Abstract
WPW syndrome with rheumatic mitral stenosis is a rare and deadly combination. We report a patient with the above combination, presenting with drug-refractory antidromic tachycardia due to left atrial flutter and marked dyspnea. She underwent successful radiofrequency ablation of left-sided accessory pathway, left atrial flutter, and percutaneous mitral commissurotomy in a single sitting.
Introduction
The association between Wolff-Parkinson-White (WPW) syndrome and rheumatic mitral stenosis is rarely reported.1, 2 The combination has profound hemodynamic significance, and treatment is challenging.
We report a patient with rheumatic mitral stenosis and WPW syndrome with refractory antidromic tachycardia due to left atrial flutter (AFL). The patient was treated successfully in a single sitting with a combined percutaneous mitral commissurotomy (PTMC) and radiofrequency ablation (RFA) of the accessory pathway and AFL. To our knowledge, this is one of the very few reported cases where successful PTMC and RFA of the accessory pathway, along with AFL, were performed in a single sitting.
Case Report
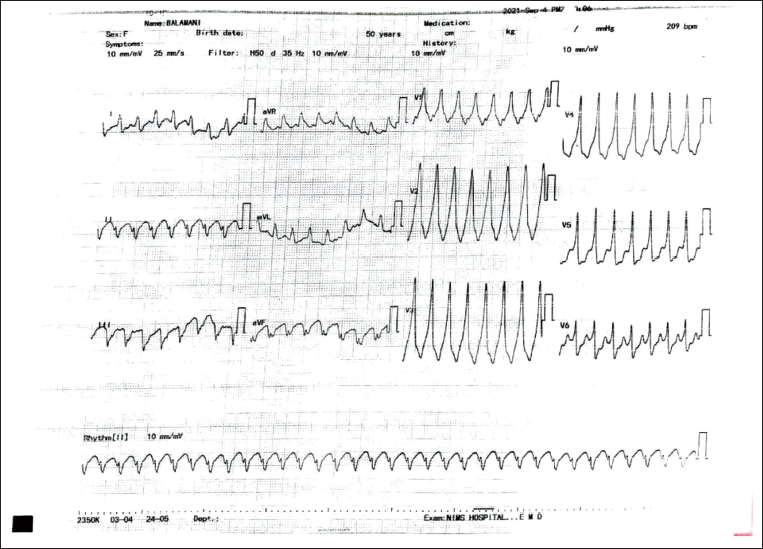
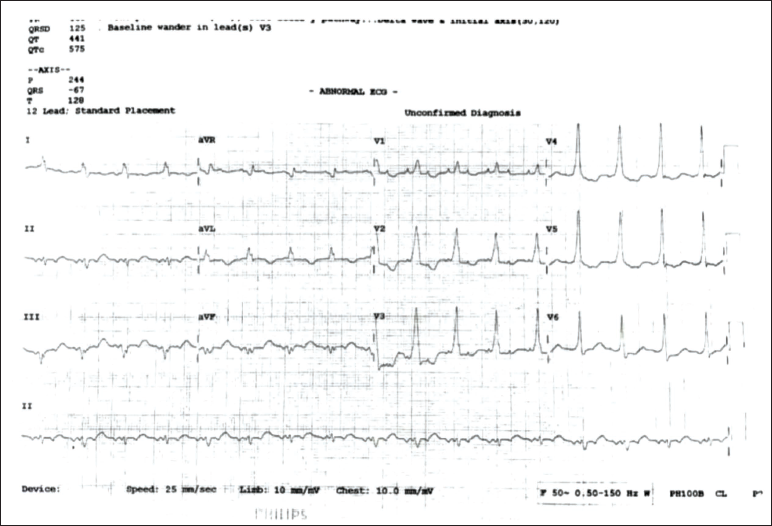
A 56-year-old female patient was referred to our tertiary care center with refractory palpitations and breathlessness lasting three months. At presentation, the patient was breathless with marked tachycardia. Her blood pressure was 90/60 mmHg. The ECG showed wide QRS tachycardia with a rate of 270 beats per minute (Figure 1), with the possibility of VT/SVT with aberrancy. The patient was given inj. amiodarone for medical cardioversion. During sinus rhythm, the ECG showed a short PR interval with a +ve delta wave in I, AVL, and V1-V6 suggestive of the left lateral accessory pathway (Figure 2). Clinical examination revealed loud S1 and P2 with a long mid-diastolic murmur at the apex. A 2D echocardiogram showed severe mitral stenosis (MVA: 0.9 cm2) with mild mitral regurgitation (MR).
ECG Showing Wide QRS Tachycardia with a Rate of 270 Beats/Minute.
ECG in Sinus Rhythm Showing Left Lateral Accessory Pathway.
Initially, we tried to control the heart rate with rate control medications (IV esmolol, IV metoprolol, and IV diltiazem) and also tried to medically cardiovert with amiodarone to stabilize the patient.
But in spite of so many attempts of drug therapy, her heart rate was continuously on the higher side, and she continued to have refractory symptoms of palpitations and breathlessness.
After informed consent, the patient was scheduled for PTMC and RFA of the accessory pathway in a single sitting. The mitral valve was semi-pliable with a Wilkins Score of 9/16 and mitral regurgitation of 4.0 cm2. Trans-esophageal echocardiography did not show any clot in the left atrium (LA) or in its appendage. As the patient was an elderly female, we did a coronary angiogram to rule out the presence of coronary artery disease. Her coronary angiogram was normal.
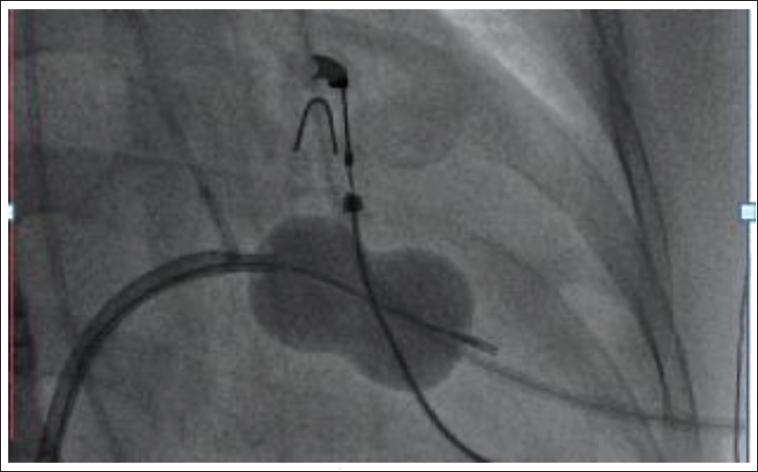
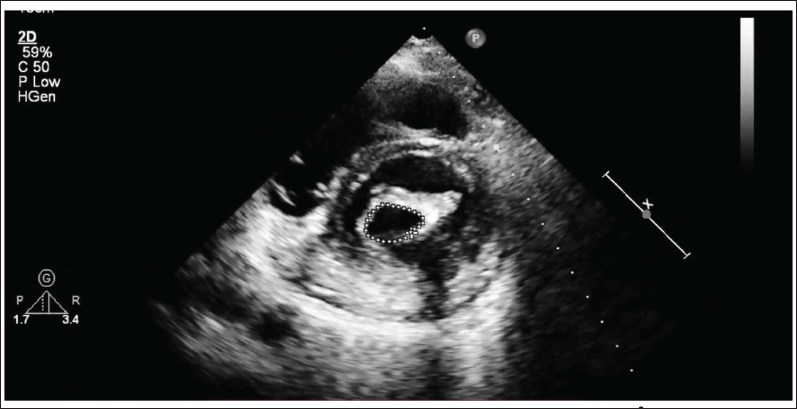
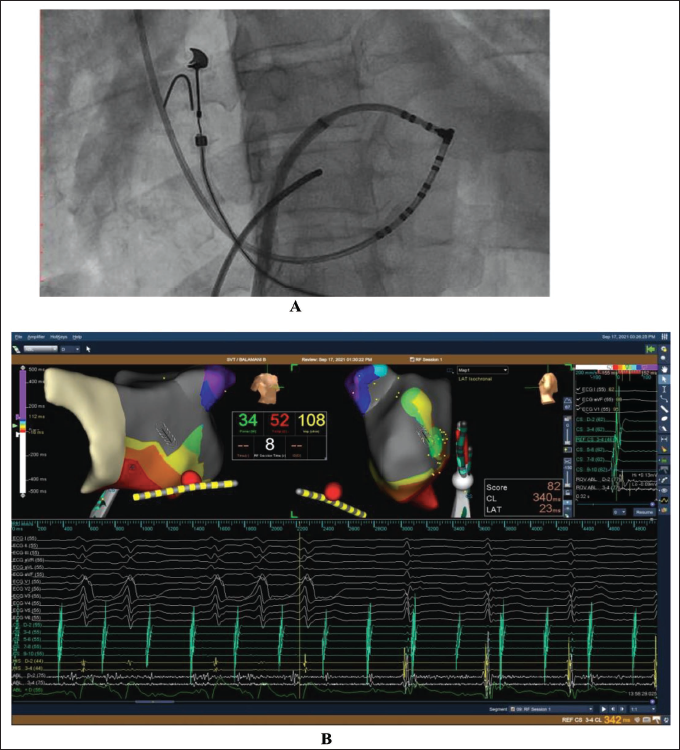
We did PTMC first through right femoral vein access. The atrial septum was punctured under fluoroscopy guidance with a 21-G Brockenbrough needle and dilated with a 14-F dilator, and a 26-mm Inoue balloon was passed across the mitral valve, and graded inflations were done (Figure 3). The mean LA pressure decreased from 28 to 8 mmHg. Mitral valve area by 2D echo planimetry was 1.9 cm2 (Figure 4) with no increase in MR.
Fluoroscopy Image in RAO Showing Inflated Inoue Balloon Across Mitral Valve.
2D Echo in Parasternal Short Axis Showing MVA of 1.9 cm2 by Planimetry.
Challenges while doing PTMC were a relatively high Wilkins score, a semi-pliable valve, mild to moderate MR, and high heart rate because of antidromic tachycardia causing instability of the septal puncture needle. However, we achieved a good result without an increase in MR severity.
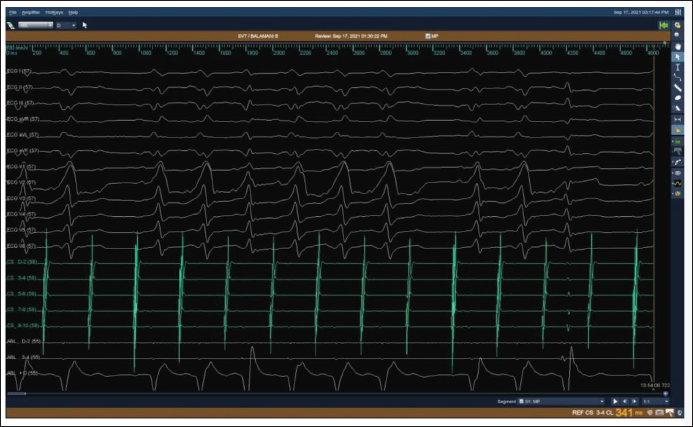
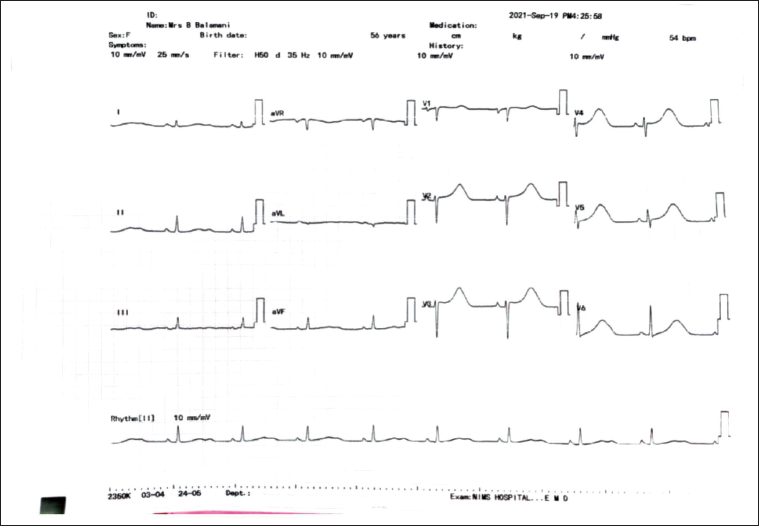
The patient was taken up for an EP study by the same team. The patient was in incessant wide QRS tachycardia when taken up for an EP study. The intracardiac electrogram activation pattern of tachycardia suggested antidromic conduction through the left lateral accessory pathway (CS distal showing a shorter AV interval than proximal) (Figure 5). The mitral annulus was mapped through the trans-septal route with a 7F-D curve Biosense Webster non-irrigation ablation catheter. At 1 ‘O’ clock position in LAO view (Figure 6A), a fused AV signal was obtained. Ablation was successful, and the preexcitation disappeared within three seconds of starting RFA (Figure 6B). However, the patient continued to be in left atrial flutter (Figure 7A). 3D mapping of left atrial flutter (Figure 7B) done with Ensite velocity (St Jude Medical Inc.) and ablation lines drawn along the LA roof terminated the AFL. The patient was in sinus rhythm (Figure 8) with no evidence of preexcitation even on the adenosine challenge, and no tachycardia was inducible in spite of isoprenaline provocation. The patient remained in sinus rhythm with no pre-excitation and mild MS at six months’ follow-up.
Intra-cardiac Electrograms Showing Short AV Interval in Distal CS During Antidromic Tachycardia.
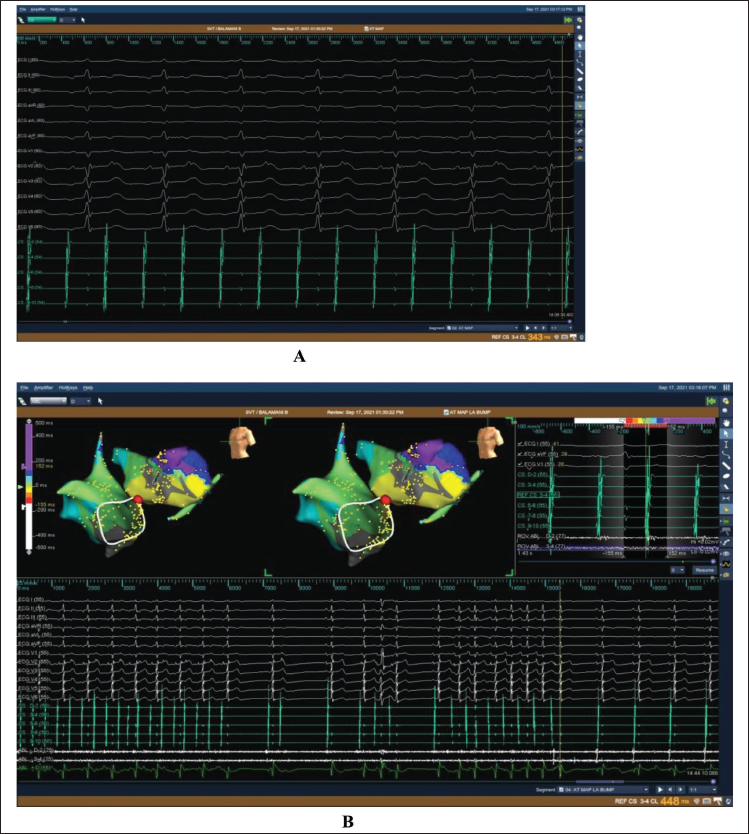
(A) Fluoroscopic Image in LAO Showing Ablation Catheter Position at Successful Ablation Site. (B) Intracardiac Electrograms Showing Successful Ablation of Left Lateral Accessory Pathway.
(A) Intracardiac Electrograms Showing Left Atrial Flutter. (B) 3D Mapping of Left Atrial Flutter and Successful Ablation.
ECG Showing Sinus Rhythm with No Pre-excitation or Atrial Flutter.
Discussion
The combination of WPW syndrome and rheumatic mitral stenosis is rarely reported in the literature. The main concern with this association is marked tachycardia due to antidromic conduction of atrial arrhythmias with increased LA pressures, resulting in pulmonary congestion and low cardiac output. This life-threatening pre-excited atrial arrhythmia could be refractory to medical therapy, as noted in our patient.
The prevalence of RHD is high in South Asia and Africa. 3 The most common manifestation of RHD is MS. The risk of having atrial fibrillation (AF) is very high, nearly up to 45% in patients with MS. 4 Patients with WPW syndrome, too, have an unexplained higher incidence of AF, about 10%-38%. 5 In WPW patients with AF, the ventricular rate may be very high, as the rate-limiting effect of the AV node is bypassed, which can result in ventricular fibrillation and sudden cardiac death. Our patient also had a ventricular rate of 250-270 beats/minute during AFL with antidromic conduction to the left ventricle.
Management depends on the patient’s clinical status and the decisions of the multidisciplinary team. In most reported cases, simultaneous RFA and mitral valve replacement were done, 6 while in some cases, RFA was performed before valve replacement. 7 Alkindi et al reported simultaneous performance of PTMC and RFA of the right posterior accessory pathway by two different teams in a patient with asymptomatic WPW and MS. 8
In contrast, our patient was very symptomatic with drug-refractory pre-excited tachycardia due to AFL and the left lateral accessory pathway, in whom successful PTMC and RFA were done by a single team in one sitting.
Alper et al performed RFA during pre-excited atrial fibrillation in a patient with right mid-septal pathway with mitral stenosis. 1 In our patient, we also ablated the left lateral accessory pathway during pre-excited AFL. Further, we mapped the left atrial AFL with 3D mapping and successfully ablated it, restoring total sinus rhythm in our patient.
Conclusion
WPW syndrome and rheumatic MS with drug-refractory pre-excited tachycardia are rare and life-threatening entities. PTMC and RFA are successful treatment options in selected cases. 3D mapping and successful RFA of the atrial arrhythmias, restoring sinus rhythm, may further enhance the treatment outcome in this rare combination, as noted in our patient.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This is a case report and the patient was not subjected to any new or experimental interventional procedures, so ethical committee approval is not necessary.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Patient gave consent to publish this case.
