Abstract
Background
Sacubitril/Valsartan is one of the pillars of heart failure (HF) management. Little is known about its uses or impact in real-world practice in the Indian population. We evaluated the effectiveness and Safety of Sacubitril/Valsartan in Heart Failure in India.
Methods
This retrospective analysis involved newly diagnosed adult patients of HF (class I–IV, per physician clinical assessment and discretion), treated in a tertiary care hospital during period of February–April 2023 and received Sacubitril/Valsartan. N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels were measured in addition to demographics, clinical symptoms, comorbidities, and left ventricular ejection fraction (LVEF). Following three months of Sacubitril/Valsartan medication, changes in LVEF, NT-proBNP levels, and clinical symptoms (graded from 1 to 10, with 10 being the maximum severity) were assessed.
Results
Out of 60 patients eligible for the study, 57% were males and 43% were females. Mean body weight was 60.26 ± 7.00 kg, while mean BP was 130/81 mmHg. 60% patients had both type 2 diabetes and hypertension as comorbidity. There was 23% improvement in LVEF (from mean LVEF of 34 to 42, P < .0001), with a significant decrease in NT-proBNP levels (mean 1220.5 pg/mL to 118.1 pg/mL, P < .0001) after treatment with Sacubitril/Valsartan for 3 months, resulted in marked improvement in clinical symptoms of breathlessness (mean score decreased from 6 to 2, P < .05), edema (mean score decreased from 5.5 to 1.8, P < .05) and palpitations (mean score decreased from 2.4 to 1.1, P < .05. 50 mg BD was the most commonly used dose of Sacubitril/Valsartan (70% patients), followed by 100 mg BD (25% patients) and rest (5%) with 200 mg BD dose. Treatment with Sacubitril/Valsartan was well tolerated, with <5% patients reporting hypotension.
Conclusion
The findings of this real-world study suggest Sacubitril/Valsartan was associated with an increase in ejection fraction translating into symptomatic improvement in Indian patients of HF.
Introduction
Heart failure (HF) is a global health concern and a major cause of morbidity and mortality. Additionally, HF significantly increases the cost burden on healthcare systems around the world. 1 Globally, 64.3 million people have been affected with HF in 2017; the highest burden of HF was noticed in Middle East, and North Africa ranging from 1133 to 1196 per 100,000 population, whereas lower burden was notified in Southeast Asia ranging from 498 to 595 per 100,000 population. With an estimated prevalence of 1.3–4.6 million people and an incidence of 0.5–1.7 per 1000 people annually, India has the highest burden of HF in Asia. 2 Modern HF medications target different pathways, like suppressing the breakdown of the renin-angiotensin-aldosterone system and endogenous natriuretic peptides at the same time.3, 4 Additionally, a number of additional potential drug-targeting methods have been studied, such as guanylate cyclase stimulators, sodium/glucose co-transporter inhibitors, and cardiac myosin activators.4, 5 Though numerous medicines aim to block neurohumoral pathways, the pathophysiology of HF may entail overstimulation of these processes. As a result, HF continues to be a leading cause of morbidity & mortality. 1
The US FDA and the European Medicines Agency (EMA) have approved sacubitril/Valsartan, a medication that contains an angiotensin receptor blocker and neprilysin inhibitor (ARNI), for the treatment of HF patients with reduced ejection fraction (HFrEF).6–8 Compared with already established drugs (e.g., Enalapril), ARNI usage had lowered cardiovascular deaths by 20% and all-cause mortality by 16%. 1 In HFrEF patients who were hospitalized for acute decompensated HF, the initiation of Sacubitril/Valsartan treatment led to a greater reduction in the levels of NT-proBNP versus enalapril therapy. 9 Sacubitril/Valsartan was recommended as part of optimal therapy by the European Society of Cardiology and the American Heart Association/American College of Cardiology clinical guidelines. Class 1A recommendations were primarily indicated in patients with persistent HF who had reduced ejection fraction.6–8
The safety and efficacy of Sacubitril/Valsartan in Indian patients with HF, however, are not well understood. Therefore, the current study aimed to evaluate the effectiveness and safety of Sacubitril/Valsartan in Indian patients with HF.
Material and Methods
This hospital-based, retrospective, cohort study was conducted at a tertiary care healthcare center in India. Patients aged >18 years, newly diagnosed with HF (NYHA class I–IV) 10 who received Sacubitril/Valsartan treatment (Tab. Sacuval, Alkem Laboratories Ltd.) twice daily for at least three months (Feb–Apr 2023) were obtained. These files were screened properly, and the data was collected for demographics, clinical assessment for symptoms and vitals, comorbid conditions, LVEF, NT-ProBNP. Patients with missing data for any of the above parameters were excluded from analysis. Out of the 155 records screened, 60 patients satisfied inclusion criteria thus were included in the study. We evaluated changes LVEF, NT-proBNP levels, and severity of clinical symptoms (breathlessness, edema, and palpitations; severity assessed on a scale of 1–10 with 10 being the worst) at baseline and after three months of treatment with Sacubitril/Valsartan. Other medications for HF were continued as per physician’s discretion.
Statistical Analysis
Data were compiled and analyzed using the statistical software SPSS version 20 (IBM Corp, Armonk, USA) and MS Excel. Categorical variables were represented by frequency tables, whereas continuous variables were presented in the mean ± SD form. Normally distributed variables were analyzed using parametric tests of significance (Student’s t-test). Association between categorical variables was tested using non-parametric tests (Chi-square test). P ≤ .05 was considered statistically significant.
Results
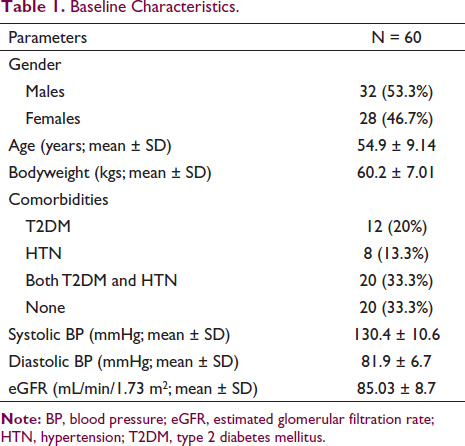
The study comprised 60 patients, with a mean age of 54.9 ± 9.1 years (32 males and 28 females). Table 1 depicts the distribution of patients based on baseline characteristics. Majority (53.3%) of the patients were aged/ ≥55 years. The mean body weight of the patients was 60.2 ± 7.01 kgs, and most patients (33.3%) had both type 2 diabetes and hypertension (Table 1). The mean estimated eGFR of the patients was 85.03 ± 8.7 mL/min/1.73 m2. At baseline, majority of the patients belonged to NYHA class II (40%) and III (26.7%), while few patients belonged to NYHA class IV (13.3%). 70% patients received 50 mg of Sacubitril/Valsartan twice daily, 25% patients received 100 mg twice daily and rest 5% received dose of 200 mg twice daily.
Baseline Characteristics.
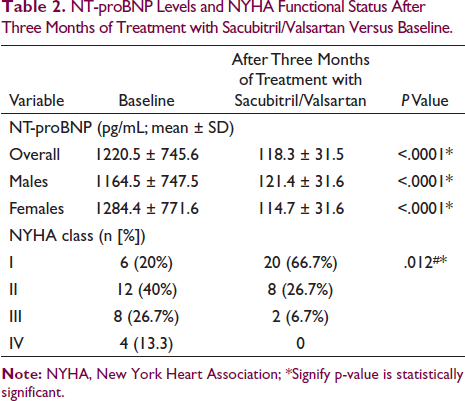
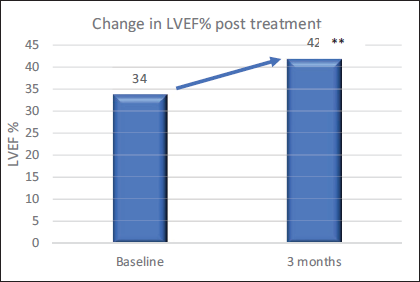
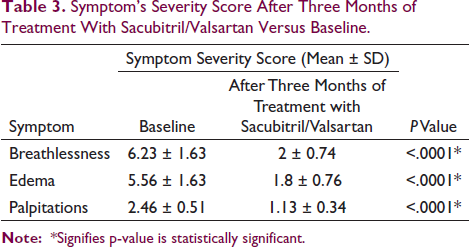
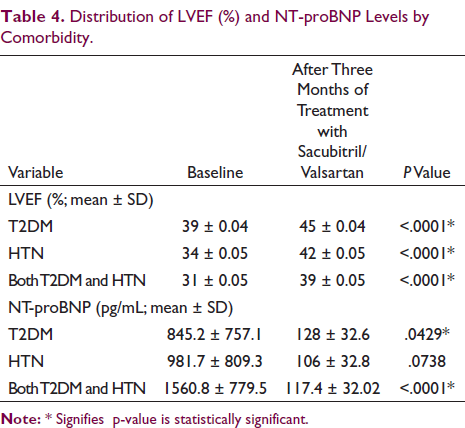
After three months of treatment with Sacubitril/Valsartan, a marked improvement was observed in functional status according to NYHA class; patients in class IV at baseline moved to class II (50%) and III (50%) after three months of treatment (Table 2). A significant 23% improvement in mean LVEF (%) was seen from 34 at baseline to 42 after three months of treatment (P < .0001; Figure 1). After three months of treatment, there was a significant drop in mean NT-proBNP levels from 1220.5 pg/mL at baseline to 118.3 pg/mL (P < .001). A marked reduction in symptom severity score was observed for all three symptoms (Table 3). Sacubitril/Valsartan was well tolerated with <5% patients reporting hypotension; mean BP was 130/81 mmHg at baseline versus 119/76 mmHg after three months of treatment. Table 4 depicts the distribution of LVEF and NT-proBNP levels (at baseline vs. post-treatment) by comorbidity; a significant difference was found in findings at baseline versus after three months of treatment (P < .05). Comorbidities had no impact on efficacy of the treatment (Table 4).
NT-proBNP Levels and NYHA Functional Status After Three Months of Treatment with Sacubitril/Valsartan Versus Baseline.
Symptom’s Severity Score After Three Months of Treatment With Sacubitril/Valsartan Versus Baseline.
Distribution of LVEF (%) and NT-proBNP Levels by Comorbidity.
Discussion
Sacubitril/Valsartan medication for three months significantly reduced the levels of NT-proBNP in this retrospective investigation of HF patients, and there was a notable improvement in LVEF and clinical BP in a real-world scenario. With the promising results from various clinical studies,11–13 For individuals suffering from HF, sacubitril/valsartan is recommended as a class 1A medication.6–8 Furthermore, there is ongoing discussion regarding the efficacy of Sacubitril/Valsartan in Asian patients with HF, as its results in trials involving Asian patients did not reach statistical significance. 14 In this study, treatment with Sacubitril/Valsartan for three months was well tolerated with only <5% patients reporting hypotension. No major difference in eGFR levels was found at the end of three months of Sacubitril/Valsartan therapy. The results of the TITRATION trial are consistent with the current study’s findings on Sacubitril/Valsartan tolerability in the Indian population. 13 In the current study, majority of patients received 50 or 100 mg twice daily; with good clinical and echocardiographic outcomes. Findings of our study are similar to various studies in Asian patients, demonstrating significant reduction in NT-proBNP and increased LVEF without serious adverse effects.15–17
In the current study, a significant improvement in mean LVEF (%) was found with 23% from 34 ± 0.04 at baseline to 42 ± 0.04 after three months of treatment (P < .0001); these findings are in concordance with the findings of previous studies.18–21 A significant rise in LVEF was observed in HRrEF patients treated with Sacubitril/Valsartan for more than six months, with a notable increase from 35.4 ± 8.9 at baseline to 43.0 ± 12.2 after six months of treatment (P < .001), according to prospective, observational cohort research done in China by Hu J et al. 15 In another prospective study (Kim H, et al.; Korea) conducted in HRrEF patients who were treated with Sacubitril/Valsartan, significant increase in LVEF (from 26.4 ± 6.2 to 35.7 ± 12.0%, P < .001) was demonstrated. 17 A marked improvement in NYHA functional class was also observed with 66.7% in class I and 26.7% in class II, and only two (6.7%) patients in class III at the end of three months; similar results were published by Khan MN et al. in HFrEF patients. 1
Sacubitril/Valsartan has been shown to be effective in a real-world scenario for HF patients with lower ejection fraction in a few recent studies. Consistent with our research, Sacubitril/Valsartan therapy was found to improve NYHA functional class, as well as reduce NT-proBNP levels and improve several hemodynamic parameters in HF patients with reduced ejection fraction in a real-world two-year follow-up study by Armentaro et al. 22 In another study on non-diabetics and diabetics with HF reduced ejection fraction, a potential role of Sacubitril/Valsartan treatment was reported in HF patients with metabolic comorbidities; a persistent metabolic improvement has been noticed over the period of 12 months. 23 In the current study, comorbidities (type 2 diabetes and hypertension) had no impact on the efficacy of the treatment. Various studies reported the discontinuation of Sacubitril/Valsartan therapy due to side effects, such as hypotension and hyperkalemia. 24 However, none of the patients discontinued therapy because of hypotension in the current study as only a few patients (<5%) reported hypotension in the current study.
Our study has certain limitations in being a retrospective study with a limited sample size and lack of a control group. Also, the shorter follow-up period remained an important limitation. Despite the same, this study demonstrated the effectiveness and safety of Sacubitril/Valsartan in Indian patients with HF.
Conclusions
In Indian adult patients with HF, Sacubitril/Valsartan is effective in improving ejection fraction and clinical outcomes and is well tolerated.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Declaration of Patient Consent
The informed consent requirement was waived by the ethical committee due to the retrospective design of the study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
