Abstract
We describe an extremely rare case of fatal cardiac mucormycosis in a renal transplant recipient, clinically manifested by focal myopericarditis and ST-elevation myocardial infarction, due to contiguous mediastinal spread. Cardiac mucormycosis is seen in 0.2–1.2% of renal transplants and carries an unfavorable prognosis despite the timely diagnosis, intensive antifungal therapies, and surgical intervention.
Keywords
Introduction
We wish to report an extremely rare presentation of ST-elevation myocardial infarction and focal myopericarditis due to cardiac mucormycosis, resulting from an adjacent spread of mediastinal infection occurring in an immunosuppressed post-renal transplant patient. Despite newer antifungal therapies and early surgical intervention for infection source control, the clinical outcomes of cardiac mucormycosis remain unfavorable with a very high mortality rate.
Case Report
The patient was a lady in her late 40s who presented with symptoms of fever, productive cough, hemoptysis, and atypical angina for 1 month. Seven months prior to the present admission, she had received an ABO-compatible renal transplant for immunoglobulin (IgA) nephropathy-related chronic kidney disease (stage G5A2). She was receiving immunosuppressive therapy, namely, tacrolimus (serum tacrolimus level of 19.9 ng ml−1 [reference range: 5–10 ng ml−1]), mycophenolate mofetil, and prednisolone. She underwent computed tomography (CT) of the thorax prior to the present hospitalization, which revealed a left-sided mediastinal cystic collection, for which CT-guided aspiration was performed. The aspirated fluid culture and biopsy were both negative for microbial growth; however, in view of her immunosuppressed state, intravenous (IV) meropenem was started empirically.
Two days after hospitalization, she developed acute-onset breathlessness with hypoxic respiratory failure in the ward, and she was transferred to the intensive care unit (ICU). On arrival in ICU, she was clinically hypoxic (oxygen saturation at ambient air was 88%), tachycardic (heart rate: 120/min), had a blood pressure of 114/67 mmHg, had tachypnoea (respiratory rate: 30/min), and had decreased left lower lobe air entry on auscultation.
Her complete blood count showed mild anemia (hemoglobin: 9.4 g dl−1) with normal leucocyte and platelet counts (platelet count 308 × 10 3 /µl; white blood cell count 5,360/cm3). A 12-lead electrocardiogram (ECG) showed sinus tachycardia with ST depression and T wave inversion in lateral precordial leads. Cardiac biomarkers were elevated: creatinine kinase-MB: 48 mg dl−1 (<25 mg dl−1), troponin-I: 0.38 ng l−1 (<0.02 ng l−1), N-terminal prohormone of brain natriuretic peptide: 19,900 ng l−1 (<500 ng l−1). Her transthoracic echocardiography revealed structurally normal valves and chambers with lateral wall hypokinesia (left ventricular (LV) ejection fraction 50%) and mild-to-moderate pericardial effusion, with a cystic lung mass abutting the pericardium laterally. The cardiac coronary tomography angiography showed about 50% stenosis in the mid-segment of the left anterior descending artery, in close proximity to the cystic lung mass, and a repeat CT imaging of the thorax showed a significant increase (compared to the scan dated 1 month ago) in the cystic lesion (8.2 × 9.1 × 11.0 cm), with extension into the anterior aspect of the left lung and mediastinum medially (Figure 1). The mild-to-moderate left-sided pericardial effusion measuring 9–11 mm with peripheral enhancement was visualized, suggestive of focal myopericarditis.
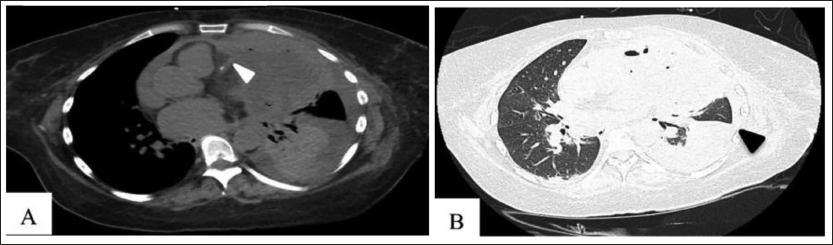
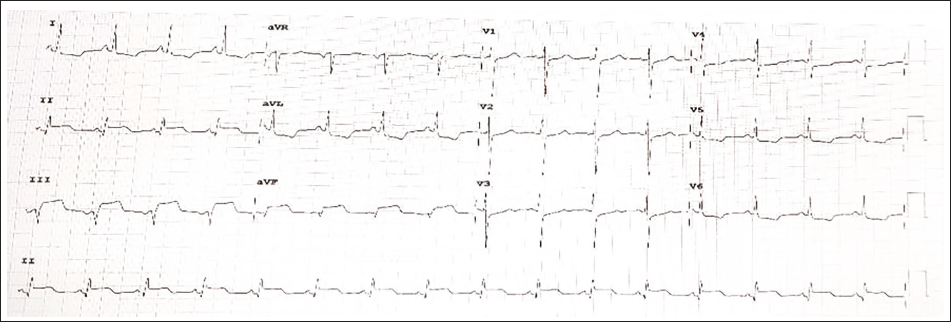
She underwent drainage of cystic mediastinal abscess via a left posterolateral thoracotomy approach. An invasive black necrotic mass was observed on gross examination. The patient reported mild chest pain on the left side 2 days after the procedure, and her ECG showed ST-segment elevation in leads II, III, aVF, and ST-segment depression in lateral leads (Figure 2); however, she remained hemodynamically stable.
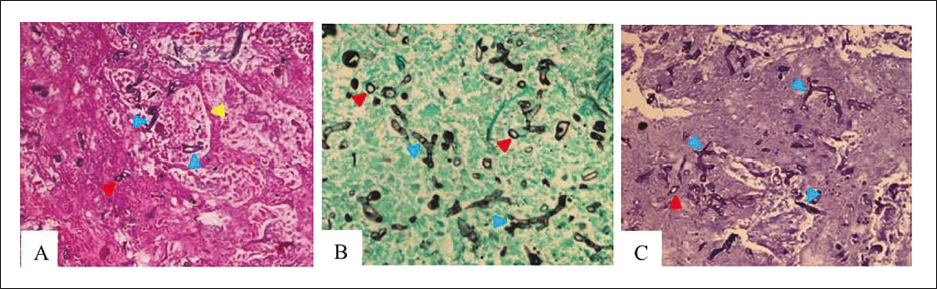
The histopathological examination of the mediastinal lesion revealed swollen branching minimally septate fungal hyphae, large infarct zones, lymphocytic and histiocytic infiltration (Figure 3). A diagnosis of disseminated mucormycosis was made, and IV liposomal amphotericin-B and posaconazole were started. The aerobic and anaerobic cultures revealed no microbial growth. The ST elevations on ECG resolved after 48 h of conservative treatment.
Histopathological Images. (A) Lung Parenchyma Histopathological Specimen (Hematoxylin and Eosin Stain), Showing Large Infarct Zones and Presence of Vascular Invasion (Yellow Arrowhead) by Minimally Septate Hyphae. Additionally, Foamy Cells, Clusters of Neutrophils, and Interstitial Edema Are Demonstrated. (B) Grocott Methenamine Silver. (C) Periodic–acid Schiff stains. The Swollen Branching Minimally Septate Hyphae (Blue Arrowheads) and Their Spore Forms (Red Arrowheads) Are Shown.
After initial improvement, her hospital course was complicated by febrile episodes and progressive dyspnea. A repeat CT scan of the thorax on day 7 showed an increase in the left upper and lower lobe collection (10.0 × 8.6 × 13.0 cm) with mediastinal compression. Antibiotics were escalated to ceftazidime-avibactam and aztreonam. In view of the respiratory compromise, a left lung pneumonectomy was performed.
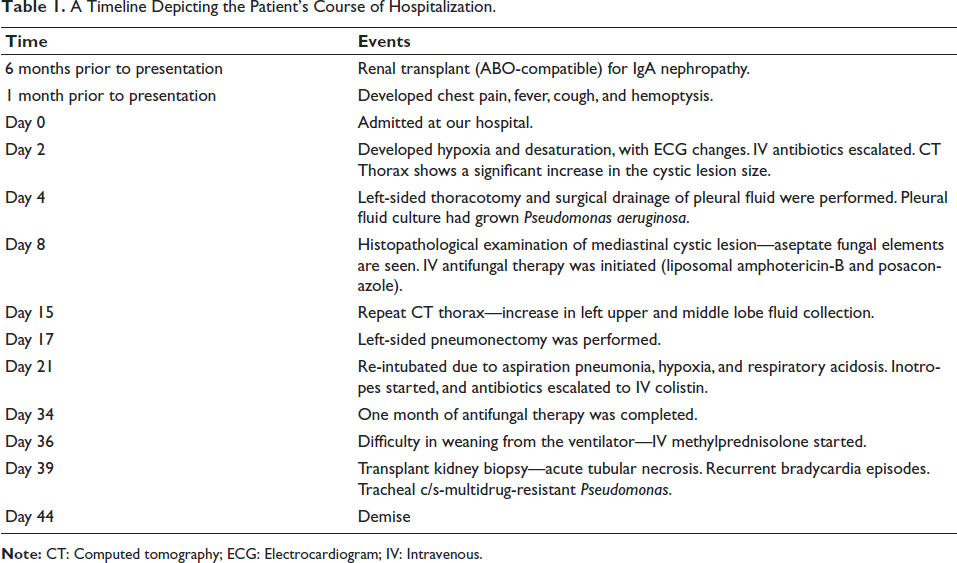
In the immediate postoperative period, she developed right lobar aspiration pneumonia and type 2 respiratory failure and was continued on mechanical ventilation. The rest of her ICU stay continued along a gradual downward spiral, with an inability to wean from ventilator support culminating in tracheostomy and development of anuria. She required frequent blood product transfusions for coagulopathy and persistent thrombocytopenia. Terminally, repeated episodes of bradycardia and persistent hypotension ensued. On the 44th day of hospitalization, she succumbed to her illness (Table 1).
A Timeline Depicting the Patient’s Course of Hospitalization.
Discussion
Mucormycosis is an invasive fungal infection with universally high morbidity and mortality rates. Conditions predisposing to invasive and disseminated infection include patients with neutropenia, innate immunity defects, those receiving prolonged corticosteroid therapy, and diabetic ketoacidosis. 1 The coronavirus disease 2019 pandemic has resulted in an increase in the incidence of mucormycosis infections. Our patient had multiple risk factors present, namely, immunosuppression, sepsis, and steroid-induced diabetes mellitus leading to persistent hyperglycemia, propagating uncontrolled fungal reproduction, and dissemination. The presence of multidrug-resistant Pseudomonas infection in the pleural cavity may have likely created a large reservoir for fungal growth. 2
Mucormycosis principally manifests as rhino-cerebral involvement, whereas cardiac manifestations are extremely rare and can be a sequela of disseminated disease or pulmonary infection that spreads adjacently to the pericardium and myocardium. 3 Cardiac mucormycosis in renal transplant patients has been sporadically reported, in which the incidence is between 0.2% and 1.2%.4, 5 Even with early treatment, cardiac involvement carries a poor prognosis.
Vasotropism is a hallmark of mucor infection, characterized by the ability of the hyphal elements to penetrate the vessel wall by adhering to and damaging the endothelial cells.5, 6 The angioinvasion and thrombosis lead to hemorrhagic tissue necrosis and infarctions. 5 Cardiac mucormycosis may manifest as arrhythmias, endocarditis leading to valvular dysfunction, myocarditis, pericarditis, and heart failure. Focal myocarditis may predispose to generalized myocardial inflammation. Septic infarction is a rare presentation.3, 7 Our patient presented with chest pain, which may be attributed to pericardial involvement. The development of a pericardial abscess following penetration of the left ventricular wall leading to a fatal myocardial rupture has been previously described. 8 Diffuse infiltration through the intra-atrial septum, both atria, and pulmonary veins from a primary mediastinal source has been reported. 9 Pathologically, the coronary arteries may be occluded with thrombi containing fungal hyphal elements, 10 leading to a transmural infarction.
The diagnosis of cardiac mucormycosis is made by observation of regional wall motion abnormalities on echocardiography and focal myocardial inflammatory involvement on cardiac CT or magnetic resonance imaging. In our patient, her critical condition precluded the performance of a coronary angiogram or endomyocardial biopsy for definite histopathological proof of cardiac invasion by mucormycosis.
Timely detection of mucormycosis infection is challenging due to frequently negative blood cultures; however, the presence of multi-organ-system infarcts should raise suspicion of disseminated mucor infection. 5 The cornerstone of mucormycosis treatment is effective antifungal therapy, surgical removal of the infection source, and control of predisposing risk factors. The antifungal agents available for the treatment of mucormycosis are liposomal amphotericin-B, posaconazole, and, most recently, isavuconazole.
In conclusion, our case demonstrates an unusual manifestation of disseminated mucormycosis with cardiac involvement. Cardiac evaluation should be considered in individuals with suspicion of mucormycosis who are immunocompromised. The multifaceted clinical features and inscrutable presentation can delay the diagnosis, resulting in poor outcomes. Therefore, a high index of suspicion, timely diagnosis, and aggressive medical treatment with optimal source control form the basis of the management of disseminated mucormycosis.
Ethical/IRB Approval
Ethical/IRB approval for this submission was not obtained, as it is a retrospective case report.
Footnotes
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
