome (ACS) in a patient with acute adrenal insufficiency (AAI) is rare and presents a diagnostic challenge to the treating physician due to its uncharacteristic presentation. AAI is an uncommon but serious condition presenting with vomiting, low blood pressure and weakness. The outcome of AAI depends on early treatment with corticosteroids. Long-term use of corticosteroids is an independent risk factor for cardiovascular disease. Corticosteroids induce a chronic inflammatory response of the vasculature, leading to atheroma formation and progression. ACS is a major cause of hospital admission and death in patients with long-term use of steroids. The most common cause of ACS is atherothrombosis. Optical coherence tomography (OCT) is an intravascular imaging modality that provides a detailed picture of atheromatous plaque morphology due to its high resolution. We hereby present a case of ACS resulting from a plaque rupture in a patient with AAI and the utility of intravascular imaging in the assessment of the underlying mechanism of ACS.
Keywords
Introduction
Acute coronary syndrome (ACS) results from plaque rupture, plaque erosion, vasospasm, or embolism. Long-term steroid use leads to chronic inflammation of the vascular wall, thereby contributing to atheroma formation and progression. Sudden withdrawal of steroids results in acute adrenal insufficiency (AAI), which poses sudden hemodynamic stress on the underlying diseased atheroma, resulting in a possible rupture or erosion of a vulnerable plaque.1–3 OCT can best visualize these characteristics of plaque morphology. The concomitant presentation of ACS with AAI is a challenging diagnosis to make owing to the non-specific symptoms at presentation ranging from nausea, vomiting, fatigue, low-grade fever, etc. We hereby present a patient with AAI resulting in a coronary plaque rupture and non-ST elevation ACS.
Case Presentation
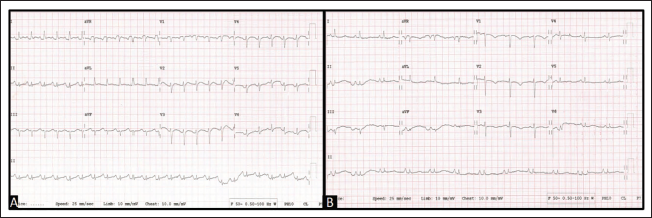
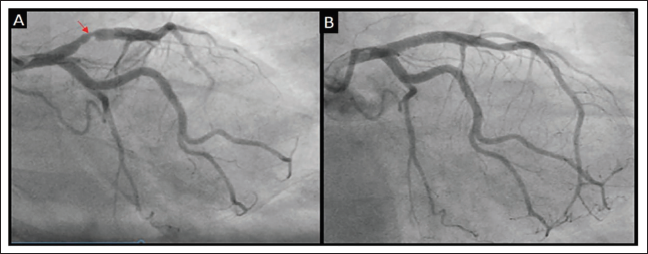
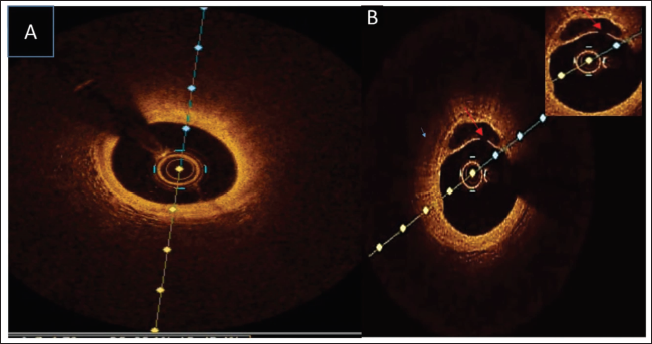
A 68-year-old woman presented with shortness of breath, vomiting, low-grade fever, and generalized weakness for 7 days. She was diagnosed with SLE (Systemic Lupus Erythematosus) and had been on oral steroids for 30 years, but stopped medication for 3 days. Her other comorbidities include hypertension and primary hypothyroidism. At admission, she was drowsy with tachycardia (pulse rate: 110/min, regular) with a blood pressure of 100/70 mm Hg. Laboratory reports upon admission, including electrolytes, blood glucose, lipid profile, and renal function tests, were within normal limits. Her electrocardiogram (ECG) on admission showed sinus tachycardia with poor R-wave progression and prolonged QTc interval (Figure 1A). A repeat ECG done 2 days later, after tachycardia settled, showed T-wave inversions in leads V2-V4 (Figure 1B). On echocardiogram, there was no regional wall motion abnormality with normal LV function. High-sensitive troponin-I was elevated (Basal sample: 831 pg/mL and 3rd-hour sample: 3,969 pg/mL, with a delta change of 378%), and NT-ProBNP was 240. These findings were consistent with ACS-NSTEMI. A coronary angiogram was done which revealed a proximal LAD 70% eccentric lesion (Figure 2A). Optical coherence tomography (OCT) imaging was done which showed a fibro-fatty lesion with a ruptured plaque in proximal LAD with no visible thrombus in the cavity (Figure 3). Coronary angioplasty (PTCA + drug-eluting stent) was done to proximal LAD using a 4 × 18 mm everolimus-eluting coronary stent system (Xience V; Abbott Vascular, CA, USA) (Figure 2B). As she was drowsy post-procedure and had hypotension (BP was 80/50 mm Hg) she was started on inotropes after adequate hydration and serum cortisol was done which was low (0.59 µg/dL and 0.38 µg/dL; normal range: 5-25 µg/dL). Based on this, a diagnosis of Addison’s crisis was made. The crisis occurred because steroids were not taken for 3 days. She was started on intravenous hydrocortisone and later switched to oral steroids (prednisolone, 1 mg/kg) as advised by an endocrinologist. Subsequently, there was a prompt improvement in sensorium, blood pressure, and general condition within 12 hours. She had an unremarkable hospital stay thereafter and was discharged on a tapering regimen of steroids.
(A) Initial ECG on Admission Showed Sinus Tachycardia with QTC Prolongation and Poor R-wave Progression. (B) ECG Taken After a Few Hours Showed T-wave Inversion in V1-V3 and Poor R-wave Progression.
(A) Coronary Angiogram (RAO Caudal View): Red Arrow Showing Proximal LAD Eccentric 70% Lesion. (B) Post-coronary Angioplasty (PTCA + DES) to LAD.
(A) Optical Coherence Tomography (OCT) Image Showing a Fibro-lipid Plaque. (B) OCT Image: Red Arrow-showing Ruptured Plaque.
Discussion
Coronary artery disease (CAD) is one of the leading causes of hospital admission and death worldwide. Atherosclerosis is one of the pathological basis of CAD. It makes the coronary arteries rigid and narrow, leading to obstruction. The initiation, progression, and destabilization of the atheromatous plaque are due to an underlying inflammatory process. 4 ACS is the most serious complication of chronic inflammation of the vascular wall, with a high mortality rate. 4 Endothelial dysfunction is currently considered the triggering factor in the pathogenesis of atherosclerotic plaque. Pro-inflammatory cytokines like TNF-alpha (tumor necrosis factor) 4 and IL-1 beta (interleukin 1) alter the endothelium by initiating a chain of events. Insult to endothelial permeability leads to accumulation of LDL, attracting circulating macrophage/monocytes and forming thrombi due to an increase in pro-aggregate and procoagulant function. 5
Addison’s crisis can either be primary or secondary. Primary adrenal insufficiency (Addison disease) is due to an underlying disease of adrenal glands and is associated with decreased cortisol and elevated ACTH. In secondary adrenal insufficiency, there is low cortisol and low to normal ACTH due to low ACTH production from the pituitary gland. Cortisol, a hormone that helps the body respond to stress, is regulated in the central nervous system. The adrenal gland produces cortisol through ACTH, which is from pituitary gland on stimulation from the hypothalamus. A common presentation of Addisonian crisis occurs with long-term steroid use, which suppresses the HPA axis. 3 It has a high mortality rate if not treated appropriately.
In patients with AAI, the diagnosis of ACS is difficult. During Addisonian crisis, inferior wall MI is most common. It can sometimes manifest with electrical disturbance like prolonged QT interval, ST-T changes, and rarely presents with dilated cardiomyopathy or stress cardiomyopathy. Prolongation of the QT interval resulting in torsade de pointes and cardiac arrest has been described in patients with adrenal insufficiency. 6 The QT interval in these patients was normalized with the administration of replacement glucocorticoids. In our case, ECG showed QT prolongation, as discussed above, with ST-T changes. The echocardiogram was normal, and hs-troponin-I was elevated with significant delta change, suggestive of ACS (NSTEMI). A strong suspicion of plaque rupture was thought of and a coronary angiogram was done with OCT imaging, which showed a 70% eccentric lesion with ruptured atherosclerotic plaque in proximal LAD.
The treatment of Addison’s disease includes glucocorticoids. Glucocorticoids stimulate atherogenesis and vascular remodelling 7 via glucocorticoid and mineralocorticoid receptors, a phenomenon which involves the local enzyme 11β-hydroxysteroid dehydrogenase. 8 In the case of long-duration cortisol treatment, there is a decrease in the production of nitric oxide, prostacyclin and an increase in thromboxane production in endothelial cells, creating an imbalance between vasoconstrictors and vasodilators.9, 10 In this scenario, she was diagnosed with SLE 30 years back and was on treatment with prednisone. Long-term use of glucocorticoids might have led to ACS, as they are known to trigger cardiovascular events. 11
This case shows an interesting scenario wherein a patient on chronic corticoid substitution with an underlying atherosclerotic burden, an acute episode of Addison’s crisis may lead to ACS. In our case, a crisis occurred because steroids were not taken for 3 days and the event was due to atherosclerotic plaque rupture as seen in intravascular imaging. The underlying pathology which led to acute events could be due to abrupt hemodynamic changes and shear stress leading to endothelial dysfunction.
Conclusion
In a patient on chronic steroid therapy, pre-existing coronary atherosclerosis can lead to ACS upon the sudden withdrawal of steroids. Severe fluid loss and low blood volume status seen in adrenal insufficiency can be the pathological basis of ACS, as in our case report. Intravascular imaging helps in the accurate identification of pathology and guides an optimal treatment strategy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
