Abstract
Objectives
Heart failure with reduced ejection fraction (HFrEF) is an advancing illness that can be managed with various drugs. In this study, we present data on the tolerance of perindopril in patients with HFrEF who were switched from angiotensin receptor neprilysin inhibitor (ARNI) to perindopril because of issues with the former’s tolerability or increased medication costs.
Methods
A total of 125 patients with HFrEF were switched to perindopril in this retrospective study carried out at the heart failure clinic. Of those, 109 patients had data accessible for a 6-month follow-up. The data of the patients who switched from ARNI to perindopril between June and December 2023 were collected. Data such as the New York Heart Association (NYHA) class, N-terminal pro hormone brain natriuretic peptide (NT-proBNP), and left ventricular ejection fraction (LVEF) were compared at baseline and 6-month follow- up with those collected after the switchover.
Results
During the transition, the average age was 58 ± 13 years (mean ± standard deviation), the disease duration was 2.6 ± 1.4 years, and 29.6% of the patients were female. The average period of follow-up following the transition was 5.7 ± 1.5 months. The 6-minute walk distance (6MWD) reduced from 344 ± 106 to 319 ± 85 m (P = .056), but the NT-pro BNP levels increased from 478 ± 170 to 490 ± 137 pg/mL (P = .56) after the transfer. All patients continued to maintain their NYHA functional class after the transition, with no significant difference seen (P = .37). There was a little improvement in LVEF from 40% ± 4% to 41.5% ± 5% (P < .001) during the follow-up after the switchover. Following the transition, no changes were observed in vital indicators, serum electrolytes, and creatinine levels. All patients successfully tolerated the transition to perindopril.
Conclusions
The transfer from ARNI to perindopril was both well tolerated and safe. The patients’ functional condition remained unchanged, as did their NT-pro BNP levels. A modest improvement in LVEF was noted. In India, switching from ARNI to perindopril is less expensive.
Keywords
Introduction
Heart failure with reduced ejection fraction (HFrEF) is a chronic progressive disorder with high morbidity and mortality rates. 1 The global prevalence of heart failure (HF) is 26 million. 2 Approximately 8 to 10 million people being affected by HF in India, and the numbers are projected to rise extensively by 2030.3, 4 Angiotensin-converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), ß-blockers (BBs), and mineralocorticoid receptor antagonists (MRAs) form the cornerstone of the HF treatment protocol. However, newer drugs are being continuously added, thereby expanding the list of effective treatments. 5
In 2016, the European Society of Cardiology and the American College of Cardiology (ACC)/American Heart Association (AHA) endorsed sacubitril/valsartan as the first agent from the novel class of medications known as angiotensin receptor neprilysin inhibitors (ARNI) for HFrEF.6, 7 It is authorized as a replacement for ACEI in ambulatory care patients who continue to experience symptoms while receiving the best possible treatment, in order to further decrease the likelihood of hospitalization or death. ARNI is suggested because of the positive outcomes observed in the
Currently, the significant hindrance to the broad use of sacubitril/valsartan in India is mostly attributed to its exorbitant price, despite its established advantages. ARNI failed to meet the cost-effectiveness criteria for preventing CV mortality and HF rehospitalization, as demonstrated in an Indian cost needed to treat and number needed to treat analysis. 3 But, currently, owing to cost considerations, many patients after initial treatment with ARNI got eventually switched back to either ACEIs or ARBs in clinical practice. Reported evidence favors the use of ACEI in the setting of HFrEF. ACEI, as a group, do not appear to have a class effect and, therefore, display differences in their clinical outcomes. Perindopril has a more specific dual-action and tissue specificity. Because of limited experience with the ARNI in ambulatory clinical practices, we conducted a retrospective evaluation of the results of transitioning from ARNI to perindopril on an outpatient basis in India.
Methods
Study Design and Patient Population
This was a retrospective observational study conducted from January to June 2023 at the Department of Cardiology, Yashoda Hospitals, Somajiguda, Hyderabad, India. The key inclusion criteria were patients >18 years old with HFrEF (New York Heart Association [NYHA] Classes I-IV and left ventricular ejection fraction [LVEF] < 40%) and with a stable sinus rhythm, who had been unable to tolerate or discontinued ARNI. Patients receiving ARBs, other ACEI, or BBs; patients with congenital heart disease, unstable angina, recent acute myocardial infarction, primary hepatic failure, or active cancer; or patients with stenotic valvular heart disease were excluded. Patients took their last dose of ARNI in the evening, and the following morning, they took their first dose of perindopril within 12 to 24 hours of the last ARNI dose as per the clinician’s advice. Following the recommendations, the ARNI was administered to all patients at maximum tolerated doses. The perindopril doses of 2, 4, and 8 mg were gradually escalated every 2 to 4 weeks until reaching the highest dose that could be tolerated by the patient. This retrospective study was approved by the institutional review board.
Data Collection
Baseline demographics and clinical data were obtained from the clinic visit immediately before the switch. Baseline conventional treatment and any modification during follow-up visits were recorded. After basic examination, perindopril dosage was prescribed at the investigator’s discretion. The collected data included a 6-minute walk distance (6MWD), NYHA functional class, brain natriuretic peptide (BNP), and renal function tests. The LVEF was obtained from an echocardiogram performed using modified Simpson’s biplane method before the switch (at baseline) and at the follow-up visit. As the right ventricular systolic pressure and right atrial pressure values are given as a range, the highest values are presented for each time point. Post-switch data of patients were obtained at a 6-months follow-up visit after switching to perindopril.
Study Outcomes
Study outcomes included changes in vital parameters, laboratory values, functional status, exercise tolerance, and LVEF.
Statistical Analysis
With the assumptions, power 80%, α 0.05, mean difference of 2.4 in LVEF, and variance 72.25 (SD = 8.5), the sample size would be 99. Considering a 10% loss to follow-up, the sample size was determined to be 109.
The statistical analysis was conducted utilizing the Windows SPSS program. The data were reported as absolute values accompanied by percentages for nominal data and as averages with standard deviation for continuous data. For the comparison of changes within the study group during subsequent visits, the Wilcoxon test for paired samples was performed. A P value < .05 was considered statistically significant.
Results
Of a total of 125 patients who had been switched from ARNI to perindopril, data of 109 patients were included in the final analysis (Figure 1). The mean age of patients was 58 ± 20 years, and the majority were males (66.7%). ARNI was used in patients in NYHA Classes I (62%) and II (38%). The mean LVEF was 38% ± 5%, and the mean BMI was 32 ± 12.4 kg/m2 (Table 1).
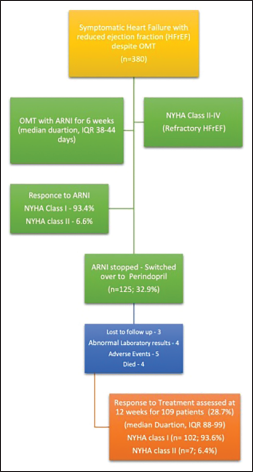
ARNI: angiotensin receptor neprilysin blocker; OMT: other medical therapy; NYHA: New York Heart Association; HFrEF: heart failure with reduced ejection fraction; IQR: inter-quartile range.
Demographics and Clinical Features of Patients Prescribed ARNI
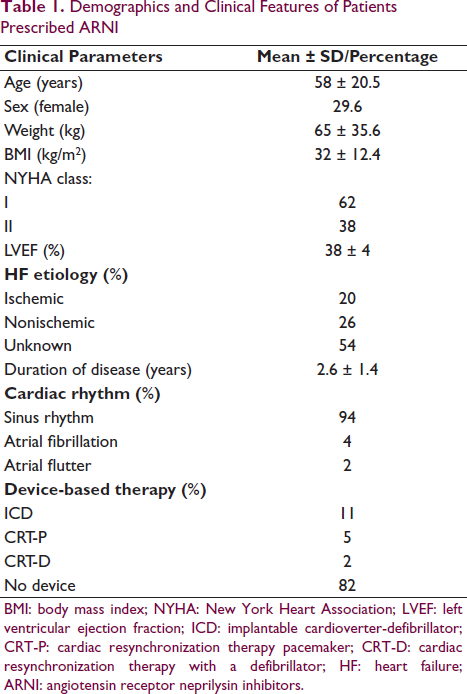
BMI: body mass index; NYHA: New York Heart Association; LVEF: left ventricular ejection fraction; ICD: implantable cardioverter-defibrillator; CRT-P: cardiac resynchronization therapy pacemaker; CRT-D: cardiac resynchronization therapy with a defibrillator; HF: heart failure; ARNI: angiotensin receptor neprilysin inhibitors.
Among 380 symptomatic patients with HFrEF who received ARNI before switching to perindopril, 32.9% (n = 125) discontinued ARNI within the first 3 months of follow-ups because of its high cost. In the follow-up period, all patients with HFrEF continued perindopril. Detailed data on pharmacological treatment at the time of switch is outlined in Table 2. The majority of patients were receiving guideline-directed medical therapy: diuretics (100%), BBs (96%), and MRA (86%).
Baseline Treatment Using ACEI/ARBs and Other Concomitant Pharmacological Treatment Before Initiating ARNI
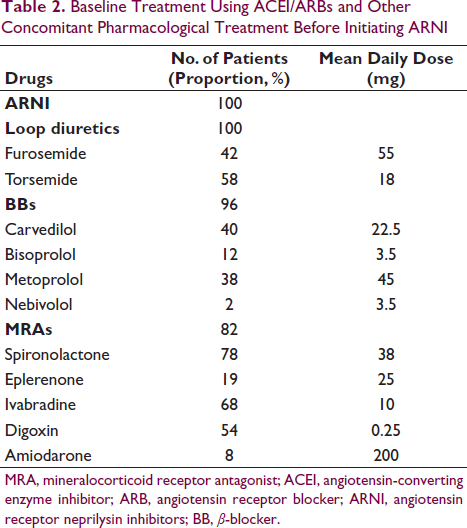
MRA, mineralocorticoid receptor antagonist; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor neprilysin inhibitors; BB, β-blocker.
The mean follow-up duration post-switch was 5.7 ± 1.5 months. All patients were alive at the time of follow-up. Mean systolic blood pressure was 134 ± 23 mmHg, whereas the mean diastolic blood pressure was 84 ± 5 mm Hg, and the pulse rate was 76 ± 28.7 per min at baseline. Vital parameters did not change and were maintained post-switch to perindopril (Table 3). BNP levels increased post-switch (478 ± 170 pg/mL vs. 490 ± 137 pg/mL, P = .93).
Vital Signs, Lab Results, Functional State, and Ejection Fraction at Baseline and at the 6-Month Period After Beginning Perindopril Medication
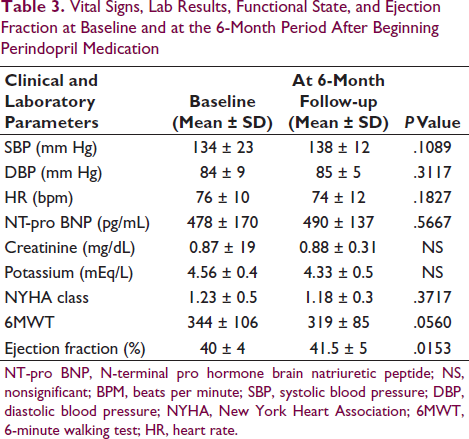
NT-pro BNP, N-terminal pro hormone brain natriuretic peptide; NS, nonsignificant; BPM, beats per minute; SBP, systolic blood pressure; DBP, diastolic blood pressure; NYHA, New York Heart Association; 6MWT, 6-minute walking test; HR, heart rate.
Both serum creatinine and potassium levels remained unchanged during pre- and post-switch. All patients who were switched to perindopril did not exhibit deteriorating symptoms at their outpatient follow-up. Pre- and post-switch 6MWDs were 344 ± 106 m and 319 ± 85 m, respectively (P = .056). Both pre- and post-switch data showed no significant change in HF symptoms in the NYHA functional class after the switch (P = .371). A marginally significant increase in ejection fraction (40% ± 4% to 41.5% ± 5%; P < 0.001) was noted at post-switch follow-up. Overall, all patients reported considerable improvement in their well-being and increased physical activity. Only 4 patients (3.2%) developed worsening HF requiring hospitalization, but they settled with increases in the doses of the guideline-directed medical therapy for the HF. Apart from a cough in 2.4% of patients, no serious adverse effects were reported; nevertheless, the cough disappeared in most cases as the drug continued, and only 0.21% of patients needed to be switched to ARBs.
Discussion
In this retrospective study, switching over to perindopril from ARNI in patients with HFrEF was found safe and effective with no change in any vital and laboratory parameters. Functional status was maintained as seen on the 6-minute walking test, and a marginal but significant increase in LVEF was observed.
In this study, patients who could not tolerate or afford ARNI because of its higher cost were included. In a post-hoc review of the PARADIGM-HF trial, dose reduction below target doses for ARNI was reported in 42% of patients that might be because of tolerability issues, which was more frequent with advanced age and in patients with lower systolic blood pressure, higher renal impairment, and severe symptomatic HF. 10 In our study, 67.1% of patients were switched to perindopril because of the tolerability issues associated with ARNIs and in 32.9% of patients because of the higher cost of ARNIs. Cost, reportedly, was one of the reasons for ARNI discontinuation for CV ailment11, 12 as treatment cost for most patients in India is not usually covered by insurance and is an out-of-pocket expense. 13 Our study showed that switching over to perindopril was safe and effective in patients who could not tolerate/afford ARNI.
It is worth mentioning that the PARADIGM-HF trial was the first study concerning this indication in which the new molecule was compared with basic ACEI- enalapril.8, 9 ACEI do not have a class effect and thus have a difference in their clinical outcomes. Perindopril has a more specific dual-action and higher tissue specificity (bradykinin/angiotensin I selectivity ratio) for perindopril, leading to a lower incidence of cough than other ACEIs.14, 15 In a meta-analysis of randomized trials, perindopril was shown to drive the magnitude of ACEI benefits in terms of reduction in strokes. 16 In agreement with this, treatment with perindopril was effective in maintaining the functional status, and it was able to improve LVEF marginally in patients with HFrEF in this study.
For switching ARNI, we preferred ACEI over ARBs for several reasons. Although they did it in different ways, both groups mitigated angiotensin II’s harmful effects on the vessel wall. With inhibition of ACE—an enzyme that metabolizes bradykinin to inactive metabolites—the bradykinin levels increase. Bradykinin then mediates an increase in nitric oxide, endothelium-derived hyperpolarizing factor, and prostaglandins, which in turn leads to vasodilation and antifibrotic, anti-inflammatory, anti-reactive oxygen species, and antithrombotic effects. 17 Bradykinin additionally opposes the adverse effects of angiotensin II and has a direct advantageous impact. 18 Only ACEIs, and not ARBs, improve coronary outcomes suggesting that ACEIs, but not ARBs, reduce the progression of atherosclerosis and the occurrence of coronary thrombosis. Selective activity of ARBs on the AT1 receptor leaves other angiotensin receptors free to be occupied by angiotensin II whose production is not affected by ARBs. The result is a change in receptors, with AT2 activity increasing and AT1 activity decreasing. Endothelial cells are negatively impacted by AT2 receptors, leading to cell death and stunted development. It appears that a significant difference between ACEIs and ARBs is their effect on endothelial apoptosis, which is reduced by bradykinin and increased by AT2 receptors.
Limitations of this study include retrospective study design, small study cohort, and limited availability of follow-up data. In addition, because of the small sample size, our study lacks the power to formally assess efficacy and safety. In addition, the lack of clinician blinding makes it difficult to generalize our findings.
Conclusion
The transition from ARNI to perindopril went well and without incident. Patients’ functional status was preserved, even if their NYHA class remained unchanged, and their LVEF showed a small but statistically significant improvement. There may be a financial benefit to switching from ARNI to perindopril in India.
Footnotes
Acknowledgement
We thank (hidden for anonymity) assistance.
Ethical Approval and Patient Consent
Patient consent and ethical approval was obtained from institutional ethical committee, and then we followed the Helsinki declaration related to clinical trials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
