Abstract
Acute promyelocytic leukemia (APML) is a myeloid leukemia classified as M3-AML. It is known for its bleeding diathesis, which is predominantly driven by disseminated intravascular coagulation and primary hyperfibrinolysis. However, thrombotic events are relatively less common. A combination of bleeding and thrombotic events is rare and poses a challenge in management. We present a case of acute promyelocytic leukemia complicated by deep venous thrombosis, pulmonary thromboembolism, and hematemesis.
Case
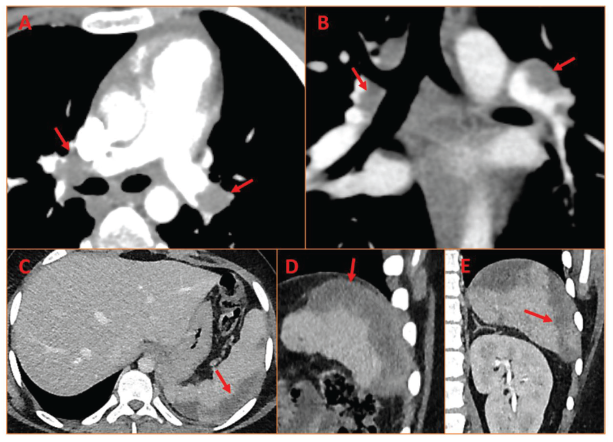
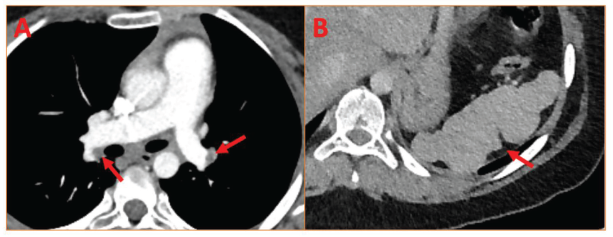
An early teenager presented with progressive right lower limb swelling followed by left lower limb swelling for three months. He was diagnosed with deep venous thrombosis (DVT) involving the right external iliac vein, common iliac vein, and origin of the left common iliac vein. He was started on Rivaroxaban. The blood thrombophilia work-up was negative, including normal prothrombin and activated partial thromboplastin time, normal fibrinogen and homocysteine levels, absence of factors C and S, and antithrombin deficiency, or factor V Leiden mutation. He was readmitted within one month with hematemesis and functional class II dyspnea. His hemoglobin, total leukocyte count, and platelet counts were 11.5 gm dL−1, 8460 mL−1, and 12000 mL−1, respectively. The echocardiogram showed mild RV dilatation with normal right ventricular function and a dilated, non-collapsible inferior caval vein. The pulmonary artery systolic pressure was 45 mmHg. The chest X-ray did not show any significant abnormalities. Given the recent history of DVT and current dyspnea, a CT pulmonary angiogram was planned to rule out pulmonary thromboembolism (PTE). His CT pulmonary angiogram showed bilateral thrombus in both left and right pulmonary arteries with involvement of segmental and sub-segmental pulmonary arteries (Figure 1(a), (b)). Abdominal CT showed hepatosplenomegaly with a hypodense, well-demarcated lesion in the spleen, suggestive of a huge splenic infarct (Figure 1(c)–(e)). His peripheral blood smear revealed 80% myeloblasts and was diagnosed as cytogenetics-proven [positive for t (15,17)(q22,12)] acute promyelocytic leukemia (APML). His international normalized ratio (INR) and d-dimer were 1.7 and 10,000 ng mL−1 (normal: 0–233 ng mL−1), respectively. He was promptly started on all-trans retinoic acid (ATRA) and arsenic trioxide (ATO) without complications and achieved complete molecular remission. After initial bleeding control, he was started on low-molecular-weight heparin, followed by Rivaroxaban maintenance therapy. Repeat CT after six months documented mild residual PTE (arrows in Figure 2(a)) and resolution of splenic infarct (arrows in Figure 2(b)), and there was no echocardiographic evidence of pulmonary hypertension or right ventricular dysfunction. There was a complete resolution of DVT.
Computed Tomography (CT) Pulmonary Angiogram in Axial (A) and Coronal (B) Sections Showing Massive Bilateral Pulmonary Thromboembolism with Filling Defects (Arrows) along with CT Abdomen in Axial (C), Sagittal (D), and Coronal (E) Sections Showing Hypodense Well-demarcated Lesion in the Spleen Suggestive of a Huge Splenic Infarct (Arrows).
(A) Six-month Follow-up CT Pulmonary Angiogram Axial Section Showing Minimal Residual Thrombus in Bilateral Pulmonary Arteries (Arrows); (B) CT Abdomen Axial Section Showing Shrunken Spleen with No Infarct and an Irregular Margin of Previously Resolved Infarct (Arrow).
Bleeding is the most common manifestation of APL, mainly attributed to disseminated intravascular coagulation (DIC), direct proteolysis of proteins, thrombocytopenia, and hyperfibrinolysis. 1 Thrombosis is relatively less caused by the release of prothrombogenic particles, including tissue factors, inflammatory cytokines, and the expression of adhesion molecules on the surface of tumor cells. 1 APL-related cardiovascular thrombotic manifestations range from coronary ischemia to cerebral ischemia and DVT/PTE. 2 This case represents a rare presenting manifestation of AML-M3 with extensive DVT, splenic infarct, and acute PTE with concomitant bleeding manifestations in the form of DIC, posing a great challenge for clinicians.
Informed Consent
Informed consent was obtained from the patient in line with COPE guidance.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
