Abstract
Background:
Sacubitril/valsartan (S/V) is a cornerstone treatment for heart failure (HF). Beneficial effects on hospitalization rates, mortality, and left ventricular remodeling have been observed in patients with heart failure and reduced ejection fraction (HFrEF). Despite the positive results, the influence of S/V on renal function during long-term follow-up has received little attention.
Aims:
We investigated the long-term effects of S/V therapy on renal function in a large cohort of patients with HFrEF. Additionally, we examined the effects of the drug in patients with chronic kidney disease (CKD) compared to those with preserved renal function and identified primary risk characteristics
Methods:
We studied 776 outpatients with HFrEF and left ventricular ejection fraction (LVEF) <40% from an observational registry of the Italian Society of Cardiology, all receiving optimized standard-of-care therapy with S/V. The patients were included in a multicentric open-label registry from 11 Italian academic hospitals. Kidney function was evaluated at baseline, after 6 months of S/V, and at 4 years. Patients were followed-up through periodic clinical visits.
Results:
During a 48-month follow-up period, 591 patients remained stable and 185 patients (24%) experienced adverse events (85 deaths and 126 hospitalizations). S/V therapy marginally affects renal function during the follow-up period (estimated glomerular filtration rate (eGFR) at baseline 72.01 vs eGFR at follow-up 70.38 ml/min/m2, p = 0.01; and creatinine was 1.06 at baseline vs 1.10 at follow-up, p < 0.04). Among patients who maintained preserved renal function, 35% were in Dose 3 and 10% dropped out of S/V therapy (p < 0.006). Univariate analysis showed that Drop-out of S/V (HR 2.73 [2.01, 3.71], p < 0.001), history of previous HF hospitalization (HR 1.75 [1.30, 2.36], p < 0.001), advanced NYHA class (HR 2.14 [1.60, 2.86], p < 0.001), NT-proBNP values >1000 pg/ml (HR 1.95[1.38, 2.77], p < 0.001), furosemide dose >50 mg (HR 2.04 [1.48, 2.82], p < 0.001), and creatinine values >1.5 mg/dl occurred during follow-up (HR 1.74 [1.24, 2.43], p < 0.001) were linked to increased risk. At multivariable analysis, increased doses of loop diuretics, advanced NYHA class, creatinine >1.5 mg/dl, and atrial fibrillation were independent predictors of adverse events.
Conclusion:
Long-term S/V therapy is associated with improved outcomes and renal protection in patients with HFrEF. This effect is more pronounced in patients who tolerate escalating doses. The positive effects of the drug are maintained in both CKD and preserved renal function. Future research may study the safety and underlying causes of current protection.
Plain language summary
Despite the beneficial results, the influence of S/V on renal function during long-term follow-up has received little attention. We investigated the long-term effects of S/V therapy on renal function in a large cohort of patients affected by HFrEF.
Introduction
Heart failure (HF) is the primary cause of morbidity and mortality, which drives up the expense of medical care. 1 When compared to valsartan or other renin-angiotensin system inhibitors (RASi), sacubitril/valsartan (S/V), a commonly used medication in cardiology, has been shown in multiple studies to reduce hospitalization and mortality in patients with heart failure and reduced ejection fraction (HFrEF).2,3 Treatment with S/V is able to reverse left ventricular remodeling and enlargement, as well as to reduce left ventricle parietal stress and congestion, in comparison to RASi or angiotensin-receptor blockers (ARB) alone.3–5 The main effects are related to reduced cardiac arrhythmic events and improved cardiac function.
Furthermore, the implementation of S/V following an early post-discharge period demonstrated favorable outcomes in terms of NT-pro B-type natriuretic peptide (NT-proBNP) reduction in clinical status and a reduction in hospitalization.6,7 S/V has been shown to have positive effects on cardiac structure and function, while its effects on renal function have received less attention. Additionally, S/V demonstrated to have a decreased all-cause mortality in symptomatic HFrEF patients with chronic kidney disease (CKD) when compared to angiotensin-converting enzyme inhibitor (ACEI)/ARB; however, these findings need to be confirmed by prospective randomized studies. In this study, the ACEI/ARB group had a significantly higher incidence of all-cause mortality associated with an increased renal adverse event rate in patients with CKD, while either the S/V and the ACEI/ARB groups did not differ in clinical or renal outcomes in patients without CKD. The protocol’s retrospective nature, the inability to distinguish between various renal dysfunction phenotypes, and the relatively narrow scope of the follow-up research; however, limit the study’s ability to provide a definitive analysis of the precise impact of S/V on renal function over longer time periods than 12 months.
Recently, the use of S/V has expanded to include patients with heart failure and preserved ejection fraction (HFpEF). Results from the PARAGON trial showed that patients with EF less than 57% and who were female were more likely to benefit from S/V, with a significant combined endpoint reduction in hospitalization and cardiovascular mortality. Studies have also been conducted on the impact of angiotensin-neprilysin inhibition on renal function in this population. According to this study, S/V attenuates the decline of estimated glomerular filtration rate (eGFR) and reduces clinically relevant kidney events similarly among patients with HFpEF with and without diabetes. 8 Despite these encouraging findings, a thorough analysis of the long-term effects of S/V on the kidneys based on concomitant disorders, baseline clinical features, and therapy combinations is currently missing. Therefore, according to current literature, there is no clear evidence regarding the eGFR and creatinine thresholds in which patients can find effective benefits from drug administration.
Our study’s objective is to assess the long-term effects of angiotensin-neprilysin inhibitor therapy on renal function in a cohort of patients with HFrEF and left ventricular ejection fraction (LVEF) < 40% who are part of an observational registry approved by the Italian Society of Cardiology. During a 48-month follow-up period, we assessed in detail how the study patients responded to S/V therapy, taking into account mortality and complications (HF recurrence and hospitalization for HF) in both patients who continued treatment (Cont) and patients who stopped the S/V therapy. In addition, we examined the impact of medication on individuals with CKD in comparison to people with maintained renal function and the potential consequences of S/V
Methods
Study design
This is a prospective study investigating ambulatory patients with HFrEF and LVEF < 40% with optimized standard-of-care therapy taking S/V therapy. The patients were included in a multicenter, open-label registry from 11 Italian academic hospitals before starting S/V therapy (NCT04397302) from December 2016 to October 2019. As required by our protocol, patients underwent echocardiography at baseline and 8–12 months after treatment. Detailed information on study protocol, patient medical history, including medications and laboratory data, as well as inclusion and exclusion criteria have been described in the previous paper. 9
Inclusion criteria
Patients were included if they had history of HF, they met at least one of the following symptoms exertional dyspnea, orthopnea, peripheral edema, and major fatigue; associated with an elevation of BNP > 100 pg/ml or NT-proBNP > 500 pg/ml, and echocardiographic evidence of systolic dysfunction with LVEF < 40%. They experienced clinical stability on a stable dose of loop diuretic at least 2 weeks preceding the study start, optimized medical therapy (defined as treatment for ⩾6 months on maximum tolerated dose of an ACEI or angiotensin-receptor antagonists (ARBs) and a beta blocker, possibly in association with a mineralocorticoid-receptor antagonist (MRA).
Exclusion criteria
We did not include patients with recent heart valve replacement, recent myocardial infarction, or coronary artery bypass graft (<3 months), history of pulmonary embolism, idiopathic pulmonary arterial hypertension, neoplastic, hematologic, and immune diseases. Subjects with end-stage renal disease or renal replacement therapy, systolic blood pressure (BP) <100 mmHg, and creatinine levels >4 mg/dl, were also excluded.
Data collection
Patients were evaluated every 3 months during early enrollment period during the first year and every 6 months for the resting time in stable patients. Kidney function was assessed at baseline after 6 months and at 4 years of follow-up. The total mean observational follow-up was 52 months. Demographic, clinical, laboratory, and echocardiographic data were collected at baseline; clinical, laboratory, and echocardiographic data were re-evaluated at the reassessment. Creatinine values and NT-proBNP were collected at baseline and during follow-up visits for a mean follow-up of 4 years. Initial study population consisted of 823 subjects, 47 patients were lost during follow-up and 776 completed the study (Supplemental Figure 1). The study was conducted according to institutional guidelines, national legal requirements, European standards, and the revised Declaration of Helsinki.
Treatment protocol
In patients taking ACEI, S/V was started after a washout period of 36-h at a preferential dose of 49/51 mg b.i.d., or 24/26 mg b.i.d. for those taking a low dose of ACE inhibitors. S/V dose was tentatively doubled every 2–3 weeks to reach the target maintenance dose of 97/103 mg b.i.d., except in patients with systolic BP less than 100 mmHg or who developed drug-related adverse events (symptomatic hypotension, hyperkalemia >5.5 mEq/l, or a decrease in eGFR (measured throughout MDRD method) to). CKD was assessed according to Kidney Disease: Improving Global Outcomes (KDIGO) classification. 10 Patients with eGFR < 60 ml/min/m2 and creatinine value >1.4 mg/dl found in two consecutive blood tests were classified as CKD. All patients provided written informed consent for anonymous collection and publication of their clinical data.
Statistical analysis
It is performed as follows: If normally distributed, quantitative variables are presented as mean ± standard deviation (SD) or as median and interquartile range (Q1–Q3) according to normality distribution evaluated by Kolmogorov-Smirnov test. Qualitative variables are reported as absolute frequencies and percentages. To evaluate differences in quantitative variables between two groups Student’s t test or Mann-Whitney test was carried out according to the normality distribution. Chi-squared test was performed to evaluate the association between qualitative variables. Kruskal-Wallis test was performed to compare creatinine and eGFR between four groups (i.e., preserved renal function, stable CKD, worsening renal function (WRF), and improvement renal function). Wilcoxon test was performed to compare eGFR and creatinine at baseline with follow-up values. Univariable Cox regression was carried out. All the significant variables at univariate analyses and interaction terms (CKD status × S/V dosage) were included in the multivariable regression. A stepwise procedure was then carried out. Hazard ratio (HR) and their 95% confidence interval (95% CI) were estimated. Multicollinearity was tested based on variance inflation factors (VIF). A p < 0.05 was considered statistically significant. Analyses were carried out with R version 4.0.0 (SPSS software).
Results
At baseline, there were 776 patients (653 males and 123 females) with HFrEF (mean FE value of 30% ± 6.57) receiving S/V treatment. At baseline patients in RASi therapy were 635 (82% of the total population), patients in beta-blockers were 719 (93%), 534 (69%) took MRA, 127 (16%) ivabradine, 84 (11%) digoxin, and 19 (2%) were taking sodium-glucose cotransporter 2 inhibitors (SGLT2i). There were 671 (86%), or patients, taking furosemide at baseline. Around 308 patients (39%) received S/V therapy at a dosage of 24/26 mg SV (Dose 1), 260 patients (34%) received 49/51 mg of SV (Dose 2), and 208 patients (27%), received a higher dose (Dose 3) of SV 97/103 mg. At enrollment, 339 (44%) patients presented a previous heart failure (pre-HF) hospitalization, 349 (45%) had a history of myocardial infarction, and 253 (32.6%) had CKD. The only variable with missing data during follow-up was NT-proBNP (118 out of 776 were missing values). All the clinical and therapeutic characteristics of enrolled population are presented in Table 1.
Characteristics of the study population at baseline.

ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin-receptor blockers; NYHA, New York Heart Association; S/V, sacubitril/valsartan.
Over a 48-month follow-up period, 591 patients remained stable and free from complications (Stable group), whereas 185 patients (24%) experienced adverse outcomes, including hospitalization and/or death (Complicated group). In this group, 126 patients underwent HF hospitalization and 85 died. Worsening heart failure requiring urgent visits was observed in 40 patients, 21 subjects in Stable group and 19 patients in the Complicated group (3.6% vs 10.3% respectively, p = 0.001). Differences in mean age were found between the two groups (63.28 ± 11.59 vs 68.06 ± 11.79). At the baseline, 463 (60%) suffered from hypertension, 253 (33%) had CKD, 169 (22%) affected by chronic obstructive pulmonary disease and 250 (32%) suffered from type 2 diabetes, 157 (20%) had atrial fibrillation (AF) and 349 (45%) presented previous myocardial infract. Similar values at baseline were found in Stable versus Complicated group in terms of body mass index, BP values, and heart rate (HR) are reported in Table 2.
Baseline characteristics of the study sample divided into Stable group without adverse outcomes and in Complicated group presenting adverse events (hospitalization and/or death) during follow-up.
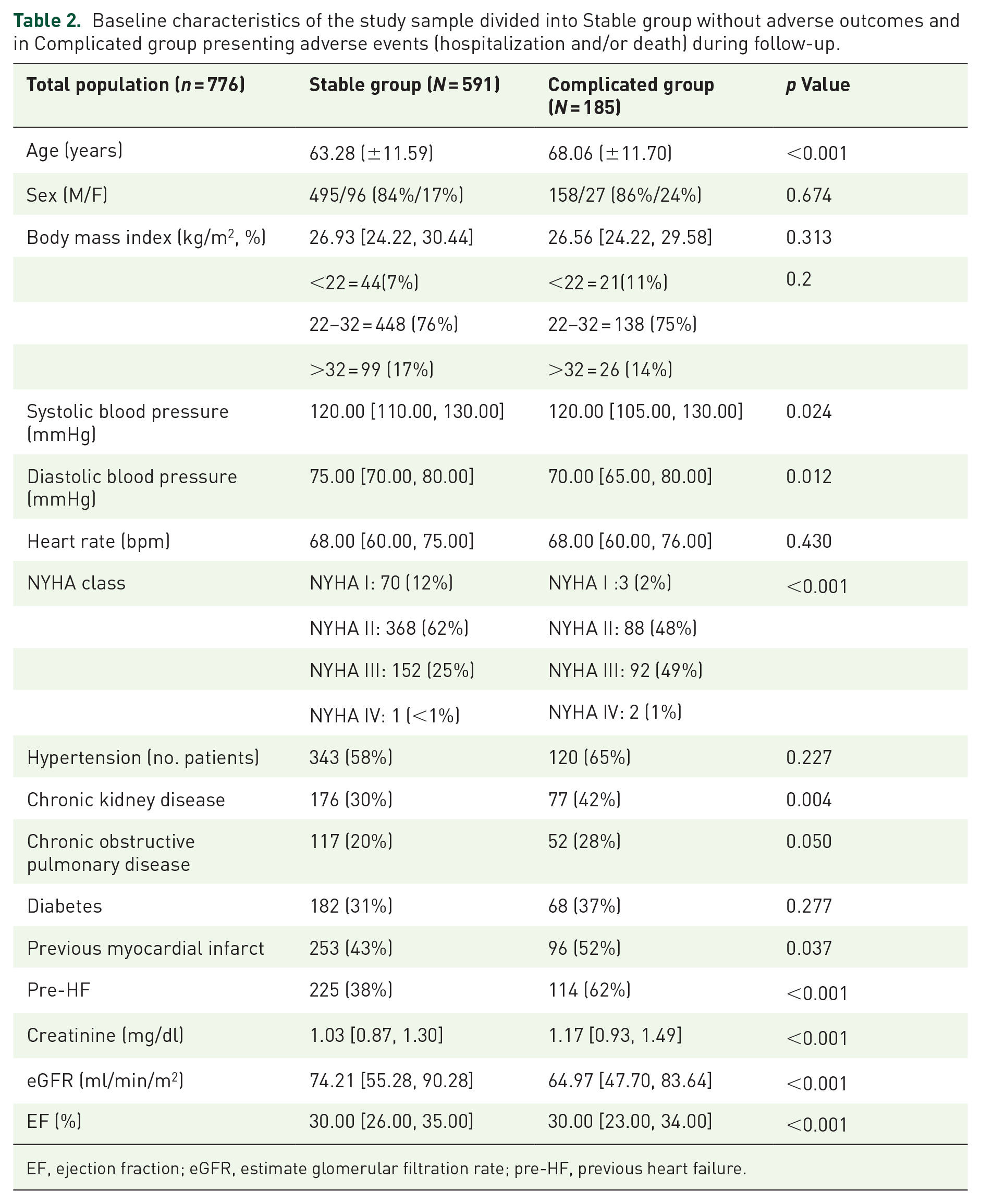
EF, ejection fraction; eGFR, estimate glomerular filtration rate; pre-HF, previous heart failure.
At baseline, in the whole population, the median creatinine value was 1.06 [0.89, 1.35] mg/dl, median eGFR was 72.01 [53.13, 89.13] l/min/m2. At the follow-up median creatinine was 1.10 [0.90, 1.34] (p = 0.04 vs baseline) and eGFR was 70.38 [54.33, 87.81] ml/min/m2 (p < 0.01 vs baseline). No significant clinical changes in renal function were observed during S/V treatment with small variations in creatinine and eGFR values.
Effects of S/V therapy on outcome according to CKD
No differences in terms of mortality were found between the CKD (CKD group) compared to subjects with preserved renal function (No CKD group; 13.8% vs 9.6%, p = 0.096). Hospitalization for HF during the follow-up was found in 74 (14%) in No CKD group compared with 52 in CKD group (20.6%; p = 0.03). Use of loop diuretics at baseline was higher in CKD patients (436 vs 235/83% vs 93%, p < 0.001). Indeed, patients with CKD presented an increased dose of furosemide at baseline compared to patients with preserved renal function (62.5 mg [25, 100] vs 50 mg [25, 75], p < 0.001; Table 3).
Principal differences in characteristics between patients with preserved renal function (No CKD group) and chronic kidney disease patients (CKD group) at baseline.

ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin-receptor blockers; eGFR, estimate glomerular filtration rate; S/V, sacubitril/valsartan.
During the follow-up, an improvement of NYHA class was observed in most of patients in both CKD and No CKD groups with a similar reduction of NT-proBNP (CKD = 1565.72 [788.87, 3455.75] pg/ml at baseline vs 1202.00 [492.53, 2761.75] pg/ml follow-up; No CKD = 901.50 [435.00, 1916.50] pg/ml at baseline vs 631.00 [229.00, 1246.00] pg/ml at follow-up). Diuretic administration was reduced in both groups during follow-up: CKD at baseline: 436 (83%) vs 384 (73%); No CKD: 235 (93%) vs 212 (84) at follow-up. This is consistent with a reduction in furosemide amount decreased in both groups (No CKD = 50 mg [25, 75] to 25 mg [0, 50]; CKD = 62.5 mg [25, 100] vs 50 mg [25, 100]). No significant changes in renal function were observed during S/V treatment with small variations in creatinine and eGFR values. Indeed, WRF was observed in few patients with no significant differences between groups (3% of No CKD patients vs 4% of CKD, p = 0.2). Similar findings were revealed regarding subjects who discontinued S/V. Moreover, increasing S/V amount from the starting dose up to the therapeutic target was possible in several patients with CKD without significant renal function deterioration. Those subjects experiencing a persistent WRF demonstrated a higher adverse event rate compared to No CKD patients (HR 1.58 [1.17, 2.12], p = 0.002; Figure 1).

Difference in survival probability between patients affected by chronic kidney disease (CKD) and patients with preserved renal function (No CKD).
Trend analysis of renal function demonstrated that most of the patients kept normal renal function (459 out of 523 patients) and a CKD worsening in patients with baseline renal dysfunction in 198 out of 253 patients. Additionally, few patients showed significant renal function changes during follow-up (64 developed WRF and 55 had significant renal function improvement). Table 4 describes the effects of S/V baseline dose and S/V follow-up dose effects on all categories.
Descriptive analysis of patients with preserved renal versus those with worsening renal function occurrence and improvement of renal function during follow-up according to S/V dose at baseline and after 1 year.

CKD, chronic kidney disease; eGFR, estimate glomerular filtration rate; S/V, sacubitril/valsartan.
Predictors of adverse outcome
Univariate analysis revealed that patients with CKD revealed an increased rate of adverse events during the follow-up period (HR 1.74 [1.24, 2.43], p < 0.001). Drop-out of S/V was related with prognosis (HR 2.73 [2.01, 3.71], p < 0.001) as well as pre-HF (HR 1.75 [1.30, 2.36], p < 0.001), AF (HR 1.83 [1.33, 2.52], p < 0.001, NYHA class (HR 2.14 [1.60, 2.86], p < 0.001), NT-proBNP values >1000 pg/ml (HR 1.95[1.38, 2.77], p < 0.001), and assumption of furosemide dosage >50 mg (HR 2.04 [1.48,2.82], p < 0.001). Conversely the use of beta-blockers demonstrated protective effects (HR 0.48 [0.32, 0.74], p < 0.001; Table 5).
Results of Cox-Univariate analysis relating to adverse outcomes during 52 months of follow-up days.

AF, atrial fibrillation; CKD, chronic kidney disease; COPD, Chronic obstructive pulmonary disease; MRA, mineralocorticoid-receptor antagonist; SGLT2i, sodium-glucose cotransporter-2 inhibitors.
Similarly, higher doses of S/V were associated with significant adverse event reduction (Dose 3: HR 0.30 [0.19, 0.45]). Multicollinearity was tested both for the model with all variables and for the final model. Initially, the VIF values for the significant variables entered had a range between 1.04 and 1.99. With the stepwise procedure, the range was reduced to 1.04–1.14. Multivariate analysis was performed on 658 patients with completed information. Multivariate analysis confirmed that elevated doses of loop diuretics, advanced NYHA class, and AF were all features associated with increased risk. Whereas mid and high S/V amounts, the contemporary use of beta-blockers was related to better outcomes. Patients in high dose of S/V (Dose 3) presented a better prognosis compared to both patients in Dose 1 and 2 (Dose 2: HR 0.63 [0.46, 0.88], p = 0.007), Dose 3: HR 0.38 [0.24, 0.60], p < 0.001). No significant interaction was observed between CKD and S/V dose (Table 6; Figure 2).
Significant data related to survival probability in hazard ratio and 95% CI of multivariable analysis adjusted for risk factors CKD, ejection fraction, and treatment.

CKD, chronic kidney disease; S/V, sacubitril/valsartan.
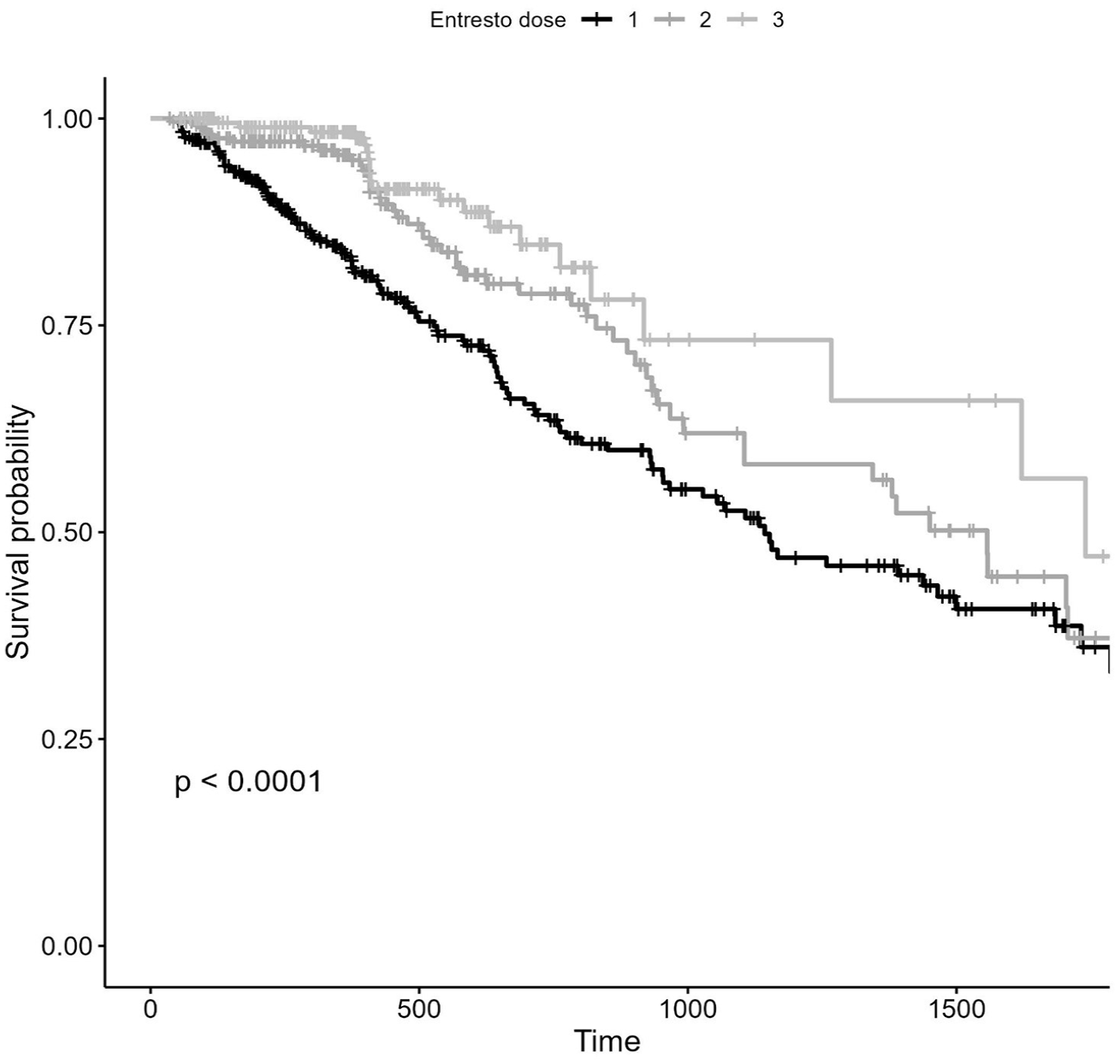
S/V dose-related prognosis evaluated in the time (days). In a follow-up of 52 months, patients taking high dose of S/V (Dose 3) presented a reduction in adverse events compared to intermediate dose (Dose 2) and low dose (Dose 1)
Effects of treatment discontinuation and up-titration
A total of 102 subjects (13% of cohort, 85 males, 17 females) dropped out S/V treatment. The main reason for discontinuation was related to significant BP reduction: 44 had symptomatic hypotension, 17 had asymptomatic hypotension, 12 had WRF, 3 had cough, 2 had gastrointestinal disorders, 1 had skin rash, 10 had hyperkalemia, and 14 had drug intolerance or angioedema.
Patients who Cont were younger compared to patients who discontinued S/V (63.70 SD ± 11.7 vs 69.18 ± 10.9, p < 0.001) Among the Drop-out group 43 patients deceased during follow-up, compared to 42 patients in the Cont group (42% vs 6%, p < 0.001). Additionally, an increase in hospitalization for HF was found in Drop-out patients compared to Cont (95 vs 31 14% vs 30.4%, p < 0.001; Figure 3). During follow-up, Drop-out group presented high values of creatinine and decreased eGFR compared with Cont group (creatinine: 1.10 [0.90, 1.30] vs 1.22 [0.96, 1.81], p < 0.001; eGFR: 71.32 [56.00, 89.08] vs 60.63 [39.32, 78.49], p < 0.001). Number of patients with a creatinine value >1.5 mg/dl was similar in both groups (116 vs 26; 17% vs 25%, p = 0.060). All clinical and laboratory characteristics of the patients who discontinued drug are presented in Supplemental Table 1.

Difference in prognosis after the follow-up with a better survival probability in patients on S/V therapy compared to Drop-out patients) presented a notable increase in adverse events.
Discussion
S/V is one of the most important pillars in the treatment of HFrEF.11,12 It is compounded by two active agents: sacubitril is a neprilysin inhibitor, and valsartan is an ARB. This combination is intended to improve cardiac function in people with HFrEF by acting synergistically. Many randomized controlled trials2,5,13,14 found that this medicine improved HF, reduced hospitalizations, improved cardiac remodeling, reduced CV mortality, and reduced sudden death. However, the long-term effects of S/V on renal function and possible medication interactions are still being disputed. 15 Treatment of WRF in HF patients on S/V may include changing drug doses, optimizing diuretic therapy, and addressing other contributing variables. 16 For these reasons, close monitoring of renal function during treatment is essential. 17 Our analysis first investigated renal function during long-term period demonstrating that the medication had favorable effects regardless of renal function. The beneficial impact persists in both the CKD and preserved renal function groups across long time period. Indeed, the minor decline in renal function observed in our study appears to be less pronounced when compared to the trend accepted by the heart failure association position paper, which indicates a mean eGFR reduction of 10 ml/min/m2 per year. 18
Several processes may play a role in the impairment of renal function with prolonged administration: Direct drug actions can have an effect on renal function. Some people’s renal function will deteriorate as a result. The current situation may be changeable or permanent: S/V can produce a temporary decrease in eGFR, followed by total or partial recovery and stabilization. This is especially critical when starting or modifying the dose of S/V, especially in people with severe CKD. 19 Other drugs may interact with S/V, potentially altering its effectiveness or safety. Potassium-sparing diuretics and mineralocorticoid antagonists, which increase potassium levels in the blood, can interfere with S/V, increasing the risk of hyperkalemia. Because of the drug’s antihypertensive effects, renal function impairment may be exacerbated in patients with low BP values, indicating decreased renal blood flow. 20
Current items are not extensively investigated in large clinical trials and they deserve a more detailed analysis. 21 Notably, the prognosis for HF patients with WRF taking S/V is still questioned and it can vary depending on the primitive kidney disorder, the severity of HF, and hemodynamic impairment. Moreover, the relationship between HF and renal function is complex, and progressive renal function decline is a common complication in patients with more advanced HF.22–24 The association between HF and progressive renal dysfunction may counteract the positive effects of S/V on cardiac deterioration and it is associated with a poorer prognosis. The prognosis is influenced by the degree of renal impairment, the extent of cardiac dysfunction, and the effectiveness of treatment. 25 Finally the drug was not tested and generally employed in patients with advanced CKD and in dialysis settings. 26 Nevertheless, a propensity analysis comparing S/V versus renin-angiotensin inhibitors showed that CKD patients have better outcomes if treated with neprilysin inhibitor. 27 Our findings validated the therapeutic benefits of S/V treatment in HFrEF patients, implying that early treatment can result in a reduction in cardiac overload and an improvement in renal metabolism and hemodynamic profile. More specifically, S/V may improve both systemic hemodynamic status and intraglomerular pressure, leading to a reduction in afferent vasoconstriction and restoration of intrarenal blood flow. The hemodynamic effects drive a neuro-hormonal downregulation and reduced renal sodium avidity. All these features may result in less use of diuretics and less expression of tubulo-glomerular feedback. The safety of S/V was confirmed in the general population as well as in patients with baseline CKD, with S/V use linked with a lower rate of renal function decrease and a better result. The current trend could be the end result of several mechanisms, including improved RAS system modulation, an improvement in congestion status facilitated by neprilysin inhibition, and a direct protective impact of this medication.21,25 A recent study looked at a simple algorithm that could predict individuals who will experience renal function deterioration, although the results were confined to the first 6 months. 26
Declining renal function is a common finding in patients with HF, and it can be due to various factors, including reduced cardiac output, congestion, and the use of medications like diuretics. 27 S/V, like other HF medications, can also affect renal function. In our sample, the WRF was observed in a restricted percentage of patients and it was not associated with increased adverse event risk. Conversely, neprilysin inhibitor seems to keep protective effects on both renal function and congestion status. 28 Looking at patients who did not tolerate the drug, we observed a more advanced NYHA class, reduced BP values more CRT prevalence suggesting a more compromised status compared to subjects continuing treatment. 29 The most important feature of drug discontinuation did not appear mainly related to renal function but to the fall in BP values. Nevertheless, independently of the cause of intolerance, Drop-out patients experienced a worse outcome in terms of hospitalization and renal function deterioration, despite mortality rate remained similar between two groups. Finally, the protective role of S/V appears much more pronounced in patients taking higher doses denoting an increased effect related to a more complete neuro-hormonal and sympathetic overdrive protection. 30
Overall, WRF and CKD in HF patients taking S/V is a challenging issue, they can be associated with similar benefits in terms of reduced hospitalizations and improved survival compared with patients without renal dysfunction, but they can potentially increase kidney-related events.
Limitations
The study’s principal weakness is the observational character of the research; nonetheless, it provides data from a lengthy period of follow-up with extensive laboratory clinical and treatment aspects. We did not compare the S/V group to a placebo group, but our primary goal was to assess changes in renal function in participants using neprilysin inhibitors. Although information from the follow-up period was recorded during follow-up visits (every 3 months during the first year and every 6 months during the resting period), certain occurrences may have been overlooked due to the transitional character of the event. Other biases could be related to the relatively small number of patients with severe CKD and the exclusion of patients with primitive renal disorders. Additionally looking at patient characteristics, subjects with drug drop-out are probably the worse group with more increased deterioration of cardio-renal function. A sub-study investigating these topics should be performed to clarify the potential eGFR cutoff in which S/V has beneficial effects from group in which drug may create severe renal events. Similarly, because of the relatively low number of patients with severe CKD in our sample, our findings cannot be extended to subjects with relevant renal dysfunction and primitive renal disease. We assessed renal function using standard metrics; further investigation should include specific markers of glomerular and tubular injury, as well as albuminuria.31–33
Conclusion
S/V treatment is associated with better outcomes and renal protection in patients with HFrEF across a long-term period. This trend is much more pronounced in people who can tolerate increasing doses. The positive effects of the drug are maintained in both CKD and preserved renal function, despite the subject developing a persistent WRF during follow-up demonstrating increased risk. Current findings highlight the beneficial properties of S/V on renal function Further studies may prospectively investigate the safety and the underlying reasons of current protection.
Supplemental Material
sj-docx-1-tak-10.1177_17539447241285136 – Supplemental material for Effects of sacubitril/valsartan on renal function and outcome in patients with heart failure and reduced ejection fraction: an Italian cohort study
Supplemental material, sj-docx-1-tak-10.1177_17539447241285136 for Effects of sacubitril/valsartan on renal function and outcome in patients with heart failure and reduced ejection fraction: an Italian cohort study by Alberto Palazzuoli, Filippo Pirrotta, Alessandra Cartocci, Elvira Delcuratolo, Frank Loyd Dini, Michele Correale, Giuseppe Dattilo, Daniele Masarone, Laura Scelsi, Stefano Ghio, Carlo Gabriele Tocchetti, Valentina Mercurio, Natale Daniele Brunetti, Savina Nodari, Francesco Barillà, Giuseppe Ambrosio and Erberto Carluccio in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
