Abstract
Complications of cardiac surgery include iatrogenic acute aortic dissection (IAAD), which occurs in 0.04%–0.06% of cases and has poorer results than spontaneous dissection. IAADs generated by thoracotomy are often associated with severe cardiovascular conditions, such as advanced atherosclerosis with three or more coronary artery lesions and persistent heart failure due to multivalvular heart disease. It is possible to experience this complication following any kind of cardiac surgery, including off-pump coronary artery bypass grafting (OPCAB) as well as following endovascular operations. We encountered early postoperative IAAD following OPCAB, which necessitated high-risk reoperation with dismal outcomes. Death rates could be reduced with better access to timely diagnosis and adequate treatment. Avoiding, diagnosing, and treating this potentially fatal complication requires tremendous effort from every member of the heart team.
Introduction
Although iatrogenic acute aortic dissection (IAAD) is considered an uncommon condition, it has the potential to be lethal. It typically manifests during surgery (intraoperative) but can also show up in the early postoperative period (between hours and 30 days) or the late postoperative period after 30 days (defined as occurring from 1 month up to many years1. From 1996 to 2007, IAAD was identified as the underlying cause of mortality in 0.7% of cardiac surgery patients reviewed by the Society of Thoracic Surgeons.2
Unfortunately, whereas Euro SCORE can predict mortality after coronary artery bypass graft (CABG) surgery and valve surgery, it cannot predict the likelihood of IAAD after CABG. The surgical fatality rate for reported cases of IAAD in the cardiac and aortic surgery literature ranges from 30% to 50%3. There are no clear recommendations for Stanford type A aortic dissections in the early postoperative period following substantial intrathoracic surgery, such as CABG. We present the case of an elderly woman who, after a conventional CABG procedure, developed IAAD in the early postoperative period and could not be revitalized, despite rigorous care.
Case Report
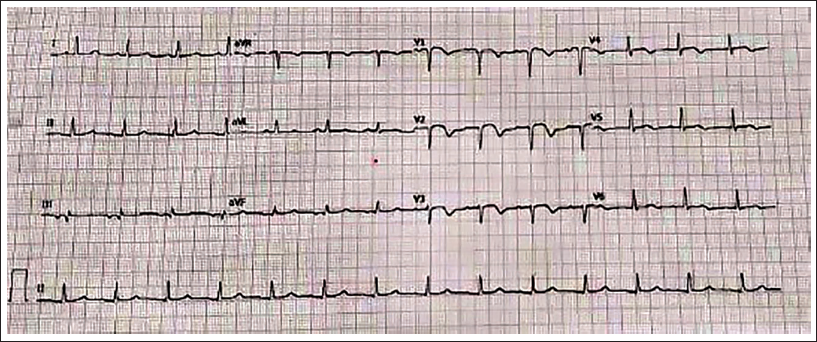
A 67-year-old female, hypertensive and diabetic, was hospitalized after complaining of retrosternal chest pain for the previous two days. Electrocardiography revealed modest ST-segment elevation in the anterior leads, as well as T-wave inversions (Figure 1). Except for elevated levels of high-sensitivity troponin I (2.3 ng/mL; normal range: 0–0.12 ng/mL), all laboratory values were normal. Echocardiography revealed hypokinesia of the distal interventricular septum, apical segments with a 48% ejection fraction, normal right ventricular function, and normal valves. Coronary angiography indicated significant stenoses of the calcified left main coronary artery, the proximal segment of the left anterior descending (LAD) artery, the first diagonal branch, and the mid-segment of the right coronary artery (RCA).
The Electrocardiogram Revealed Mild ST-segment Elevation in KLeads V1-3, as Well as Significant T-wave Inversions in All Precordial (V1-6) Leads. Inferolateral Leads had ST-segment Flattening (II, III, aVF; I, aVL).
After careful deliberation, the cardiac team has chosen to propose a complete surgical revascularization. The patient underwent an elective off-pump coronary artery bypass graft (OPCAB) procedure. The surgery was performed by a midline sternotomy, using the left internal mammary artery for the LAD artery and two saphenous vein grafts for the OM and distal segment of the RCA. Due to the narrow diameter of the diagonals, they were not grafted. Due to the significant atherosclerosis of the aorta, we performed a procedure where we partially clamped the upper part of the aorta, created a single connection at the upper end, and attached a saphenous vein graft to the RCA graft, which was already in place. Upon completion of the procedure, neither direct visualization during surgery nor transesophageal echocardiography detected any abnormalities in the ascending aorta, such as hemorrhage or dissection. The patient’s cardiovascular system remained stable, and they experienced a seamless surgical procedure.
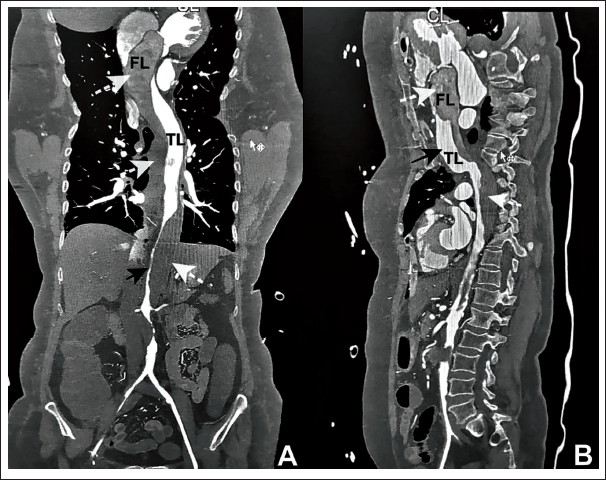
After some time in the intensive care unit, there was a progressive decrease in urine production, accompanied by varying levels of pressure. During the early hours of the first day after surgery, the femoral arterial line became dampened, resulting in the inability to monitor blood pressure. However, the radial line continued to function properly and provided clear pressure tracings. Subsequently, the patient developed anuria within the following hour. The patient exhibited coldness in the lower extremities and warmth in the upper extremities. The patient’s vital signs deteriorated, exhibiting sinus tachycardia, hypotension, and a decrease in oxygen saturation. A bedside transthoracic echocardiogram was performed, revealing the presence of an intimal flap in the ascending aorta. The level of inotropic assistance was increased. Following the temporary improvement in the essential physiological indicators, an urgent computed tomography aortogram was performed, unveiling a type A aortic dissection that extends to the iliac arteries (Figure 2A and B). There was a thrombosed false lumen with compression of the true lumen in the infrarenal aorta, with no perfusion below. Mesenteric vessels were involved in the dissection, resulting in malperfusion, and even the arch vessels were involved except the innominate artery, which was arising from the true lumen and was dissection-free. There were signs of bowel ischemia and splenic infarcts. The patient was taken up for an emergency Bentall procedure after explaining the current situation and the risk involved to the patient’s family.
(A and B) Acute Iatrogenic Stanford Type A Aortic Dissection with Compression of the True Lumen (TL; Thick Black Arrows) by the False Lumen (FL; Thick White Dashed Arrows), Leading to Extensive Malperfusion of the Mesenteric and Iliac Arteries, as Seen on Computed Tomography (CT) Angiography in Anteroposterior (Panel A) and Sagittal (Panel B) Views.
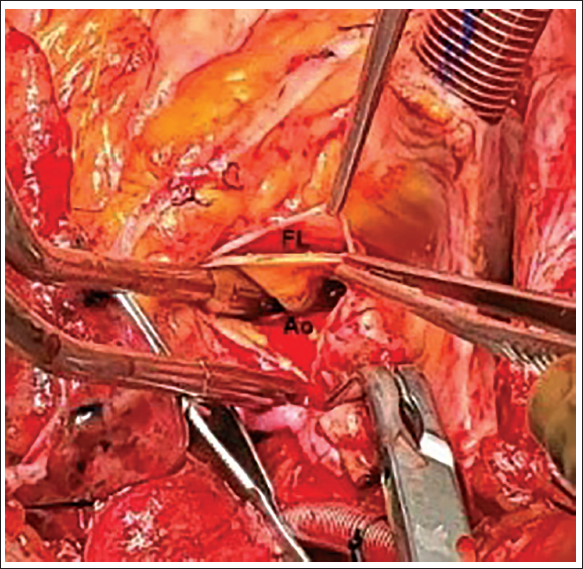
At first, the surgical procedure comprised inserting an 8-mm Dacron graft into the right axillary artery, which was unaffected and originated from the main artery. Upon reevaluation, the sternum was surgically reopened, revealing a dissected ascending aorta. We endeavored to perform a cardiopulmonary bypass (CPB) procedure by inserting cannulas into the axillary artery and right atrium. We were unable to establish flow by axillary cannulation due to very high perfusion pressures exceeding 250 mm Hg, which led to ongoing distention of the left ventricle. Several efforts were made to establish CPB, but both the flow of oxygenated blood into the arteries and the return of deoxygenated blood through the veins were unsuccessful, most likely because the infrarenal aorta was completely blocked. We cannulated the ascending aorta after cross-clamping because we had no other arterial access (Figure 3). Right after the aortotomy was finished, the left ventricle was evacuated. The flap that emerged from the right coronary sinus was visible, although it did not affect the coronary arteries. We successfully identified the location of entry at the proximal anastomosis throughout our research. The CPB procedure failed due to the absence of satisfactory venous return. The heart had cardiac arrest and was unresponsive to resuscitation efforts.
Intraoperative Photograph Demonstrated the Flap of the Dissection Detected in the Posterior Wall of the Ascending Aorta (True Lumen; Ao ) Showing a False Lumen (FL) Separated Using Two Forceps, Which Extended Further Down Up to the Iliac Arteries.
Discussion
Dissection of the aorta may happen spontaneously because of clinical circumstances, such as hypertension, trauma, or medial necrosis.4 A rare but possibly catastrophic IAAD occurs in surgical patients during and after CABG.3 Emory University researchers highlighted the occurrence of this problem in the initial postoperative period following CABG in 1978.5 There is an imminent risk of death if the dissection is not recognized and treated quickly.
When detected within the first two weeks after CPB, IAAD is considered early postoperative IAAD, and when diagnosed more than 30 days after cardiac surgery, IAAD is considered late postoperative IAAD.1, 6 In the period 1978–1990, IAAD varied from 0.04% to 0.29% of cardiac patients (0.12%–0.16%).7, 8 IAAD occurred in 90% of patients during surgery.3, 9 There was just one patient (0.06%) with IAAD among 1,233 cardiac surgical operations at our institution between July 2019 and January 2021, similar to the incidence reported by Shea et al. 3
While preliminary research suggested that OPCAB was riskier than on-pump CABG, subsequent research using larger samples and more databases debunked this.7, 8 Any of several sites, including the cardioplegia, the aortic cannulation, the aortic cross-clamp, the partial occlusion clamp, the aortotomy, or the vein anastomosis, can be the site of a tear in a patient with retrograde IAA.8 Although cannulation and aortic cross-clamping are not required for OPCAB, a side-biting clamp, and proximal anastomoses can enhance the risk of dissection due to a pulsatile arterial pressure pattern.7 Such invasive procedures done directly on the ascending aorta wall may result in intimal injury, splitting, and, eventually, dissection, which is exacerbated by underlying medical disorders, such as uncontrolled hypertension, cystic medial-necrosis, and/or atherosclerosis. 5
Hand inspection of the ascending aorta may assist the surgeon in determining the locations of the aortic cannula, saphenous vein anastomosis, and cross-clamping.8 Our patient had hypertension, but a physical examination during cannulation did not reveal aortic plaque. Preventing early postoperative dissection in CABG requires avoiding aortic clamping with a beating heart, maintaining low systemic blood pressure during proximal anastomoses (80 mm Hg), and avoiding partial occluding clamps. By carefully monitoring perfusion pressure throughout the intervention, preventing excessively high jet pressures with vasodilators, and relocating the arterial cannula, we feel that the risk of IAAD can be minimized.8,10
Due to the high risk and potential fatality of intraoperative aortic dissection, it is important to closely monitor patients for any pathological changes in the aorta wall until the surgery is finished. Urgent repairs are necessary to prevent the development of dissection growth and its associated effects.5, 10 The position and extent of the intimal tear dictate the shape of the repair. Patients with localized dissection should undergo primary repair. Patients with substantial injuries require prompt patchplasty or graft interposition.4, 9
Prosthetic grafts can be used to replace aortas that have become dilated, calcified, or affected by extensive atherosclerosis. Extra-corporeal membrane oxygenation (ECMO) was employed in specific reported instances following surgery to effectively manage IAAD. The community remains polarized about the comparative merits of implementing ECMO as a last-resort treatment for critically ill patients, weighing the risks against the potential advantages. In the case of IAAD, patient outcomes are improved by both rapid diagnosis and surgical therapy. 9
Conclusion
Iatrogenic acute aortic dissection, is a severe and unexpected complication that can occur during heart surgery. The occurrence of IAAD has been linked to age, hypertension, and atherosclerosis of the ascending aorta. Although IAAD is rare, it is crucial to take preventive measures due to the increasing number of older individuals requiring heart surgery. Our example stands out not due to the IAAD but rather because of the irreparability and technological difficulties involved in operating the CPB. Ultimately, while the existing evidence suggests that minimally invasive surgery carries no risks, more comprehensive randomized research is necessary to establish definitive results.
Contributorship Statement
PJ and GPK are responsible for the writing manuscript, collection of data, and preparation of the article. SSK is a chief cardiothoracic surgeon who operated patient and GA is an assistant surgeon who helped in writing a part of the surgical details of the case.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from a patient included in the study, and additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
