Abstract
Acute thrombocytopenia in Acute Coronary Syndrome patients may pose a diagnostic challenge due to multifactorial etiologies. It is a recognized, but rare complication of treatment with glycoprotein IIb/IIIa receptor antagonists, which are commonly used in the management of Acute Coronary Syndrome. Diagnosis is made after ruling out other common causes, one of them being Heparin-induced thrombocytopenia. Management includes discontinuation of GPRA & in severe cases might require steroids. We present a case of a 38-year-old gentleman with acute inferior wall MI, who developed glycoprotein IIb/IIIa receptor antagonists-induced acute symptomatic thrombocytopenia.
Keywords
Introduction
Thrombocytopenia in a critically ill patient is one of the most common hematological disorders encountered in the ICU. Causes can be multifactorial, but in patients with ACS, exposure to multiple inciting agents, including oral antiplatelets, Heparin, GP IIb/ IIIa inhibitors offers a challenge in diagnosing the cause and subsequent management. Several differentials may be considered for etiology, including laboratory-related factors like abnormal clumping, pseudo-thrombocytopenia (exposure to lab EDTA or citrate/low temperature), dilutional (after massive transfusion), viral infections, vitamin B12 deficiency, disseminated intravascular coagulation, intra-aortic balloon counter-pulsation (IABP) related & drug-induced thrombocytopenia. We report a case of GPRA-induced refractory thrombocytopenia, which had to be managed using intravenous steroids.
Case Report
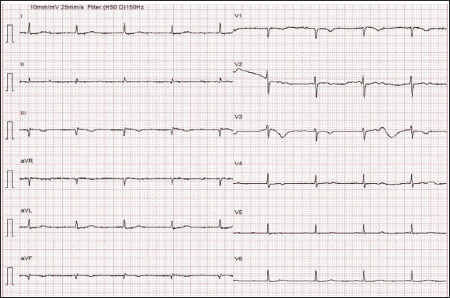
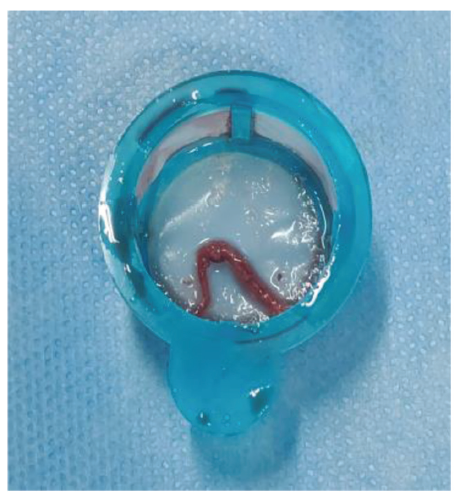
A 38-year-old gentleman, working as an insurance advisor, having risk factors of obesity, type 2 diabetes mellitus, dyslipidemia and a family history of coronary artery disease, had acute inferior wall STEMI with complete heart block. He underwent primary Percutaneous Coronary Intervention (PCI) to the Right Coronary Artery (RCA) at a local hospital and was compliant with post-PCI dual antiplatelets. One week post-PCI, he had recurrent episodes of syncope and was reviewed in an emergency. His vitals were stable (temperature: 97.8°F, blood pressure: 148/96 mm Hg, respiratory rate: 22 per minute, heart rate: 55/minute, and his oxygen saturation 98% on room air). On cardiac examination, he had RVS3; no murmurs or rubs were heard. Electrocardiogram showed q waves in leads II, III and aVF, complete AV dissociation with narrow QRS escape (Figure 1). Troponins were 1.2 ng/ml. Echo was suggestive of mild LV dysfunction (LVEF 47%) & RV dysfunction (TAPSE 14 mm). He underwent a check CAG, which showed subacute stent thrombosis with high thrombus burden. He underwent thrombus aspiration (Figure 2) & PCI to RCA (2 DES-2.25 × 38 for distal RCA & 3.5 × 22 mm for ostio-proximal RCA). In view of complete heart block, he underwent semi-permanent pacemaker implantation and was planned for a reassessment of return to sinus rhythm after revascularization. During the procedure, he had one episode of hemodynamically stable, sustained ventricular tachycardia, which was pharmacologically reverted to sinus rhythm with IV amiodarone.
Electrocardiogram Showing Q Waves in Leads II, III, Complete AV Dissociation with Narrow QRS Escape.
Thrombus Aspirated from Right Coronary Artery.
In view of the significant thrombus burden, he was started on IV heparin infusion & IV tirofiban infusion (0.15 mcg/kg/min). Eight hours post-procedure, he developed oral mucosal bleeding & hematuria. On physical examination, subconjunctival hemorrhage, needle puncture point bleeds, few ecchymoses over both arms and chest were noted. The patient did not complain of any chest pain. Blood investigations showed severe thrombocytopenia with platelet counts declining from 2.23 × 10 5 /µL (pre-procedure) to 0.06 × 10 5 /µL (Figure 3). Blood peripheral smear showed reduced platelets without any abnormal aggregation or clumping. Other blood investigations showed hemoglobin 12.4 g/dL & TLC 5900 /µL, normal PT/INR & aPTT. Doppler screening of lower limb vessels for deep vein thrombosis was negative.
Platelet Count After Starting IV Tirofiban and Response to IV Methylprednisolone.
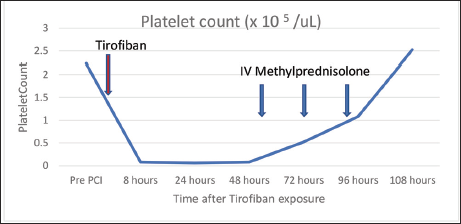
In view of the history of stent thrombosis & severe thrombocytopenia, clinical suspicion of Type II Heparin-induced thrombocytopenia (HIT) was made initially, and HIT antibody assay (against Heparin & Platelet Factor 4 complex) was done, which was negative. Other prothrombotic work-ups were done including ANA, Lupus Anticoagulant, Antiphospholipid, Anti Cardiolipin & β2 glycoprotein antibodies, Protein C & S-all were within normal limits. Heparin, Tirofiban & oral antiplatelets were withheld. However, even after stopping these medications, the patient continued to have persistent thrombocytopenia with minor bleeding manifestations. He was given four-units platelet transfusion, but had no improvement. Subsequently, he was managed with IV pulse methylprednisolone (1 gm once daily for three days), after which platelet counts normalized (Final count 2.53 × 10 5 /µL). He was sequentially re-introduced with oral antiplatelets (Aspirin 75 mg once daily, Ticagrelor 90 mg twice daily). There was no recurrence of thrombocytopenia. An electrophysiological study done after two weeks of PCI showed an AH interval of 192 ms, HV interval of 52 ms, with persistent CHB with a narrow QRS complex. He underwent permanent dual chamber pacemaker implantation four weeks after PCI.
Discussion
Acute thrombocytopenia is a recognized, but rare complication of treatment with glycoprotein IIb/IIIa receptor antagonist (GPRA). The close differential that needs to be excluded is Heparin-induced Thrombocytopenia, which can be either Type I (non-immune mediated, the onset being two to three days after exposure to high dose heparin, and has mild thrombocytopenia with platelet counts rarely below 0.5 × 10 5 , resolves spontaneously, with no sequelae) or Type II (immune-mediated, onset being 5–14 days after exposure with any dose, with severe thromboembolic sequelae, positive for antibodies against Heparin-PF4 complex or Platelet serotonin release assay). 1 One should have a high clinical suspicion as definitive management includes the use of direct thrombin inhibitors or fondaparinux.
Studies have shown several patterns of GPRA-induced thrombocytopenia, including acute pattern (within 12 hours of first exposure), sub-acute pattern (12 hours after a second exposure), the delayed type (which manifests in five to seven days of treatment), platelet clumping type causing pseudo-thrombocytopenia, and a variant causing anaphylaxis. 2 One to three percent of patients on GPRA are noted to have pseudo-thrombocytopenia. 3 Incidence of true thrombocytopenia was seen with Tirofiban in 1.1% in the PRISM trial, 4 1.9% in the PRISM plus trial 5 and 1.1% in the RESTORE trial. 6 However incidence of severe thrombocytopenia (<0.5 × 10 5 /µL) was very rare (0.1%–0.5%). 7 Pathogenesis of platelet destruction, in most cases, can be secondary to the development of drug-dependent circulating IgG against platelet IIb/IIIa receptors. Flow Cytometry is used to detect these antibodies. This entity is more associated with bleeding rather than thrombosis as compared to HIT. Studies have also shown a higher incidence of thrombocytopenia with abciximab (2.4%) compared to tirofiban (0.5%). 8 In the EPIC study, patients with GPRA-induced thrombocytopenia were found to have high risk of mortality and revascularization. 9 Regular monitoring of platelet counts, discontinuation of GPRA and judicious use of platelet transfusion (only in presence of bleeding & platelet counts <0.2 × 10 5 /µL) remains the cornerstone of treatment. 3 Steroids and IVIG are reserved for refractory cases. 3 Thienopyridine antiplatelets are known to cause mild thrombotic thrombocytopenia. Isolated severe thrombocytopenia is rarely seen with aspirin and clopidogrel. 10 Ticagrelor is also rarely implicated. 11 Management includes minimizing the dosage or change of medications.
In the index patient, even though all agents suspected to cause thrombocytopenia were withheld, platelet counts improved only after giving IV steroids. Prior heparin exposure during PCI & subsequent stent thrombosis made heparin-induced thrombocytopenia (Type II) a likely possibility. However, severe thrombocytopenia with bleeding is rarely encountered with HIT, which is more associated with thrombotic complications. 1 Also HIT antibody assay (against Heparin-Platelet Factor 4 complex) was negative, ruling out the possibility. There was no recurrence of thrombocytopenia after re-initiation of aspirin & ticagrelor. All technical causes & pseudo-thrombocytopenia was also ruled out. So the GPRAs (Tirofiban) mediated thrombocytopenia was considered as most likely diagnosis. GPRAs are widely used in the management of patients with acute coronary syndromes. Immune mechanisms responsible for severe thrombocytopenia seen with GPRAs have been hypothesized for all three agents currently available, although specific laboratory tests are not available for use in routine practice. 12 Although the incidence of severe thrombocytopenia is relatively low, the implications for patients are potentially life threatening.
Conclusion
This case presents a diagnostic dilemma in a patient with acute severe symptomatic thrombocytopenia, post PCI. Appropriate diagnosis of etiology is essential as management differs according to the cause. HIT & pseud-thrombocytopenia should be ruled out in such cases. As the patient requires to be on long term antiplatelet therapy after ACS & PCI, he should be followed up diligently. Avoidance of the inciting medication in any further future event is essential.
Abbreviations
ACS - Acute Coronary Syndrome
ANA - Antinuclear antibodies
CHB - Complete Heart block
GPRA - GPIIb/IIIa receptor Antagonist
HIT - Heparin Induced Thrombocytopenia
LV - Left Ventricle
EF - Ejection Fraction
PCI - Percutaneous Coronary Intervention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The authors confirm that the consent for submission and publication of this case report has been obtained from the patient in line with the Committee on Publication Ethics guidance.
