Abstract
Previous studies have shown that plaque rupture (PR) is the most important cause of acute coronary syndrome (ACS). However, the potential impact of intra-stent PR on stent failure manifesting as ACS and the optical coherence tomography (OCT) features of these lesions was still less clear. Therefore, we aimed to investigate the characteristics of OCT-defined in-stent PR and identify the potential risk factors for PR in ACS patients. All 213 ACS patients were stratified into two groups according to the occurrence of in-stent PR. The baseline characteristics of all patients were investigated. A multivariable logistic regression was performed to identify factors associated with the formation of PR. OCT findings showed that 80 patients had in-stent PR, and 133 patients were without PR. Patients who suffered in-stent PR had higher blood lipid levels (P < .050) and longer duration of stent failure (P = .001). Moreover, thrombolysis in myocardial infarction (TIMI) 0/1 flow was more prone to the formation of PR (P = .010), and lesions in the PR group harbored more macrophages and cholesterol crystals (P < .001 and P = .024). On multivariate analysis, presentation of myocardial infarction (MI) and OCT findings of lipidic neointima length showed a 3.6-fold (P = .004) and 1.3-fold (P < .001) higher risk for occurrence of in-stent PR. ACS patients who suffered in-stent PR had a longer duration of stent failure and more ISR occurring in non-culprit vessels. Given the distinguishing features between ACS patients with in-stent PR and non-PR, potential targeted therapy was warranted to improve the prognosis of patients with in-stent PR.
Keywords
Introduction
Patients undergoing percutaneous coronary intervention (PCI) with newer-generation drug-eluting stent (DES) implantation still experience late or very late post-implantation stent failure that presents as in-stent restenosis (ISR) or stent thrombosis. 1 The main mechanical factors contributing to stent failure are under expansion or fracture, and the main biologic factors include local inflammation, aggressive neointimal proliferation, late acquired malapposition, or neoatherosclerosis.2,3 Meanwhile, stent failure is also one of the main mechanisms of the acute coronary syndrome (ACS). The prevalence of ACS in the setting of ISR can be as high as 60% to 70%, with a myocardial infarction (MI) prevalence of 5% to 10%. 4 It is vital for the individualized treatment and secondary prevention of ACS patients to clarify its potential pathophysiological mechanisms and specific characteristics. Histopathologic and optical coherence tomography (OCT) studies have shown that plaque rupture (PR) is the most crucial cause of ACS in non-stent vessels, 5 however, the potential impact of PR in the setting of ISR or stent thrombosis and the detailed OCT features in those lesions is less clear.6,7 Therefore, we aimed to investigate the incidence and characteristics of OCT-defined neointimal PR in ACS patients and identify the potential risk factors for ACS patients with late stent failure.
Methods
Study Design and Eligible Criteria
This was a retrospective study, and 213 ACS patients from the Second Affiliated Hospital of Harbin Medical University were included between Jan 2018 and Dec 2020. The inclusion criteria were as follows: age > 20 years; late and very late (>30 days from implantation) symptomatic DES failure irrespective of clinical presentation; and sufficient OCT images obtained at the time of stent failure. Exclusion criteria for OCT examination were angiographically left main disease; coronary artery bypass grafting (CABG); congestive heart failure or cardiogenic shock; renal insufficiency with a baseline serum creatinine level >1.5 mg/dL or poor OCT image quality. According to the results of OCT images, we also excluded patients who suffered secondary PR caused by thrombectomy or predilatation. ST-elevation MI (STEMI) was defined as continuous chest pain lasting >30 min, arrival at the hospital within 12 h from symptom-onset, ST-segment elevation > 0.1 mV in >2 contiguous leads or new left bundle-branch block, and elevated cardiac markers (creatine kinase-MB or troponin T/I). Non-ST-elevation MI (NSTEMI) was defined as ischemic symptoms in the absence of ST-elevation with elevated cardiac markers. Unstable angina pectoris (UAP) was defined as newly developed/accelerating chest symptoms on exertion or rest angina within 2 weeks.8,9
The baseline characteristics included medical history, clinical and angiographic data, and recent medications were collected. Patients were routinely treated with aspirin, clopidogrel (or ticagrelor), and unfractionated or low-weight heparin according to current international guidelines. The protocol was approved by the Ethics Committee and Institutional Review Board of the Second Affiliated Hospital of Harbin Medical University, and informed consent forms were obtained from all included patients.
Cardiac Catheterization and Angiographic Analysis
Potential strategies to treat the lesions due to stent failure were decided by the operators. Manual aspiration thrombectomy (Export aspiration catheter; Medtronic, Santa Rosa, CA, USA) before OCT imaging in patients with a sizable occlusive thrombus or TIMI flow grade <2 was allowed. Stent failure included ISR and stent thrombosis. ISR was either ≥50% luminal narrowing at follow-up angiography with symptoms or signs of ischemia (at rest or with stress) or ≥70% luminal narrowing at follow-up angiography without clinical symptoms or signs. Definite stent thrombosis was defined according to the Academic Research Consortium criteria. 10
Coronary angiography at the time of stent failure was performed using guiding catheters after intracoronary injection of 100–200 μg of nitroglycerin. Angiographic images were analyzed using CAAS 5.10.1 software (Pie Medical Imaging BV, Maastricht, the Netherlands) at the intravascular imaging and physiology core lab in the Second Affiliated Hospital of Harbin Medical University. Lesions’ location and length, minimal lumen diameter (MLD), diameter stenosis (DS), and initial and post-procedure TIMI flow were measured by an independent investigator (T.C.) who was blinded to clinical and OCT data. Detailed analytical methods were mainly referred to previous studies.11,12
OCT Image Acquisition and Analysis
OCT images were captured using frequency-domain OCT (C7-XR, ILUMIEN, or ILUMIEN OPTIS OCT imaging systems; Abbott Medical) at a pullback speed of 20–25 mm/s. 13 Each frame (every 0.2 mm) was analyzed by two independent investigators (H.Y. and H.L.), who were blinded to baseline characteristics and angiographic findings of patients by using proprietary software (Abbott Medical) after confirming the calibration settings.
Stent and intra-stent lumen cross-sectional area (CSA) was measured at the minimum lumen area (MLA) site to include the minimum stent area (MSA) and maximum neointimal hyperplasia (NIH) CSA. Stent CSA was measured by joining the middle points of the strut blooming artifacts. If the stent was covered by high signal attenuation tissue, stent CSA was delineated using proximal and distal slices. The percentage of NIH was calculated as ([stent–lumen CSA]/stent CSA) × 100. Proximal and distal reference lumen CSAs were the slices consisting of the largest lumen CSA within 5 mm proximal and distal to the stent edges before significant side branches (>1.5 mm in diameter).
Under expansion was defined as the ratio of in-stent MLA to average reference area <80%. 14 Neointimal tissue at the intra-stent MLA site was assessed qualitatively to characterize the pattern as neoatherosclerosis versus non-neoatherosclerosis, homogeneous versus heterogeneous, or mean neointimal thickness <100 μm. Neoatherosclerosis was defined as NIH that contained lipid (the intra-NIH signal-poor region with diffuse borders), calcification (intra-NIH signal-poor area with a well-delineated border), or neointimal rupture (disruption in the fibrous cap connecting the lumen with the underlying NIH).15–17 In-stent thin-cap fibroatheroma (TCFA)-like neointima was defined as lipidic neointima with fibrous cap thickness ≤65 μm. Homogeneous restenosis showed uniform signal properties (high or low intensity) without focal variation or attenuation. Heterogeneous restenotic tissue had variable optical properties and backscattering patterns and was divided into layered (concentric layers with an endoluminal high backscattering layer and an abluminal layer with low backscattering optical properties) or nonlayered. Macrophage accumulation was high-intensity, signal-rich, linear regions with sharp attenuation and a narrow trailing shadow that changed frame by frame. A malapposed strut had a measured distance between its inner surface and the adjacent surface of the vessel greater than the sum of the thickness of the strut plus polymer. Coronary evagination was the presence of an outward protrusion of the lumen between well-apposed struts with a maximum protrusion depth >10% of the mean stent diameter within the same frame. Stent fracture was a separation of the stent into two pieces with ≥120° of the stent circumference without visible struts.18,19
Based on OCT findings, primary PR was identified by a fibrous cap discontinuity, creating communication between a cavity within the plaque and the arterial lumen. The hallmarks of primary PR were a thin fibrous cap, large lipid core, and fibrin-rich thrombus. However, most of the secondary PR caused by thrombectomy or predilatation came from plaque erosion (PE) which was characterized by a plaque covered by a thrombus (typically a white, platelet-rich thrombus), a thick intima, and a small or absent lipid core. 20
Statistical Analysis
The Kolmogorov–Smirnov normality test was performed to examine whether the data showed normal distribution. Continuous variables were denoted by mean ± SD if normally distributed, and the difference between groups was tested using an independent sample t-test. Skewed data were expressed as median and range, and comparisons were performed using the Mann–Whitney U test. Categorical variables were reported as numbers and percentages, and the Pearson chi-squared test was used to compare categorical variables.
A multivariable backward stepwise logistic regression approach was used to examine the relationship between potential risk factors and PR in ACS patients. A two-sided P-value of <0.05 was deemed to be statistically significant. Analyses were performed using SPSS software version 18.0 (SPSS Inc., Chicago, Illinois, USA).
Results
Demographic and Clinical Characteristics
A total of 213 ACS patients were enrolled in this study, and all underwent OCT imaging at the time of stent failure manifesting as ACS. 10 patients had very late stent thrombosis (VLST) without NIH, while the remaining 203 patients presented with NIH-related ISR. All 213 ACS patients consisted of 78 (36.6%) STEMI, 58 (27.2%) NSTEMI, and 77 (36.2%) UAP. Overall, 80 (37.6%) had OCT-identified neointima PR, and 133 (62.4%) were without PR. In the PR group, all 80 patients were classified as in-stent PR in this study. The follow-up time in this study was from the index procedure to stent failure. Follow-up was conducted by telephone or clinical visit every 3 months after the index procedure. Given the unpredictable nature of coronary accidents, coronary angiography was systematically advised at any time in case of suspected developments of stent failure manifesting as ACS. The median follow-up duration was 8.7 years (IQR: 6.3 to 12.5 years) for patients with PR and 6.9 years (IQR: 4.8 to 10.3 years) for patients without PR.
The baseline characteristics of all patients at the time of ACS presence were illustrated in Table 1. There was no statistical difference between the two groups in terms of gender, age, weight, history of diabetes, hypertension, chronic kidney disease or smoking, and other risk factors. Overall, more than 90% of stents were the second-generation DES or DES with biodegradable polymer. The demographic data showed insignificant differences between the two groups except for the duration of stent failure and clinical presentation. Patients diagnosed with STEMI were more prone to the formation of PR (P < .001). As for laboratory data, patients who suffered PR had significantly higher total cholesterol (TC), low-density lipoprotein-cholesterol (LDL-C), and high-sensitivity C-reactive protein (hs-CRP) (all P < .05). Moreover, the patients in the PR group were also with significantly higher peak creatine phosphokinase-MB (CK-MB) and cardiac troponin I (cTnI). Medications of antiplatelet therapy were similar between the two groups.
Clinical Characteristics of Patients with or Without Plaque Rupture in the Presence of the Acute Coronary Syndrome.

Values expressed as n (%), mean ± SD, or median (range).
CKD, chronic kidney disease; HDL-C, high-density lipoprotein cholesterol; hs-CRP, high-sensitive C-reactive protein; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; PR, plaque rupture; TC, total cholesterol; TG, triglyceride
Angiographic Findings
Angiographic and procedural characteristics of patients at the time of stent failure in the presence of PR were shown in Table 2. The left anterior descending artery (LAD) was the prominent location of the culprit's vessel in both groups. People who suffered PR were also more likely to have a longer duration of stent failure (P = .001). Overall, no significant difference existed in the location of PR versus non-PR lesions either by vessel or distribution (proximal vs non-proximal). The types of stents were similar between the two groups (P = .080), while the characteristics of lesions before angiographic intervention were also comparable between the two groups (all P > .05). Importantly, TIMI 0/1 flow was most common in the PR group, while TIMI 3 flow often occurred in patients without PR (P = .010). What's more, the incidence of ISR in non-culprit vessels was also significantly higher in the PR group (P < .001).
Angiographic and Procedural Characteristics of Patients with and without Plaque Rupture.

Values expressed as n (%), mean ± SD, or median (25th–75th percentiles).
BMS, bare metal stent; DES, drug-eluting stent; ISR, instent restenosis; LAD, left anterior descending artery; LCX, left circumflex artery; MLD, minimal lumen diameter; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction
OCT Findings
A total of 133 lesions in 133 ACS patients without PR and 80 lesions in 80 patients in the PR group were quantitatively analyzed. The OCT morphometric data according to the presentation time in both groups were illustrated in Figure 1 and summarized in Table 3. The most common type of culprit plaque was the fibrous plaque in the non-PR group and the lipid plaque in the PR group. Neoatherosclerosis was seen in all patients with PR versus only 54.9% of patients without PR (P < .001). When compared with the non-PR group, TCFA-like neointima was more common in the PR group (P = .001). Meanwhile, the incidence of calcification was similar between the two groups (P = .238), and the arc and length of calcium (P = .080 and P = .162) were also similar. Furthermore, coronary artery plaque in the PR group harbored much more macrophages and cholesterol crystals (P < .001 and P = .024). However, there was no statistical difference in the presence of stent fracture, malapposition, or underexpansion between the PR and non-PR groups.
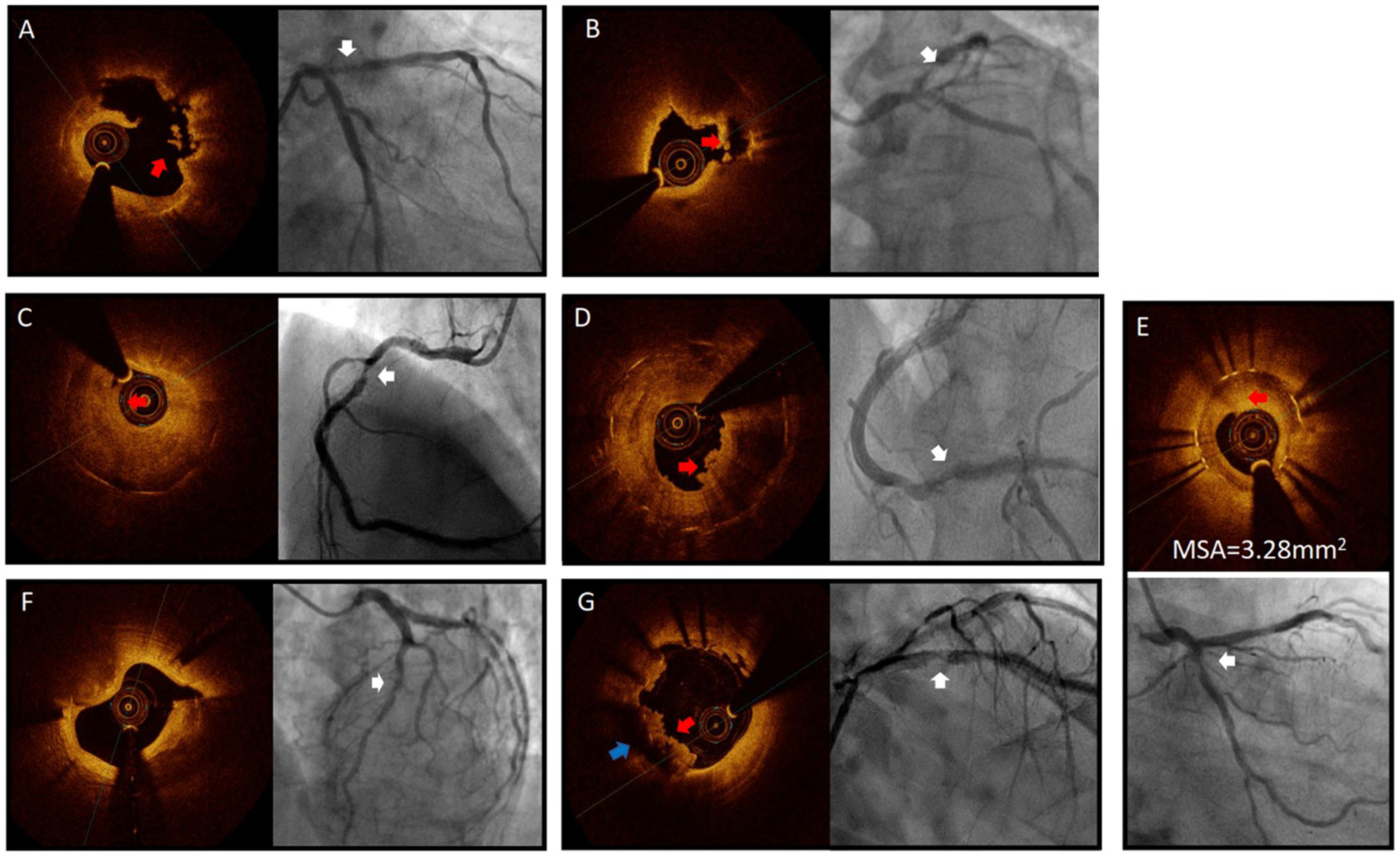
Mechanisms of stent failure in patients presenting with ACS (white arrows on the angiograms). (A) and (B) Plaque rupture (red arrows on the OCT with the blue arrow in (B) showing macrophage formation). Note the near absence of stent struts caused by high attenuation and backscattering of the lipidic neoatherosclerotic neointima. (C) and (D) Lesions with neoatherosclerosis, but without plaque rupture (the red arrow showed a thrombus attached to the vascular wall). (D) Stent underexpansion. (F) Calcified nodules. (G) Stent malapposition (blue arrow) with superimposed thrombus (red arrow) (Color online).
OCT Findings of in-Stent ACS Patients with and without Plaque Rupture.

Values expressed as n(%), mean ± SD. VLST, very late stent thrombosis; TCFA, thin cap fibroatheroma; NIH, neointimal hyperplasia.
Non-Culprit ISR Lesions
Overall, 48.8% (104/213) of all included patients had in-stent lesions in non-culprit vessels, and nearly half of them suffered ISR (44.2%, 46/104). The incidence of restenosis in secondary (non-target) stents was significantly higher in the PR group versus the non-PR group (65.9% vs 30.2%, P < .001) (Table 2). When compared with the non-PR group, longer neoatherosclerosis (P < .001), higher rate of thin cap fibroatheroma (TCFA) (P = .001), and thinner fibrous cap (P = .040) were also found in the PR group based on the OCT findings of ISR in non-culprit vessels (Table 3).
Predictors for PR in ACS Patients
Results of the multivariate analysis revealed that previous history of MI (combining STEMI and NSTEMI) and OCT findings of lipidic neointima length showed a 3.6-fold and 1.3-fold higher risk for occurrence of PR in all patients [(OR: 3.583, 95% CI: 1.493 to 8.599, P = .004) and (OR: 1.316, 95% CI: 1.187 to 1.459, P < .001)], however, fibrous cap thickness (OR: 0.992, 95% CI: 0.986 to 0.998, P = .005) was reversely correlated with the occurrence of neointimal PR in patients with ACS (Table 4).
Multivariable Model for Predicting Risk Factor of Plaque Rupture in ACS Patients.

Abbreviation: LDL, low-density lipoprotein cholesterol; MI, myocardial infarction; TCFA, thin cap fibroatheroma
Discussion
This study intended to investigate the incidence and characteristics of in-stent PR in ACS patients and identify the potential risk factors for patients with late stent failure. The findings of this study provided evidence for potential targeted therapy for ACS patients with PR, allowing for the distinguishing features compared with patients without PR. The results demonstrated that 72.0% of ACS patients suffered stent failure by around 5 years on average after stent implantation. Among them, 37.6% of patients have underlying neoatherosclerosis and PR, especially those presenting with STEMI/NSTEMI. In-stent neoatherosclerosis was frequently observed in patients with VLST, and in-stent PR was identified as the dominant pathological mechanism causing VLST in 30% of patients presenting beyond the 1-year time frame.
Intravascular imaging-initially with intravascular ultrasound (IVUS) and now with OCT has provided insights into the mechanisms of stent failure and differentiated ISR from stent thrombosis.21,22 However, little progress has been made in identifying the proper approaches to prevent and treat neoatherosclerosis, partly because there is no consensus on the interpretation of different optical patterns of neointima and their histopathological basis. 23
The results of this study elucidated that patients with neointima PR were associated with significantly higher indexes of cardiac injury and incidence of TIMI 0/1. Moreover, TC and LDL-C were also higher in the setting of PR, which was consistent with findings from a previous study indicating that stent age after DES implantation together with hyperlipidemia and the absence of statin therapy was regarded as the potential risk factors for PR. 24 According to our OCT findings, nearly 90% of patients in the PR group suffered OCT-defined thrombus, and the incidence of macrophage infiltration, as well as cholesterol crystals, was significantly higher in the PR group versus patients without PR. Thus, macrophage infiltration and cholesterol crystals might contribute to rupture-prone vulnerable neointima in patients with PR. Consistent with our results, previous studies also indicated that infiltration of foam cells into the neointima could be considered as the initial stage of neo-atherosclerotic plaque formation, but no differences were found in the level of neointimal calcium.23,25 Given this, neoatherosclerosis after DES implantation in patients with PR was likely to be associated with discriminated characteristics, such as vascular healing and inflammatory response.
On basis of previous research, once developed, neoatherosclerosis was with a soaring incidence of stent failure compared with stable neointima.26,27 The current study extended this observation by showing that the rupture of neointima may not only be related to stent failure but also triggered the recurrence of ACS, which referred to poor prognosis of PR patients. Elevated levels of lipids in serum and inflammation were the two most essential factors in the pathogenesis of atherosclerosis and PR, which have been proved in previous papers.28,29 Likewise, our study also reported the clinical significance of these indices in in-stent PR-related ACS patients, and there were also higher levels of serum lipids and more vulnerable plaque features in the PR group. Thus, certain laboratory data, such as TC, LDL-C, and hs-CRP levels, should be taken into vital consideration during clinical follow-up. In addition, the length of lipidic neointima and fibrous cap thickness were clarified to be closely related to in-stent PR and the recurrence of ACS in the current study. Most importantly, for those with a previous history of MI or longer lipidic neointima, a more aggressive lipid-lowering therapy (higher dose of statin or concomitant use of a new generation of lipid-lowering drugs, such as PSK9 inhibitors) and strict clinical or even radiographic follow-up for those potential high-risk patients were necessary to prevent them from severe adverse events (SAE).
This study had several limitations. First, this study included several different DESs. Moreover, clinical presentations and underlying lesion morphology were unavailable at the time of the original stent implantation(s). Second, manual thrombectomy or pre-dilatation with small-sized balloons was performed in a considerable number of patients, which might affect the underlying morphology of atherosclerotic lesions. However, the operators were experienced, and special care was taken to avoid extra damage to lesions. Third, the presence of a massive thrombus might reduce the ability of OCT to recognize the underlying plaque characteristics.
Conclusions
Compared with non-PR patients, ACS patients who suffered in-stent PR experienced a longer duration of stenting to stent failure and had more ISR occurring in non-culprit vessels. Allowing for higher blood lipids level and previous history of MI was associated with increased risk for the formation of neoatherosclerosis and PR in ACS patients, our findings provide suggestions for a potential optimized lipid-lowering treatment plan for ACS patients with in-stent PR. When considering the distinguishing features of OCT findings between patients with PR and non-PR, potentially targeted therapy is warranted to improve the prognosis of PR patients.
Footnotes
Author's Contribution
Conception and design: H.Y, B.Y.
Development of methodology: J.N.D, C.F, S.Q.J.
Acquisition of data: H.Y, J.N.D, C.F.
Writing, review, and revision of the manuscript: H.Y, J.N.D, C.F, B.Y. G.S.M
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study protocol was approved by the Institutional Ethics Committee of Harbin Medical University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China, National Natural Science Foundation of China (Grant Nos. 2016YFC1301100 and 81330033).
