Abstract
Coronary stent infection (CSI) is a rare but fatal complication of percutaneous coronary intervention (PCI) occurring in less than 0.1% cases. CSI usually manifests as fever and acute coronary syndrome but in some cases presentation may be confusing. Without prompt diagnosis and appropriate treatment, the outcome is poor with mortality reaching 30%. Here, we present 5 cases of CSI resulting in coronary artery aneurysms. Interestingly, CSI occurred during the first stent implantation, mostly in post-acute myocardial infarction patients. Blood culture, pericardial fluid analysis, and coronary angiogram were used as diagnostic tools in our cases. Three of our patients had infective endocarditis involving native mitral and aortic valves and one patient had pericardial effusion and cardiac tamponade. Stent thrombosis and aneurysm formation were observed in all cases. All patients received appropriate and adequate antibiotic therapy before undergoing coronary angiogram and surgery. Cardiac catheterization laboratory hardware reuse, repeated procedures, complex procedures requiring instrumentation, and prolonged indwelling catheters may increase the risk of bacteremia and stent infection. Currently, there are no established criteria or protocols for diagnosis and management of CSI.
Keywords
Introduction
The first reported case of coronary stent infection (CSI) was in 1993. The most common causative organism was Staphylococcus aureus (80%) followed by Pseudomonas aeruginosa (20%). Hospital acquired infections are more often encountered in patients who are admitted to critical care areas or in patients who undergo invasive procedures or surgeries.1, 2 Infection following percutaneous coronary intervention (PCI) and cardiac catheterization is known but extremely rare. PCI generally requires the insertion of multiple devices into arterial circulation and this may be responsible for development of bacteremia or even septicemia. 3 This risk is further increased with reuse of sterilized cath lab hardware and during complex interventions. Diagnosing CSI is challenging. Majority of patients present with fever and symptoms suggestive of acute coronary syndrome (ACS). Clinician should be aware of the possibility for early diagnosis. Delay in identifying symptoms may delay the diagnosis and treatment with disastrous consequences. Diagnostic criteria and guideline directed management strategies for CSI are yet to be established.
Case Summary
Case: 1
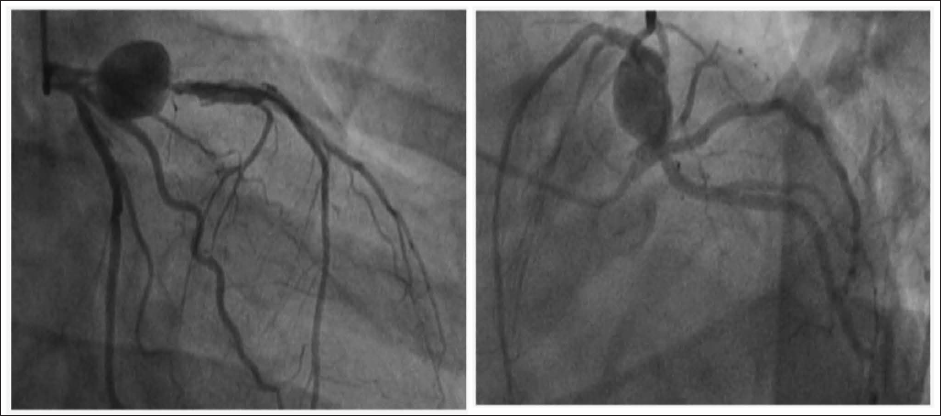
A 49-year-old male presented to a local hospital with chest pain. Electrocardiogram (ECG) was suggestive of evolving anterior wall myocardial infarction (STE AWMI) and was thrombolysed with 15 lakh units of Streptokinase. Four days later coronary angiogram (CAG) was done which showed 90% discrete lesion in proximal left anterior descending artery (LAD). Subsequently, he was subjected to percutaneous transluminal coronary angioplasty (PTCA) and stenting with drug eluting stent (DES) to LAD. Two weeks after PTCA, patient developed fever which was treated initially with oral antibiotics at local Hospital. Blood culture demonstrated growth of P. aeruginosa. Patient was brought to our hospital as fever continued inspite of antibiotics. Transthoracic echocardiography showed severe MR with large mobile vegetation on anterior mitral leaflet (AML). A diagnosis of Pseudomonas septicemia with infective endocarditis of native mitral valve was considered and initiated the treatment with IV Meropenem 2 gm 3 times daily for 6 weeks and IV Gentamycin 80 mg twice daily for 2 weeks followed by IV Ciprofloxacin 200 mg twice daily for 4 weeks. After completion of the antibiotic course, he was taken up for CAG which revealed a huge fusiform aneurysm involving the proximal LAD, stent thrombosis and severe narrowing of origin of first diagonal branch (Figure 1). Subsequently, he underwent coronary artery bypass surgery (CABGS), mitral valve replacement (MVR), explantation of LAD stent and repair of aneurysm. Subsequent recovery was uneventful. After surgery, he was kept on IV antibiotics for additional 1 week.
Coronary Angiogram Showing Aneurysmal Dilatation of Proximal Stented Segment of LAD.
Case: 2
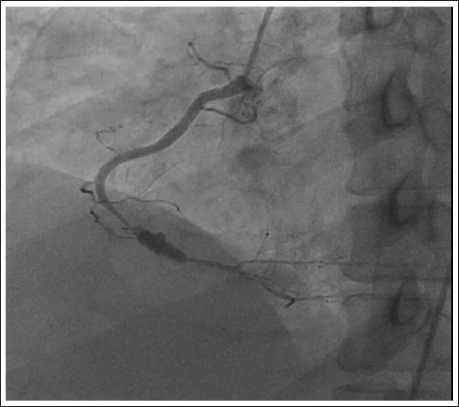
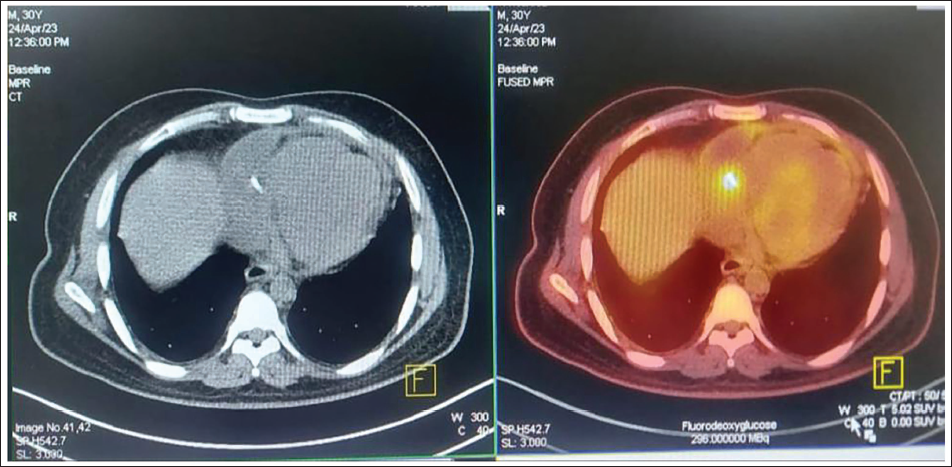
A 30-year-old male with preexisting hypertension (HTN), diabetes mellitus (DM), and pulmonary tuberculosis presented to local hospital with chest pain of 2 hours duration. ECG showed changes of acute ST segment elevation (STE) inferior wall MI. He was thrombolysed with 15 lakh units of Streptokinase. CAG revealed double vessel disease. Subsequently, PTCA and stenting of distal RCA as well as proximal RCA were done. Four days later, patient complained of chest discomfort, fever, and cough which was managed conservatively. As condition worsened he was brought to our hospital where transthoracic echocardiogram (TTE) showed pericardial effusion with impending cardiac tamponade, therefore, emergency pericardiocentesis was done and 200 ml of straw colored pericardial fluid was aspirated and sent for analysis. Pericardial fluid grew P. aeruginosa with multidrug resistance. Suspecting CSI, FDG PET CT was done, which revealed hypermetabolic focus around distal RCA stent, which was suggestive of stent infection (Figure 3). Patient was started on Ceftazidime 2.5 gm and Aztreonam 2 gm. CAG revealed diffuse ISR with aneurysm in distal RCA (Figure 2). Subsequently, he underwent CABGS and stent explantation.
Coronary Angiogram Showing Aneurysm of Distal Stented Segment of RCA.
FDG PET CT Revealing Hypermetabolic Focus Around Distal RCA Stent.
Case: 3
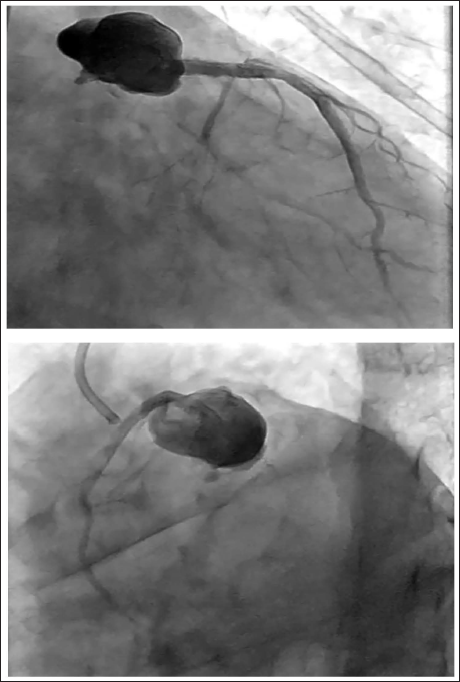
A 54-year-old male with a previous history of HTN and DM presented to local hospital with chest pain. ECG showed acute STE anterior wall MI. He was thrombolysed with tenecteplase. He underwent rescue PTCA and stenting of proximal LAD with 3.5 × 30 mm2 endeavor resolute stent. Two weeks later he developed fever, breathlessness, and reinfarction. CAG showed triple vessel disease (TVD) (70% luminal loss in proximal portion of stent in LAD, 80% stenosis in proximal segment of LCX, 80% stenosis in mid-segment of RCA) and a large aneurysm arising from LAD ostium (Figure 4). CABGS, LAD aneurysm repair and removal of stent was done.
Coronary Angiogram Showing Large Aneurysm Arising from Stented Portion of Proximal LAD.
Case: 4
A 62-year-old diabetic presented with ACS. CAG revealed TVD, ad hoc PTCA, and stenting of RCA was done. Following coronary angioplasty, he had recurrent episodes of fever for 2 months. Urine culture and blood culture grew P. aeruginosa. On further evaluation, 2D echo showed severe AR due to IE of native aortic valve, CAG revealed severe TVD. He was subjected to coronary artery bypass grafting and aortic valve replacement. Postoperative recovery was uneventful.
Case: 5
A 52-year-old female with past h/o HTN, DM, and hypothyroidism presented to a government hospital with acute STE anterior wall MI and was thrombolysed with tenecteplase. CAG revealed 2 vessel disease. PTCA and stenting of LAD was performed. One month later patient presented with fever and reinfarction. Echo showed severe MR. CAG revealed LMCA disease with aneurysm, LAD stent thrombosis, critical lesion in LCX and PDA. Plain old balloon angioplasty (POBA) was done to LAD along with intracoronary Faximab bolus administration. Blood culture was positive for P. aeruginosa. The patient underwent coronary artery bypass grafting and mitral valve repair subsequently.
Discussion
Though complications are inherent to cardiac interventions however, occur infrequently. Some of them are life threatening. Complications of no reflow phenomenon and stent thrombosis occur in around 2% of patients while coronary artery endarteritis and stent infection occurs in less than 0.1% cases. The first case of coronary artery stent infection was described after Palmaz-Schatz stent implantation in a 66 years old women in 1993. 4 Majority of patients present with fever and chest discomfort. High index of suspicion is required when patients present with fever and constitutional symptoms of 4 to 6 weeks duration following PCI. 5 Infection can manifest early or late following stent implantation. On the basis of timing of onset, CSI is classified as early onset if infection occurs within 10 days of coronary intervention, which responds to medical therapy alone, otherwise classified as late onset if infection occurs >10 days after implantation, requiring combined surgical and medical therapy. 6 These empirical definitions were based on the evaluation and treatment of foreign body infections in orthopedic surgery.
Currently, there is no universal standard for diagnosing CSI, Dieter et al proposed a criteria for suspecting CSI. 7 A definitive diagnosis of CSI was made on the basis of postmortem findings or surgical material examination revealing an infected coronary artery stent complex. The presence of 3 or more of the following criteria indicate possible CSI: coronary stent implantation within the past 4 weeks; repeated procedures through same vascular access; complications at the site of puncture; fever above 101.5°F, documented bacteremia, leukocytosis without any obvious focus of infection; ACS; or cardiac imaging supporting persistent inflammation.
Coronary angiography and blood cultures were the most commonly done investigations after suspecting CSI. Multi-detector CT angiography is the current imaging modality of choice for evaluating suspected mycotic aneurysms because it has an added advantage of rapid image acquisition, high spatial resolution, and multi planar capabilities.8, 9 Only limitations are radiation and usage of iodinated contrast media. Other advanced diagnostic imaging modalities are PET CT and cardiac MRI. In our case series, all patients underwent a combination of imaging modalities.
As per the literature, S. aureus was the most common organism isolated followed by P. aeruginosa and Streptococcus organisms. Pseudomonas was the most common organism identified in our case series. CSI cases are commonly associated with infected aneurysms. These infected aneurysms are formed by arterial injury and direct infection. 13
Potential complications of mycotic coronary artery aneurysms include rupture of aneurysm, cardiac tamponade, fistula formation, myocardial ischemia or infarction secondary to septic embolization and sudden cardiac death.10, 11, 12 Mortality rate associated with mycotic coronary aneurysm is 43% to 53%.14, 15, 16 When CSI is suspected, prompt and appropriate antibiotic therapy active against Pseudomonas and Staphylococcus should be initiated until causative agent is identified. Mycotic aneurysm excision along with stent explantation is highly recommended.17, 18 The mechanism of stent induced aneurysm has been classified by Aoki et al 19 into 3 types.
Conclusion
CSI is a rare complication of PCI. Increasing number of coronary interventions including complex procedures may increase the incidence of stent infections further. Suspicion, early diagnosis, and prompt treatment may reduce the morbidity and mortality. Adequate measures aimed at prevention are pivotal. One common feature in our series is all our patients had interventions following acute STEMI and 4 out of 5 patients’ infection were with P. aeruginosa. All our patients underwent surgery and recovered completely.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval has been obtained from Institute Ethics committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained for publication of clinical data as well as images for academic purpose only from the family.
