Abstract
Objective:
Patients with atrial fibrillation (AF) are at increased risk of thromboembolic events originating mainly from left atrial appendage thrombus (LAAT). Patients with atrial flutter (AFL) are treated with anticoagulation based on the same criteria as patients with AF. However, whether patients with AFL have similar thromboembolic risk as AF is unclear. In the current study we aimed to estimate the prevalence of LAAT in patients with AFL undergoing trans-esophageal echocardiography (TEE).
Methods/results:
We included 438 patients (404 with AF and 34 with AFL) scheduled for TEE to rule out LAAT before cardioversion (patients who reported no or inadequate anticoagulation before cardioversion). Demographic and echocardiographic data were compared between patients with and without LAAT. Despite a similar CHA2DS2-VASC score (3.8 ± 1.3 vs 3.4 ± 1.5 in the AF and AFL groups, respectively, P = .09), LAAT was documented in 12 (2.8%) in the AF group and in no patient in the AFL group (P < .0001).
Conclusion:
Based on our results and previous studies, it seems reasonable to re-evaluate the need for oral anticoagulation in specific populations with AFL such as those with solitary AFL (without a history of AF episodes) undergoing successful ablation and in those with low CHA2DS2-VASC score.
Introduction
Atrial fibrillation (AF) is the most common persistent arrhythmia in the elderly, with increasing incidence overtime. 1 This global pandemic is associated with several complications such as thromboembolic events and heart failure.1,2 The main goal in managing patients with AF is to prevent thromboembolic complications, particularly cerebrovascular and cardiovascular events. Most embolic events originate from the formation of left atrial appendage thrombus (LAAT) due to blood stasis in the non-functioning atria. 3 The indication for oral anticoagulation in patients with AF is based on the thromboembolic risk estimated by the CHA2DS2-VASC score. Compared to AF, atrial flutter (AFL) is considered a more organized arrhythmia that is usually located in the right atrium and involves the cavo-tricuspid isthmus. 4 Although the anticoagulation strategy in patients with AFL is similar to AF, the embolic risk may be different since previous studies that have focused on the incidence of LAAT in patients with AF compared to AFL by transesophageal echocardiography (TEE) showed a wide range of results.5-10 These ambiguous results may be explained by variations in the study design and inclusion criteria (before cardioversion, before ablation, or as a screening test), different anticoagulation status of the patients, and unspecified diagnosis (fibrillation-flutter diagnosis). Although it is well established to prescribe oral anticoagulants for both AF and AFL depending on the CHA2DS2-VASC score, the difference in the nature of the arrhythmia may provide insights for difference in the thromboembolic risk. In the current study we aimed to investigate the incidence of LAAT between two well defined groups of patients (AF vs AFL) who did not receive oral anticoagulation.
Methods
Study population
Between January 2018 and September 2021, 506 consecutive patients had a documented TEE study in the Cardiology Department at Galilee Medical Center. The TEE was performed to rule out LAAT in patients with AF or AFL without adequate anticoagulation in whom the arrhythmia was documented for more than 48 hours or of unknown duration and were scheduled for elective cardioversion. Patients were defined as having AFL only if they have solitary flutter without concomitant report of previous AF episodes.
Inclusion criteria
We included patients with clear definition of the arrhythmia (solitary AF or solitary AFL) with complete TEE study in whom the indication is well defined and all the echocardiographic parameters are available.
Exclusion criteria
Patients who had TEE as part of endocarditis workup (to exclude endocardial vegetations) or cerebrovascular accident (searching for embolic origin without the documentation of arrhythmia) were excluded. A total of 438 patients were finally identified and included in the analysis.
Transesophageal echocardiography
All the echocardiographic studies were performed by a cardiologist (echocardiography specialist) using Philips Epiq-7 machine, and EPIQ X8-2t transducer (Phillips, Adnover, MA, USA). During the study, patients were mildly sedated using low doses of midazolam and propofol. The presence or absence of LAAT was confirmed with direct visualization of the left atrial appendage (LAA) using color doppler and left atrial velocity. The standard views were the short axis aortic valve view (30°-60°) and the 2-chamber view (80°-100°) when the probe is in the mid-esophageal position. In equivocal cases, multiple views and 3D echocardiography were also used. Detection of sludge or spontaneous echo contrast were reported but not considered as equivalent to thrombus for this purpose. Left atrial diameter (LAD) and left atrial area (LAA) were also measured. All patients with clear LAA without evidence of thrombus underwent synchronized electrical cardioversion.
Data extraction
We retrospectively collected demographic and echocardiographic data for all patients as shown in Table 1. Echocardiographic parameters included left ventricular ejection function (LVEF), spontaneous echo contrast (SEC), atrial sludge, and LAAT. CHA2DS2-VASC score was calculated for each patient in which congestive heart failure, hypertension, age between 65 and 74, diabetes, female gender, and vascular disease were given one point whereas age above 75 and previous systemic embolism (including CVA or TIA) were given 2 points.
Baseline characteristics of the patients.

Abbreviations: AF, atrial fibrillation; AFL, atrial flutter; CKD, chronic kidney disease; CVA, cerebrovascular accident; LVEF, left ventricular ejection fraction.
Follow-up
Patients with documented LAAT were followed for events including hospitalizations cerebrovascular events, transient ischemic attack, or other systemic events. In patients with documented embolic events, adherence to oral anticoagulation was assessed based on computerized files.
Statistical methods
Categorical variables are presented as percentages, while continuous variables by mean with standard deviation (SD). We used Fisher’s exact test and Chi square test to compare categorical variables between the 2 groups. Independent sample t-test and Mann-Whitney tests were used for continuous variables. All tests were conducted at a two-sided overall 5% significance level (α = .05). Statistical analysis was performed using IBM SPSS statistics, version 27. The study was approved by the local ethical committee of Galilee Medical Center.
Results
A total of 438 patients (34 with AFL and 404 with AF) were included in the final analysis. LAAT was documented in 12 (3%) of the AF group and in none (0%) of the AFL group (P < .0001). The flowchart of the study design is shown in Figure 1.
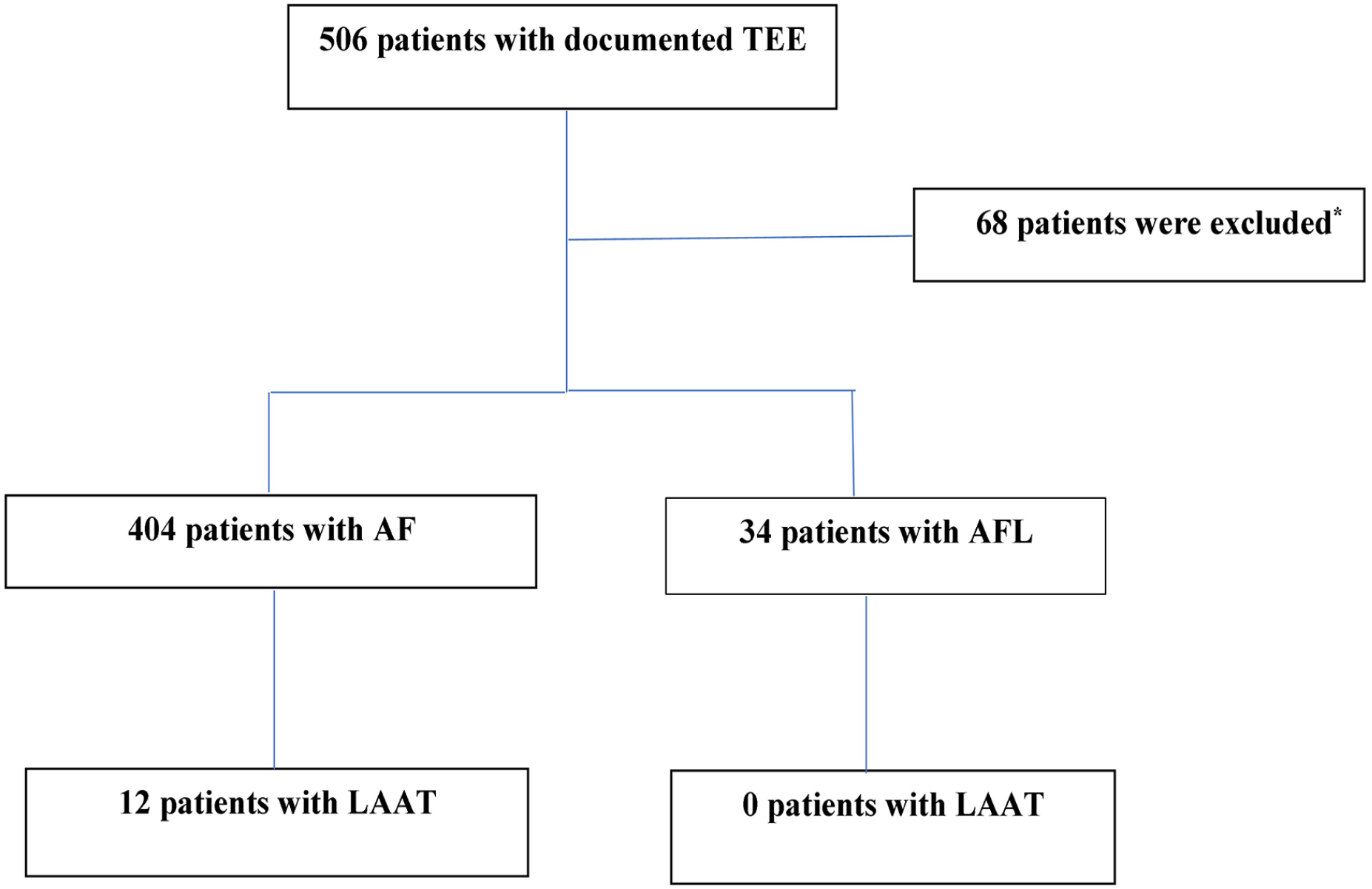
Flowchart of the study.
The demographic and clinical characteristics of the patients are given in Table 1.
As may be predicted, patients with LAAT had larger LAD (42.1 ± 3.0 mm vs 34.2 ± 2.1 mm, P < .0001) and LAA (20.4 ± 2.1 cm2 vs 18.5 ± 1.8 cm2, P < .0001) than patients without LAAT. Although the CHA2DS2-VASC score was not significantly different between the groups (3.8 ± 1.3 vs 3.4 ± 1.5, in the AF and AFL groups respectively, P = .09), LAAT was observed only in the AFL group. Demonstrative images of LAAT during TEE are given in Figure 2 (standard TEE) and Figure 3 (3D-TEE).

LAAT as demonstrated in patients with atrial fibrillation during TEE.
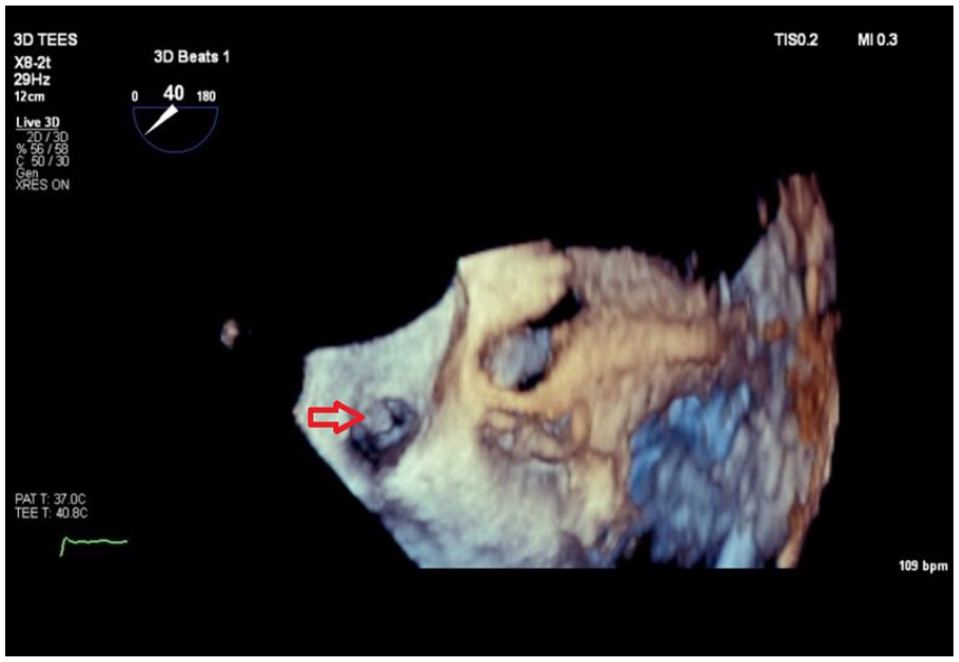
LAAT as demonstrated during 3D TEE.
The baseline characteristics and echocardiographic data of the patients in the AF group with and without LAAT are given in Table 2.
Baseline characteristics and echocardiographic parameters of patients in the AF group with versus without LAAT.

Abbreviations: CKD, chronic kidney disease; CVA, cerebrovascular accident; IHD, ischemic heart disease; LAA, left atrial area; LAD, left atrial diameter; LVEF, left ventricular ejection fraction; SEC; spontaneous echo contrast.
Follow-up
After a median follow up of 3 (IQR, 0.5-4) years, 2 patients (16.7%) in the AF group with documented LAAT were hospitalized for embolic events (1 for TIA and 1 for retinal artery occlusion) despite adequate oral anticoagulation, whereas no patients in the AFL group had embolic events during the follow-up period.
Discussion
In the current study, we assessed the prevalence of LAAT in patients with AF and AFL during TEE. To make a clear distinction, we included only patients with diagnosis of solitary AF or AFL and without adequate anticoagulation therapy who were scheduled for TEE before electrical cardioversion. Among 34 patients with AFL, no one had LAAT detected during TEE. As may expected, patients with LAAT in the AF group had larger LAD and LAA along with more evidence of SEC in the left atrium. The difference in electromechanical mechanism between AFL and AF may partly explain the difference in embolic events between the 2 conditions. The origin of AF is usually related to the pulmonary veins and the left atrium with chaotic non-functioning atrial activity whereas the more organized AFL circuit is usually located in the right atrium and typically related to the cavo-tricuspid isthmus. Overall, studies showed that the risk of ischemic stroke in AFL is lower than AF, particularly when including solitary AFL compared to AFL with coexistent AF. 11 Nevertheless, studies aimed to evaluate the incidence of LAAT in patients with AFL showed conflicting results. In one study comparing the risk of stroke in 59 patients with AFL with matched cohort of AF, showed higher risk in the AFL group. 12 However, in this study, 56% of the patients in the AFL group developed subsequent AF during the study period and some of them were treated with aspirin or warfarin. In another retrospective study of 191 unselected patients referred to AF treatment (cardioversion or ablation), 4 of them had acute embolism following ablation or cardioversion and 9 patients had thromboembolic events during the follow-up period. 13 These differences emphasize the difficulties in interpretation of the data focusing on the risk of thromboembolic risk in AFL compared to AF. The heterogeneity in the reported data may result from different study designs and population, mixed anticoagulation status (anticoagulated and non-anticoagulated patients), and vague primary diagnosis (so called fibrillation-flutter arrhythmia). Moreover, many of previous studies included patients treated with aspirin or warfarin rather than new oral anticoagulant drugs. The difference in embolic risk between AFL and AF may be explained by the different mechanisms of arrhythmia. The left atrial appendage has a mechanical function by completing the reservoir role of the atria in diastole. Due to its blind end shape and its reduced blood flow velocity, the majority of thrombi identified in AF originate inside the LAA. 14 Schmidt et al studied the thrombogenity in AFL and found that among 139 with AFL, 15 had thrombogenic milieu (defined as the induction of SEC and reduced mechanical appendage function). All the patients with such thrombogenic milieu had a previous history of AF and only 2 had LAAT. 6 SEC is seen during TEE as “smoke” and it is related to conditions with low flow in the atrium such as significant mitral stenosis and AF, is often accompanied by decreased atrial blood flow velocity, and is associated with increased risk of thromboembolic events. The standard method for SEC quantification was proposed by Fatkin et al 15 and based on visual grading (grades 0-4+) in which 0 denotes absence of echogenicity and 4+ an intense echo density and very slow swirling patterns in the LAA with similar density in the appendage. Previous studies have shown a correlation between the presence of SEC with high blood viscosity parameters such as high hematocrit and fibrinogen levels.16-18 In the absence of adequate anticoagulation, SEC can progress to sludge and thrombus and finally may lead to systemic embolism.19,20 In the study by Grimm et al, 21 patients with AFL had higher LAA flow velocity and less pronounced post cardioversion- atrial dysfunction compared to patients with AF. We did not have the data regarding atrial velocity in our study population, but the finding of larger atrial size and higher proportion of patients with SEC in patients with AF may be translated to lower atrial velocity in this group. Our study included patients with AFL with no documented history of AF episodes and without effective anticoagulation and subsequently reflect the natural history of this arrhythmia. Currently, oral anticoagulation is still recommended in AFL since these patients often have risk factors for thromboembolic events due to co-morbidities and concomitant episodes of AF. However, based on our studies and other similar studies, it is reasonable to re-evaluate the need for oral anticoagulation in some patients such as those undergoing successful AFL ablation without documented previous episodes of AF or in patients with solitary AFL with high bleeding risk and low CHA2DS2-VASC score.
Limitations
Our study has the following limitations: first, although our patients with AFL had no documented previous episodes of AF, it is difficult to differentiate them from those with combined arrhythmia since patients may have alternating asymptomatic episodes of AF and AFL. Based on our results and previous studies, the prevalence of atrial thrombi in patients with AFL seems to be less common than in AF. Yet, in older patients with comorbidities and long episodes of flutter, the risk may be similar. Therefore, despite these results, the approach to anticoagulation should be performed case by case.
Future perspective
Despite the small size of the current study and its retrospective design, the consistency of the results with previous studies raise the possibility of managing patients with AFL in a different approach. In patients with high bleeding risk and low CHA2DS2-VASC score,1,2 anticoagulation may not be necessary.
Conclusions
When compared to AF, the prevalence of LAAT is lower in patients with AFL. The risk of LAAT in patients with AFL and therefore the recommendation for anticoagulation depends on other comorbidities and the coexistence of AF episodes. In specific populations such as patients with high bleeding risk and low CHA2DS2-VASC score, anticoagulation strategy may be more lenient than in patients with AF. Larger prospective studies are warranted for this purpose.
Footnotes
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
