Abstract
Heparin is one of the most widely used drugs in the world. It has been described as a lifesaving drug due to its roles in treating many serious diseases and illnesses including kidney dialysis, surgery, cardiac-invasive, heart attack, cardiac arrhythmia, acute coronary syndrome, pulmonary embolism, stroke, deep vein thrombosis, blood clot prevention, and many other related uses. Heparin drug products currently approved in the United States are obtained from porcine intestinal mucosa sourced from pigs, the majority of which is imported from China. However, due to the heparin contamination crisis (2008) and potential shortage and to safeguard the quality of current and future heparin supply chains including raw material, Food and Drug administration (FDA) posted a notification on its website titled “FDA Encourages Reintroduction of Bovine-Sourced Heparin”. This perspective is intended to address the history of regulatory and scientific background of heparin drug products obtained from bovine and porcine sources and general recommendations for improving the quality of current heparin manufacturing process including Critical Quality Attributes (CQA), control management, process control, related tests, limits, etc. Additionally, a general plan with systematic steps is proposed for diversifying heparin supply chains by reintroduction of bovine sourced heparin to the US market.
Introduction
In June 2015, the US Food and Drug Administration (FDA) held a
The World Health Organization lists heparin as an essential drug product. Its use as an anticoagulant allows for successful lifesaving interventional and surgical procedures, and its therapeutic use to eliminate unwanted blood clotting has saved a countless number of lives. This paper will review the history of heparin drug products with recommendations for its future.
Heparin Chemical Structure
Heparin, a complex pharmaceutical agent, has been used clinically since the early part of the 20th century in the prevention and initial treatment of thrombosis. Heparin is a linear, highly sulfated polysaccharide consisting of alternating glucosamine and uronic acid monosaccharide residues (Figure 1). Heparin does not possess a defined single structure or organized template like proteins or nucleic acids. Rather, it is a complex material composed of random sequences of various saccharide chains with various lengths and various degrees of sulfation with an average molecular weight ranging between 16 and 18 kDa. 1

Illustrates the complexity of heparin's structure as a highly sulfated polysaccharide consisting of alternating glucosamine and uronic acid monosaccharide residues.
Heparin is mostly a polymer of disaccharides with each unit containing an uronic acid and glucosamine. When the various stereoisomers, sugars, and sulfation patterns are combined, there are potentially 32 different disaccharide units to be included in what is labeled “heparin.” This makes heparin one of the most information-dense molecules in biology. Due to the lack of a template, complete sequencing of heparin polysaccharide chains is not feasible compared to nucleic acids and protein.
Although heparin is a complex sulfated glycosaminoglycan and it is comprised of polysaccharide chains of different molecular weights, it has an organized and defined composition of disaccharide units and patterns of sulfation. Each chain is comprised of a disaccharide repeat unit of
Figure 1 illustrates the complexity of heparin's structure showing the major repeating unit which consists of iduronic acid and glucosamine residues (Figure 2). Sequences A-E is the pentasaccharide sequence required for the high-affinity binding of heparin to the antithrombin (AT) molecule.
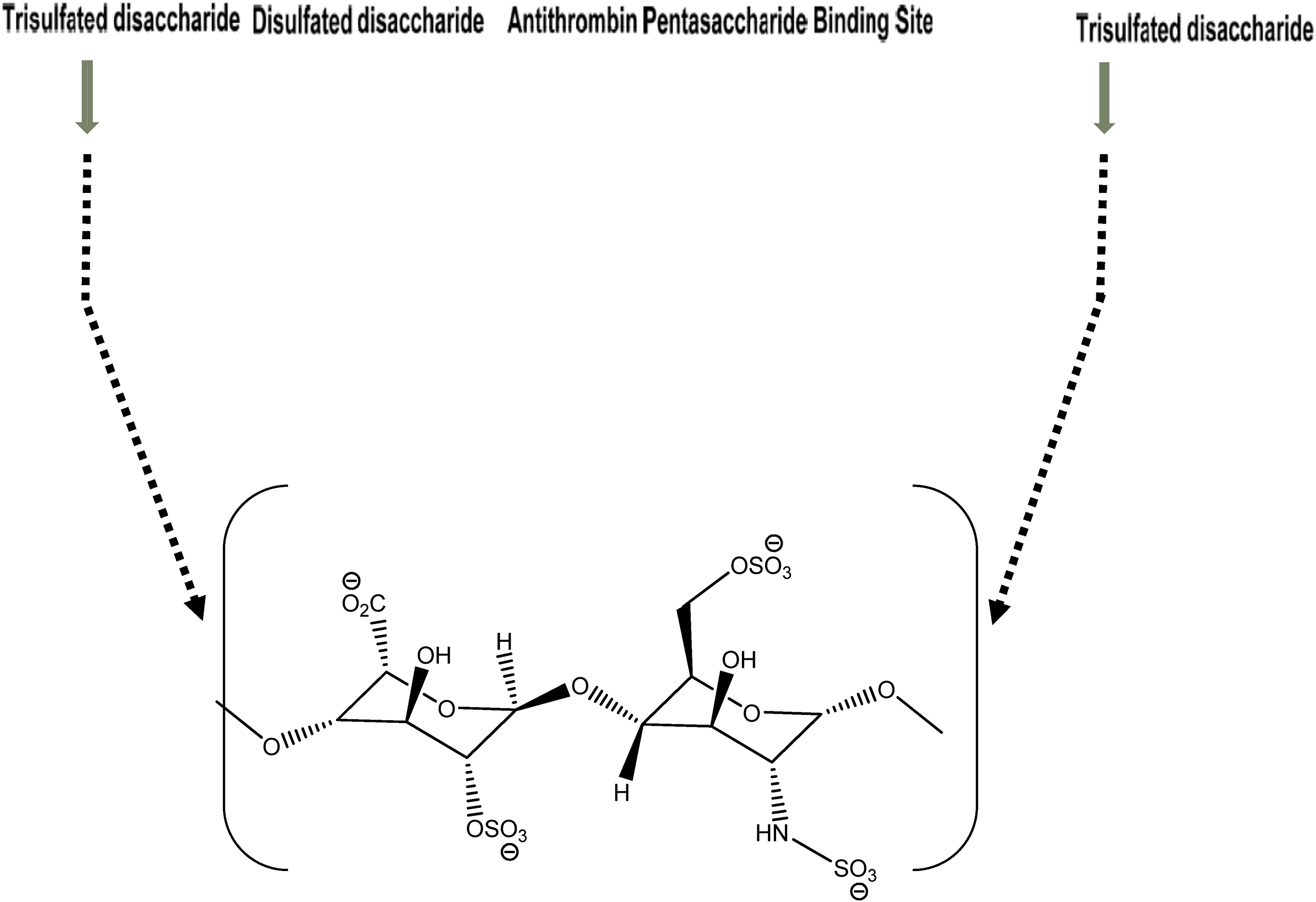
Showing the major repeating units which consist of iduronic acid and glucosamine residues.
From Figure 2, a number of disaccharides that serve as building units of heparin can be identified. Complete enzymatic depolymerization of heparin with three polysaccharide lyase enzymes (eg, heparin lyase I, heparin lyase II, heparin lyase III) produces eight variously saturated disaccharides. These disaccharide units, isolated and structurally characterized, reveal nonsulfated, monosulfated, disulfated, and trisulfated disaccharides.2–4 Therefore, the complexity and polydispersity of heparin are determined with
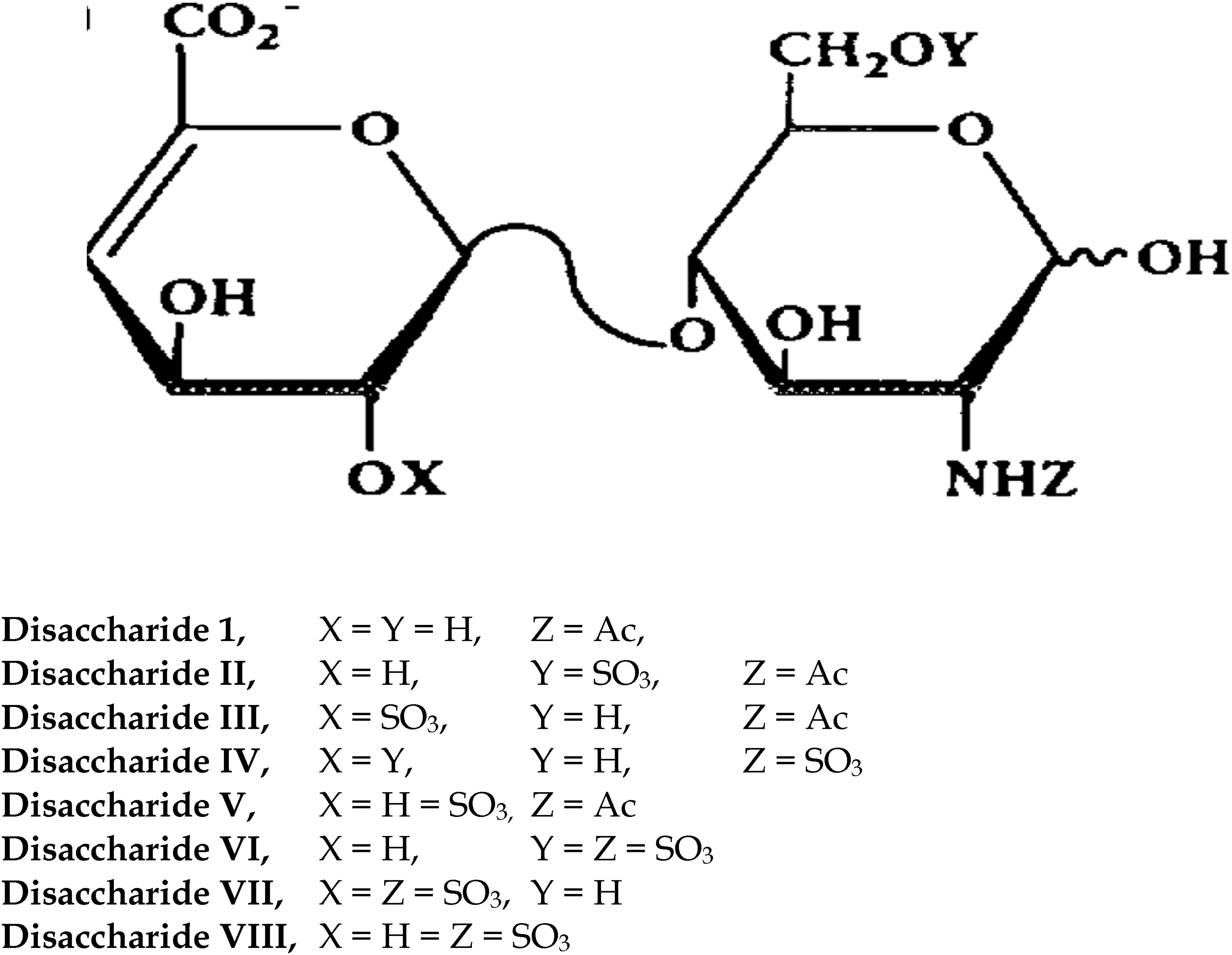
Disaccharide composition of heparin obtained by enzymatic treatment.
Anticoagulant Activity of Heparin
Heparin has been used successfully as an injectable antithrombotic medicine since the 1930s. Heparin is second only to insulin as a natural therapeutic agent used in many indications including, but not limited to, kidney dialysis, surgery, cardiac-invasive procedures, treatment of heart attack, cardiac arrhythmia, acute coronary syndrome, pulmonary embolism, stroke, and deep vein thrombosis, prevention of blood clotting, and anticoagulant coating of devices. Recently, heparin has been used as an anticoagulant agent to treat patients with the SARS-Cov-2 virus that causes COVID-19 to control blood clot formation which clogs blood vessels preventing blood oxygenation. This additional clinical application of heparin may result in increased global demands for heparin drug products. 6
The biological activity of heparin that targets blood clotting results from its binding to the serine protease inhibitor AT which in turn binds to the serine proteases thrombin (coagulation factor IIa) factors Xa, IXa, XIa, and XIIa. Heparin binding causes a conformational change in AT that result in enhanced inhibition of thrombin and other serine proteases involved in the blood clotting cascade, as shown in Figure 4.

Thrombin inactivation by heparin.
The sulfated polysaccharide chains of heparin possess structural and conformational properties which are critical to the function of the biological potency of heparin as an anticoagulant agent. The polysulfated polysaccharide sequences have variable binding affinities to AT. The resulting conformational changes in AT result in variable inhibitory kinetics against thrombin, factor Xa, and other proteases involved in blood clotting.
Heparin Drug Product
Heparin was first discovered by Jay McLean and William Henry Howell in 1916. Clinical trials were conducted in the 1930s to confirm its safety and efficacy as an anticoagulant agent. It is considered as one of the oldest drugs still available to patients.
The original FDA-approved New Drug Application (NDA) for heparin drug products was in 1939. This heparin was sourced from cows (bovine lung). More heparin application approvals followed in the 1940s, but these heparins were sourced from porcine intestinal mucosa. All the above approved heparin applications were sourced from domestic US animals (cows and pigs). Eventually, US heparin market supplies could not meet the increased demand for heparin. Therefore, between 1970 and 1980, several NDAs for porcine heparin drug products were approved using heparin active pharmaceutical ingredient (API) imported from China.
There have been about 60 years of safe and effective use of bovine lung heparin in patients in the United States. However, because of the spread of BSE or “Mad Cow” disease in Europe in the 1990s, concerns appeared about the possible introduction of transmissible spongiform encephalopathy agents into bovine lung heparin. This led to the removal of bovine lung heparin from the US market in the late 1990s with replacement by porcine heparin. Figure 5 depicts this timeline.

Historical timeline of regulatory heparin animal-sourced applications in the United States.
In 2008, some samples of porcine heparin were found to be contaminated with over-sulfated chondroitin sulfate (OSCS) resulting in multiple deaths for patients throughout the world. 7 Since then, FDA has worked diligently and continuously to provide additional analytical assessments of heparin to further safeguard heparin drug products and related heparin supply chains. Center for Drug Evaluation and Research (CDER), a division of FDA, remains interested in alternative sources to porcine heparin to diversify and enable a proactive approach to address possible shortages or contamination of the global porcine heparin supply.
For the 6 years between 2008 and 2014, FDA worked with USP, international heparin academic regulatory agencies, experts, and heparin manufacturers to update the heparin USP monograph to protect the quality and safety of heparin (Figure 6). The work conducted over two stages defined new specifications and incorporated advanced analytical techniques that identify the heparin product, assess the purity, assure no contamination or adulteration of the product, and assess the potency of heparin. The new monograph introduced modern analytical methods and Critical Quality Attributes (CQA) that tightened the allowable levels of impurities including organic solvents, chondroitin sulfate, dermatan sulfate, protein, and nucleic acids. Orthogonal analytical methods and tests included were molecular weight distribution to assure the consistency of heparin products obtained from various suppliers, as well as H1-nuclear magnetic resonance (NMR) and SAX-high-performance liquid chromatography (HPLC) to ensure the identification of heparin product and absence of OSCS. 8

United States Pharmacopeia (USP) heparin sodium monograph updating stages from 2009 to 2014.
Heparin Manufacturing
The manufacturing process of heparin has not changed substantially since its introduction, and the manufacturing process of porcine and bovine heparin are essentially the same. The manufacturing process starts with collecting lung or intestine tissues at animal slaughterhouses where meat is harvested for human consumption. Animals should be healthy and suitable for human consumption certified by the local/federal health authorities.
For good manufacturing practice (GMP), the following steps should be considered before starting the manufacture of heparin:
Controlling the supply chain starts with having dedicated farms that can supply animals (cows or pigs) (preferably closed herds) to slaughterhouses. The animals should be certified to be healthy and suitable for human consumption. Traceability of the animals back to the farm of origin should be documented. Animals should be inspected by health officials.
Slaughterhouses should be dedicated to cows or pigs only. No other animals should be used in the same area so that the mucosa obtained from the animals cannot be mixed or contaminated with ruminants or tissues from animals of different species. These houses should be inspected by local health officials.
The extraction process should be performed by trained personnel using proper equipment, hygiene, clothes, etc.
The collected mucosa should be stored in appropriate containers with the addition of preservatives, usually sodium bisulfite, before shipping/transportation to the manufacturing facility. Addition of a preservative has many functions; stabilizes the mucosa, limits microbial growth, and halts the intrinsic enzymatic digestion which liberates polysaccharide chains from proteoglycan complexes. Storing mucosa for an extended period may also result in the desulfation process due to natural enzymatic/chemical reactions within the mucosa.
Mucosa is usually transported by trucks to the manufacturing facility where it is pumped directly into reaction tanks/vessels where crude heparin is produced (Figure 7).

General manufacturing process for crude heparin (porcine or bovine)*.
Large quantities of mucosa (thousands of kilograms) are typically used to obtain a desirable yield of heparin. It is estimated that each 1 kg of mucosa will produce between 10 and 260 mg of heparin. 9
The two most important steps in the initial GMP preparation of heparin, crude heparin from animal tissue, are the enzymatic digestion (proteolytic enzymes) and NaOH treatment. The material is treated with anion exchange resins.
Crude heparin is unpurified, unfractionated heparin (UFH) material which contains some impurities including peptides/protein, lipids, oligonucleotides, and other non-heparin polysaccharides (eg, heparan sulfate, dermatan sulfate, chondroitin sulfate). In 2013, the FDA published a
Certain methods are used on the crude heparin material released from tissue to detect foreign or contaminating material. Potential methods include polymerase chain reaction (PCR) and immunochemical assays. The absence of porcine species in bovine heparin and vice versa is a further safeguard for the quality, identity, and purity of heparin drug products and to comply with regulatory labeling requirements.
The FDA published
Some of the most common tests performed on crude heparin are (refer to FDA PCR for the identification of heparin animal species (ie porcine or bovine) and absence of any other species. Biological activity to show anti-Xa activity, which is usually less than the purified API heparin due to the presence of impurities in the crude material (eg, nucleotides, proteins/peptides, dermatan sulfate, chondroitin sulfate). NMR to confirm the basic features of heparin as a polysaccharide sulfated complex containing the main major repeating units of
Most crude heparin materials are a complex of heparin polysaccharide mixtures bound to an ion exchange resin, forming a brown colored solid material. The complex is very stable and can be shipped or stored for a long time. However, some manufacturers release heparin from the resin followed by precipitation and consider this material as a crude heparin.
Figure 8 shows the GMP process of converting crude heparin to API. Heparin sodium API obtained from porcine and bovine sources is a white powder. The API should meet the current updated USP specifications and related tests, stored in appropriate containers with appropriate labeling. Heparin sodium drug product (injection) is API formulated into the appropriate solution (water, saline). The drug product solution is sterilized and filled into the appropriate container/closure system labeled with the strength/activity in U/mg.

General manufacturing method for the preparation of heparin sodium (porcine or bovine) active pharmaceutical ingredient (API)*.
Some of the most important steps in the API manufacturing process are the treatment of heparin with NaOH, potassium permagante (KMnO4), and hydrogen peroxide (H2O2). The use of a strong base such as NAOH and a strong oxidizing agent including KMnO4 and H2O2 has been utilized since the discovery of heparin in the 1920s. It is obvious that the scientists and researchers at that time were aware of the risk associated with a drug obtained from an animal source and considered the possibility that some pathogens and viruses may be carried over from the starting animal tissue material to the final drug product. They designed this harsh and methodical treatment during the heparin manufacturing process for the purpose of viral inactivation and removal of bacterial endotoxins.
Quality Risk Management Approach/In-Process Control
Heparin manufacturing process is a complex laborious process that spans days/weeks, involves many steps, and deals with extremely large quantities of mucosa starting material (tens of thousands of pounds) together with large size reaction vessels. The main objective of heparin manufacturing process is to extract the desired sulfated polysaccharide materials from mucosa followed by purification and removal of impurities (eg, lipids, proteins/peptides, nucleic acids, etc.). In addition, removal of related polysaccharides such as dermatan sulfate, heparan sulfate, and chondroitin sulfate is required.
Throughout this process, some heparin batches may fail to meet the USP specifications having a low-quality profile in terms of low potency, loss of sulfation, molecular weight variation, crude batches with different color, chemical or enzymatic modifications, etc. This can be attributed to the sheer size of the manufacturing production process and multiple steps used. Specifics including starting material, equipment, reagents, excipients, manpower, records/documentation, in-process testing, release testing, etc., also play a role.
Therefore, controlling and monitoring the manufacturing process throughout its cycle is needed to ensure the pharmaceutical quality of the final product. A Quality Risk Management Approach (QRMA), a risk-based process of in-process controls that monitor various stages of the manufacturing process, should be implemented. This involves identification, monitoring, and evaluation of CQA, Critical Process Parameters (CPP), and Operating Ranges and Time (ORT) with respect to monitoring and control of the in-process control and testing of the animals, mucosa, raw materials/reagents, crude heparin, and production of the final heparin sodium drug substance (Figure 9).

Quality Risk Management Approach (QRMA); in-process control.
The QRMA describes in detail the various critical stages of the manufacturing process where in-process control must be used to monitor and control CQA, CPP, and OTR to eliminate any deviations or changes from the standard manufacturing process.
Such deviations and changes usually happen to batches manufactured at the same site where one or more of the followings failures can occur:
Batches not meeting potency USP specification of not more than 180 U/mg. The manufacturer may not reject or reprocess the batch, but they may use one of the following to address this failure:
Blending or mixing low-quality batch(es) with high-quality batches; because this is not GMP practice it is a short-term solution. Releasing such below standard product to the market; however, this may result in quality issues and subsequent recalls. To avoid such failures, manufacturers should check the entire manufacturing process and examine its various stages by reviewing records, operation ranges, quality of the reagents/excipients, process steps, etc. Failures and potency loss can be due to many reasons including:
Storage conditions for the mucosa; longer storage may result in degradations from intrinsic enzymes that are present in the mucosa. Varying times and conditions used during the oxidation/bleaching reactions. Water/alcohol fractionation step. Changes in the phases concentration and/or fractionation times may result in loss of sulfates and/or some polysaccharide chains particularly the pentasaccharide anti-thrombin binding site responsible for most of the anticoagulant activities.11,12 Chemical modification of heparin and formation of unnatural sugar units. This is usually due to: KMnO4treatment that produces chemical modification and formation of acetyl group on some heparin saccharide chains.
13
Controlling this oxidation step by optimizing amount and times of this treatment will reduce the formation of acetyl groups. Variation in molecular weight. This can be due to:
Quality of the proteolytic enzyme. Due to the size of the reactors, duration/times of treatment may vary which may result in excessive depolymerization. Loss of sulfation due to improper fractionation/precipitation step. Different colors of the crude heparin batches. This can be due to:
Improper oxidation/bleaching step (mixing times). Quality of the resins, considering that sometimes resins are used multiple times (recycled/regenerated). Conduct a hazard analysis and identify preventive measures for each step of the manufacturing process. Establish the corrective action to be taken when monitoring indicates that the critical control points are not in a state of control. Establish the appropriate critical limits and ranges in the process control. Establish a system to monitor the critical control points. Product Identify and control potential quality issues during development and manufacturing.
Quality risk management is designed to have a quality product. The focus is aimed at the manufacturing process so that it consistently delivers the intended performance of the product. An added feature is that it enhances the knowledge of product performance over a wide range of material attributes and assesses critical attributes of crude materials, API processing options, and process parameters during the life cycle of the product. ICH-Q11 described the critical control points which should be implemented especially for a complex manufacturing process such as that for heparin. These points include:
During the 2008 heparin contamination crisis, FDA evaluated the risk assessment and mode of failure during heparin manufacturing processes. The investigation was based on the manufacturing historical data obtained from heparin manufacturers and suppliers over several years. From these investigations, it was clear that the presence of OSCS was the culprit of the contamination of heparin drug products. Evaluation and assessment of the critical parameters, attributes, and control points of the various stages of the manufacturing process provided the key to the stages or steps where things had gone wrong resulting in poor, low-quality products including external contamination. Figure 10 illustrates that the most probable cause of a low-quality heparin product is external contamination with non-heparin materials.

Probable causes of failures during heparin manufacturing process including external contamination and/or adulteration.
Alternative Source Material for Heparin
It has been reported that there will be a global increase in demand for heparin and the possibility exists that supplies will not meet demands in the future.14,15 It was estimated that the heparin worldwide market size is expected to reach about $10 billion by 2027 from $990 million in 2017. This estimation is based on the global heparin market size (eg, value, capacity, production, and consumption) in key regions such as North America, Europe, and Asia-Pacific (China, Japan).
In the United States, currently, all heparin marketed approved products including UFH and low-molecular-weight heparin (LMWH) are obtained from porcine intestinal mucosae and most of the API is imported from China.
Depending on a single source is a risk for such a lifesaving drug as heparin, especially if the supply cannot meet the demand. In fact, the Chinese pig population has faced many serious problems in the last decade, including but not limited to:
In 2007, blue ear pig disease (highly pathogenic strain) swept through 10 provinces in China destroying large number of the pig population. In 2008, adulteration of heparin with the semi-synthetic, nonnatural molecule OSCS by Chinese manufacturers resulted in death and injury for patients worldwide, including the US. Between 2018 and 2020, African swine fever (ASF) swept through the pig population in China.
Therefore, in order to make heparin drug products available for the US patients and safeguarding heparin supplies, an alternative source for heparin should be sought.
16
Accordingly, FDA encourages all prospective manufacturers and suppliers of heparin, particularly bovine heparin but also other animal-sourced products such as sheep and goat, to develop a good manufacturing process capability and related quality controls for their products. FDA also welcomes engagement with sponsors considering the use of non-porcine animal-sourced products through the Pre-Investigational New Drug (Pre-IND) process. FDA will consider meeting requests, as appropriate, to discuss prospective applications with respect to demonstrating safety, efficacy, quality, and purity. 17
Over the past 90 years, heparin has come to be well understood in terms of structure and anticoagulant activity. Manufacturing processes have been well established including detailed quality control measures. With this robust management, heparin drug products can be produced today with the assured outcome of a pure, unadulterated, biologically active material. Because of the potential shortage of porcine starting material, it is incumbent upon the heparin community to determine and establish alternate sources of starting material. The first logical approach is to revisit bovine heparin. Other animal tissue, however, will likely provide further options for useful alternatives.
Bovine Heparin
Although bovine heparin is not currently available in the US, it is still manufactured and commercially available in other countries, including South America (Brazil, Argentina), with a long history of safety. As we indicated previously, bovine heparin was withdrawn from the US market due to the spread of BSE in Europe in the 1990s. However, the US bovine heparin drug product was not withdrawn due to quality or safety reasons; it was withdrawn as purely a precautionary measure.
Heparin obtained from bovine and porcine sources are comparable in their polydispersity as polysaccharide heterogeneous sulfated mixtures that consist of major repeating units of iduronic acid and glucosamine residues. The only difference found between porcine and bovine heparin is in the potency where bovine heparin is about two-thirds the potency of porcine heparin. This potency difference may be due to the variation in distribution of the sulfate groups on the polysaccharide chains. 18 To circumvent this issue, dosing is clinically adjusted to meet porcine heparin potency.
Early studies comparing porcine and bovine heparin products, including LMWH as reported in 1990 19 identified by gradient polyacrylamide gel electrophoresis (PAGE) analysis and related activities that there is a slight difference in the degree of sulfation and the molecular weight distribution between porcine and bovine unfractionated materials. Controlled partial enzymatic digestion with heparinase 1 enzyme indicated that there is a difference in the composition of polysaccharide building block units between porcine and bovine heparin materials. Activity analysis showed that porcine heparin has higher anti-Xa activity compared to bovine heparin (172 vs 155 U/mg, respectively).
Publications in subsequent years comparing the molecular profile and chemical characteristics between unfractionated porcine and bovine heparins20–23 using more advanced analytical techniques and instrumentations such as H1 and C13 NMR, HPLC-ultraviolet (HPLC-UV), and LCmass spectroscopy (LC-MS), confirmed that there are subtle differences between bovine and porcine heparin. Comparing the disaccharide building units of bovine and porcine heparin the total amount of sulfation in bovine or porcine heparin is similar; however, there is a slight difference in the distribution of sulfates and N-acetyl groups as shown in Figure 11. These differences in the composition of the disaccharide building units between porcine and bovine are expected for the following reasons:
Different animals were sourced to produce heparin API. API heparin batches, porcine or bovine, manufactured at the same site and under the same strategy controlled conditions, will be found to exhibit:
Slight variation in the degree of sulfation; Variation in molecular weight distribution; Different potency/activity range.

Proton nuclear magnetic resonance (NMR) spectroscopy of porcine mucosa heparin compared to bovine mucosa heparin. Signals 1, 2, and 3 corresponding to H5 + H6 of 6-
The above variations are depicted in the structural features and molecular profile obtained from 600 MHz proton spectra of heparin obtained from porcine intestinal mucosa and bovine intestinal mucosa. The spectra of the two animal sources show considerable comparability between the two heparin samples as reported by Mauri et al. 25 (Figure 11).
These differences do not seem to produce a significant risk to patients due to the length of historical use of bovine heparin in the United States (60 years) and to the continuing use of bovine heparin in Brazil for the last 40 years.
With respect to activity, recent studies demonstrated that bovine heparin has less anti-Xa potency (130 U/mg) compared to porcine heparin (185 U/mg). However, if bovine heparin potency concentration is adjusted to porcine potency (eg, when vialed), identical concentration-response curves in the activated partial thromboplastin time (aPTT) and anti-protease assays are obtained. In addition, protamine neutralization studies indicate that bovine heparin requires a higher amount of protamine sulfate, which can be accomplished when using the aPTT or anti-Xa assay for adjustment. Therefore, the anticoagulant activity of porcine and bovine heparin, when used at adjusted anti-Xa unit levels, is comparable. 26
Baig et al. 27 reported that the use of bovine heparin in patients under dialysis in place of porcine heparin, and that the lower anti-Xa activity of the bovine heparin did not significantly affect the patients or the operation of the dialyzer. This indicates that the use of bovine heparin in dialysis, regardless of its lower potency compared to porcine, does not impact the efficacy and safety of the patients. This also confirms the possibility that porcine heparin can be substituted for bovine heparin for dialysis. The availability of bovine heparin becomes critical in the event of a porcine heparin shortage considering the large and increasing number of dialysis patients in the United States who undergo dialysis several times a week.
Demands for heparin have been increasing worldwide due to its various lifesaving indications. Yet, there is little growth in the United States and the European Union (EU) for alternate heparin sources and the FDA is concerned that without additional domestic or foreign supply, a serious shortage could result. Demand has increased dramatically worldwide in the last decades and the prices have been going up alarmingly. Therefore, the reintroduction of bovine heparin, particularly bovine lung sourced heparin, presents many advantages including:
Diversifying animal sources. Increase heparin production to combat any potential shortages. Alternative sources will reduce dependency on the imported porcine material that has the potential shortcomings of adulteration, contamination, viruses, and diseases. A new market will be open for US heparin manufacturers that can use domestic cows for heparin production. Bovine lung is a clean organ compared to intestines.
To introduce bovine heparin to the US market, FDA will promote dialogues and meetings to discuss and review these applications beginning with Pre-IND process. However, and as reported before, one of the main challenges for introducing bovine heparin product is the concern over a potential risk of the presence of BSE in bovine sourced heparin.
The BSE agent caused a previously unrecognized variant of Creutzfeldt–Jakob disease (vCJD) in humans, most likely due to consumption of contaminated beef products. There has never been a documented or suggestive transmission of Transmissible Spongiform encephalopathy (TSE) to humans from a medical product of bovine origin. This is also true in the UK, where a very large number of cattle were infected with the BSE agent in the 1980s and 1990s28,29
World Health Organization has classified US and South America as having negligible BSE risk while Canada is under controlled BSE risk. Therefore, FDA encourages dug applications coming from such countries using bovine sourced heparin. The supply chain should be safeguarded and controlled adequately to obtain a safe bovine product. This includes obtaining raw heparin from well-controlled and monitored animals (cows) fit and healthy for human consumption. In addition, dedicated slaughterhouses for cows should be certified by local, state, and federal health authorities.
As stated previously, bovine heparin was available on the US market for about 60 years. It was withdrawn, for precautionary reasons, from the US market in 1999 due to the outbreak of the BSE disease in Europe. It has a long history of safety and efficacy. Bovine heparin drug product has been available in Brazil and it is listed in the Brazilian Pharmacopeia. Bovine heparin is also available in Argentina. Recently, bovine heparin is being manufactured in China and India. Currently, the United States does not have a monograph for bovine heparin due to the absence of this material in the US market. However, the current US monograph for porcine sourced heparin can be used with some adjustment for tests including potency for bovine heparin. It is anticipated that FDA will work with USP to establish a monograph for bovine heparin when the bovine drug product will be approved and becomes commercially available on the US market.
Two major concerns are associated with the introduction of bovine sourced heparin. First is the issue of BSE (Mad Cow disease). However, a recent study demonstrated that the heparin manufacturing process that includes the use of NaOH and oxidation reaction with KMnO4e will inactivate BSE. Such treatment will improve the safety profile of bovine heparin.
29
This work demonstrated that the heparin manufacturing process itself is capable of BSE clearance in bovine heparin. The following are the critical steps of the inactivation procedure:
Crude heparin treatment with NaOH (pH 12, 20 h, 50°C). Filtration with diatomaceous earth. Bleaching and oxidation steps using H2O2 and KMnO4 (pH 6.0, 20 h, 50°C).
The second concern is immunogenicity. FDA has published a
FDA would work with all prospective manufacturers and suppliers of bovine heparin products to develop a good manufacturing process capability with related quality controls for their products.
The Future of Heparin
FDA continues to work unabated toward safeguarding heparin by controlling the source of materials and the manufacturing processes. The QRMA and related in-process controls should be used to manufacture this animal-derived product to ensure that manufacturers implement and comply with cGMP for monitoring quality and production processes.
The quality and traceability of raw materials should also comply with the FDA appropriate requirements including controls, and best remanufacturing practices allowing for the early detection of any problems and ensure the availability of safe and good quality of the final heparin drug products.
FDA is acting proactively to eliminate the possibility of a heparin shortage in the future. Without a foreign supply, there could be a serious shortage of heparin in the US. FDA recommends that sponsors interested in meeting with FDA prepare a comprehensive Pre-IND/IND meeting package with the appropriate regulatory and scientific data to support the current stage of drug development.
The meeting package may include, but is not limited to, the following information:
Brief description of the Chemistry, Manufacturing, and Controls Information. The regulations in 21 CFR 312.23(a)(7)(i) emphasize the graded nature of chemistry, manufacturing, and controls (CMC) information needed as development under an IND application progresses. Description of the candidate product, including physical, chemical, and/or biological characteristics, as well as its source. Description of the dosage form and information related to the dosage form. Quantitative composition of the drug product. A brief description of adequate test methods used to ensure the identity, strength, quality, purity, and potency accompanied by the test results, or a certificate of analysis, of the product lots used in toxicological studies and intended for the proposed human study. The grade and quality (eg, USP, NF, ACS) of excipients used in the manufacture of the investigational candidate product. Name and address of the manufacturer(s) (if different from the sponsor). The method of preparation of the proposed heparin product lots used in preclinical studies and intended for the proposed human study, including a brief description of the method of manufacture and the packaging procedure, as appropriate, with a description of the container and closure system. Appropriate stability data to support the requested/proposed shelf life/expiry dating of the drug substance and the drug product. Guidance for Industry. Heparin for Drug and Medical Device Use: Monitoring Crude Heparin for Quality, June 2013. Guidance for Industry and Food and Drug Administration Staff. Heparin-Containing Medical Devices and Combination Products: Recommendations for Labeling and Safety Testing, July 2018. Guidance for Industry. Immunogenicity-Related Considerations for Low-Molecular Weight Heparin, February 2016. FDA, proactively, in collaboration with the USP and stakeholders, has initiated compendial improvements for safeguarding drug supply chains for heparin (from porcine sources) with emphasis on CQA like sensitive detection methods for potency, contaminants, and impurities. See The US Regulatory and Pharmacopeia Response to the Global Heparin Contamination Crisis.
8
Diversifying Global Heparin Supply Chain; Reintroduction of Bovine Heparin in the United States.
16
Listed below are the related initiatives performed by FDA:
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
