Abstract
Brugada phenocopies (BrP) are clinical entities that are etiologically distinct from true congenital Brugada syndrome. BrP are characterized by type 1 or 2 Brugada electrocardiogram (ECG) patterns in precordial leads V1-V3. However, BrP is elicited by various underlying clinical conditions such as myocardial ischemia, pulmonary embolism, electrolyte abnormalities, or poor ECG filters. Upon resolution of the inciting underlying pathological condition, the BrP ECG subsequently normalizes.
Takotsubo (octopus fishing pot) cardiomyopathy (TCM) also known as stress cardiomyopathy is an acute cardiac condition characterized by transient systolic dysfunction of the left ventricular apex and mid-ventricle with depressed LV function mimicking acute coronary syndrome (ACS) and recovers within a few weeks. TCM is most commonly seen in postmenopausal women with intense physical and or emotional stress. We are reporting a rare case of BrP in a patient with TCM masquerading as ACS posing a diagnostic and therapeutic challenge.
Abbreviations
ABS: apical ballooning syndrome
BrP: Brugada phenocopies
BrS: Brugada syndrome
SCD: sudden cardiac death
TCM: takotsubo cardiomyopathy
Introduction
Brugada syndrome (BrS) is an autosomal-dominant inherited arrhythmic disorder characterized by ST elevation with successive negative T wave in the right precordial leads without structural cardiac abnormalities. 1 Patients are at risk for sudden cardiac death (SCD) due to ventricular fibrillation (VF) and are responsible for up to 20% of sudden death in patients with structurally normal hearts. 2
Brugada phenocopies (BrP) are clinical entities that have electrocardiograph (ECG) patterns that are identical to true congenital BrS but are elicited by various other factors, such as myocardial ischemia, metabolic abnormalities, mechanical mediastinal compression, and poor ECG filters. 3 BrP is an emerging entity with incompletely understood pathophysiological mechanisms.
Takotsubo (octopus fishing pot) cardiomyopathy (TCM) was first described in Japan in 1990 by Sato et al is also known as stress cardiomyopathy or broken heart syndrome or apical ballooning syndrome (ABS). 4 It is an acute cardiac condition characterized by transient systolic dysfunction of the left ventricular (LV) apex and mid-ventricle with depressed LV function mimicking acute coronary syndrome (ACS) and recovers within a few weeks. 4
Here we report a rare case of BrP precipitated possibly by TCM, who was successfully treated conservatively.
Case Details
A 60-year-old-female with no comorbidities presenting with a history of retrosternal chest discomfort, diaphoresis followed by a sudden onset loss of consciousness for 1 hour. She was brought to EMD unconscious and struggling to breathe. The patient had a history of vague chest discomfort and intermittent breathlessness for 15 days, which coincided with the death of her brother.
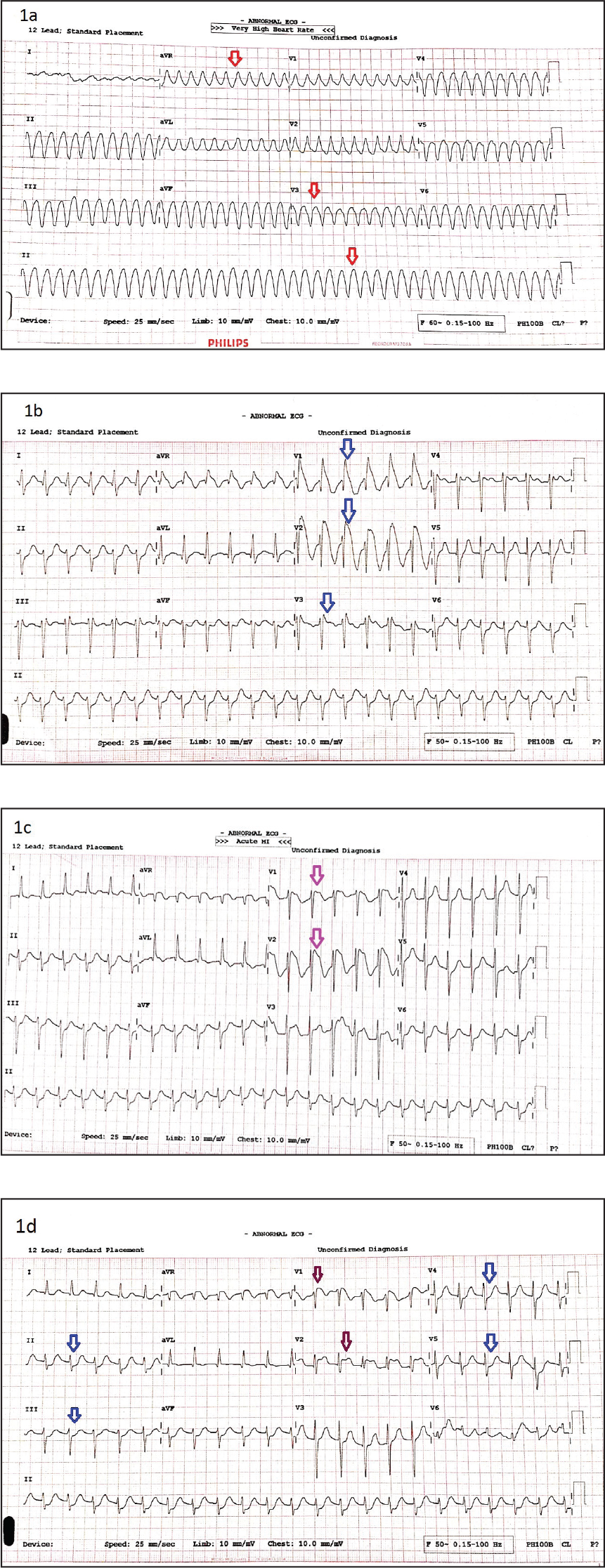
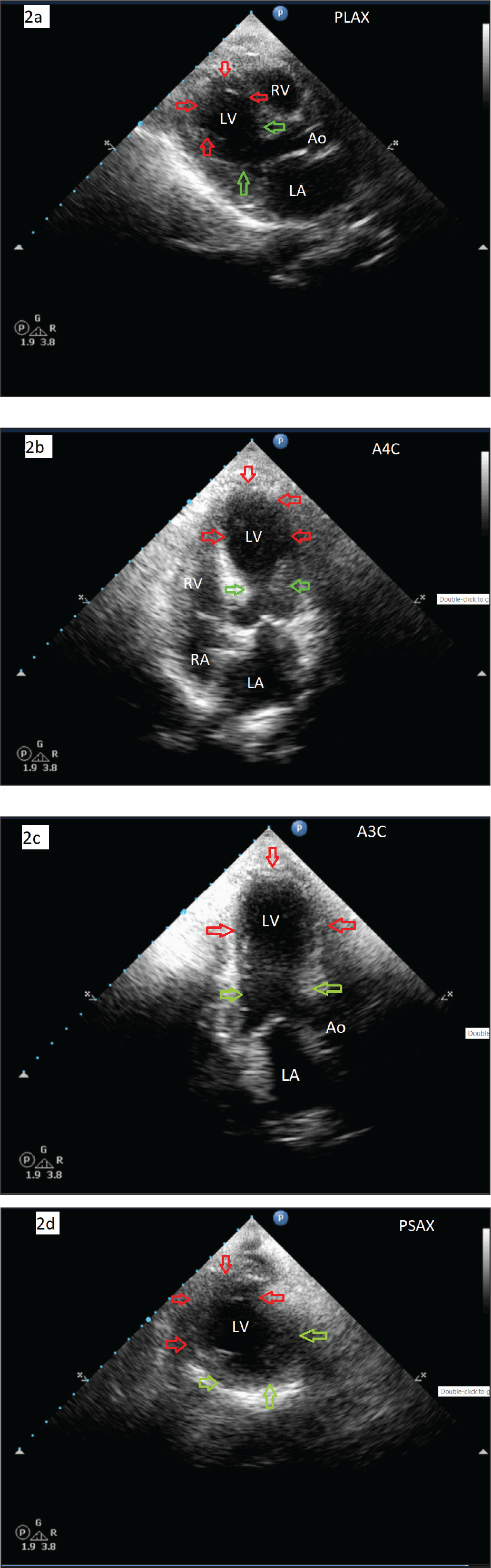
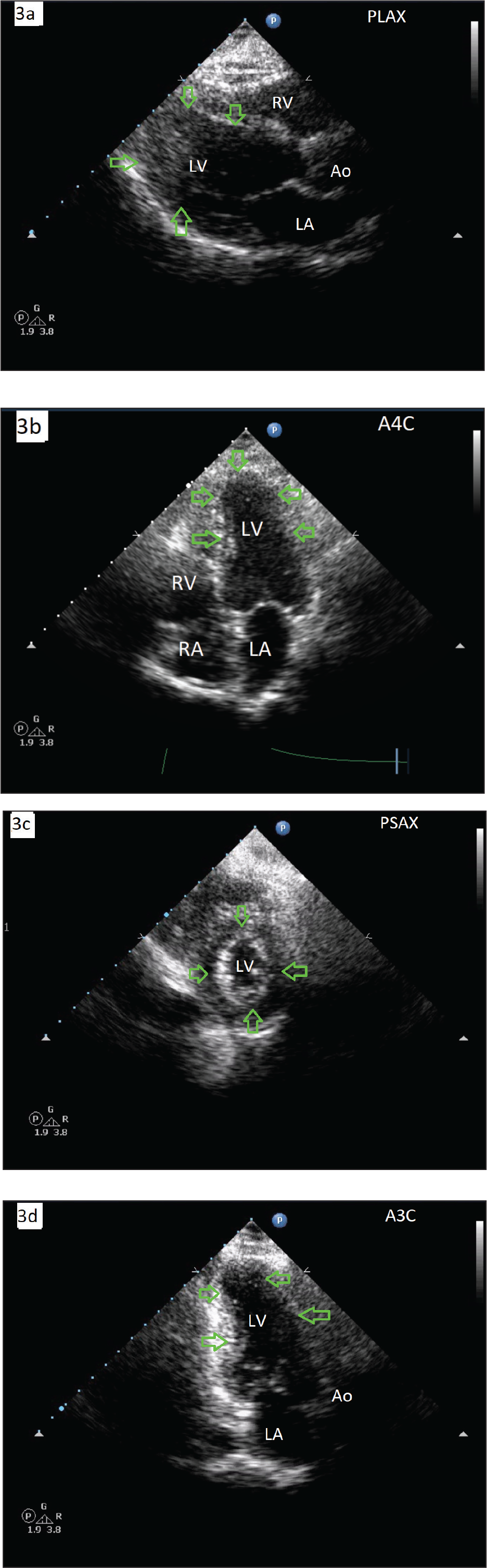
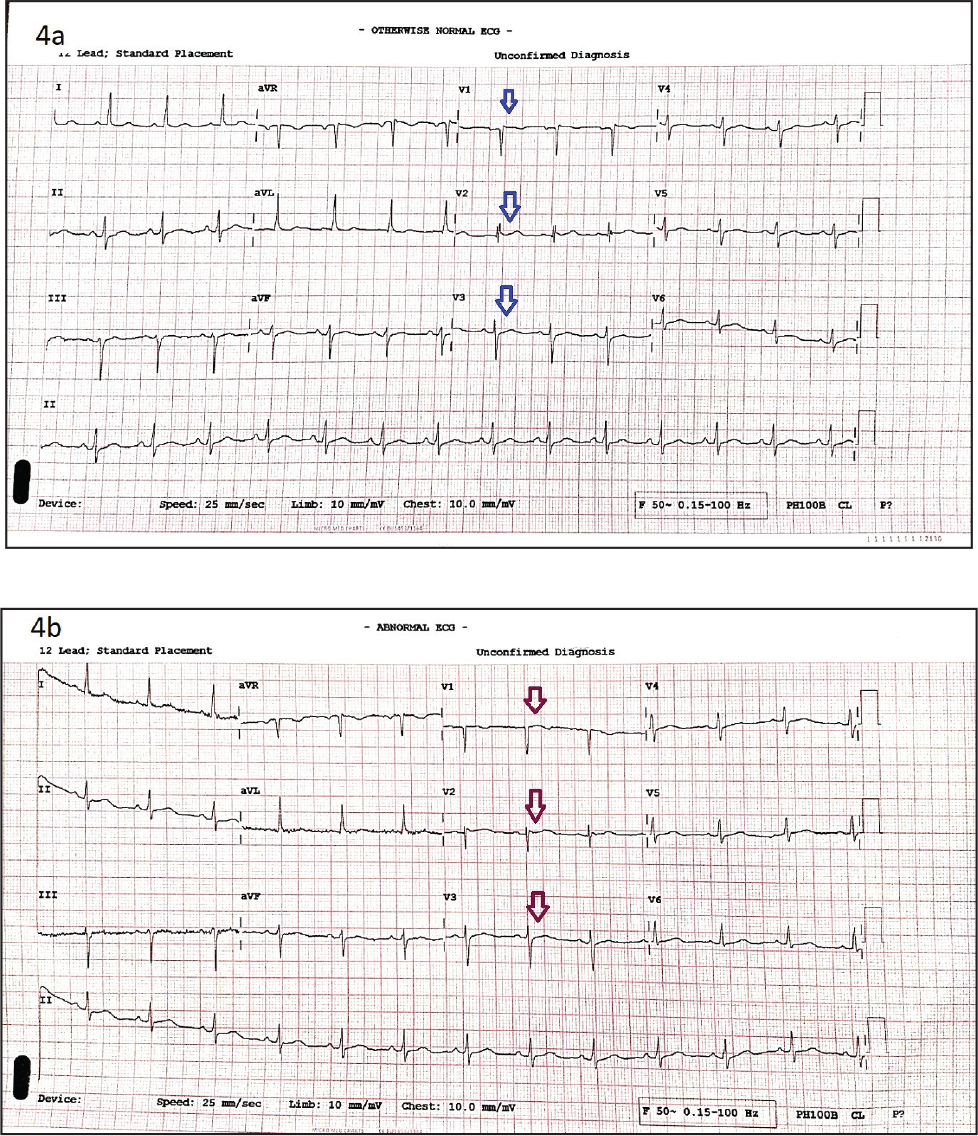
The patient had pulse-less electrical activity, and ECG showed (Figure 1a) ventricular tachycardia (VT). She was cardioverted using 200 joules direct current (DC) shock, intubated, and put on mechanical ventilation. Postcardioversion ECG in sinus rhythm showed sinus tachycardia with incomplete right bundle branch block (RBBB) and ST elevation in V1 to V3 with subtle ST depression in I, II, and V4-V6. AVR had J point elevation with ST coving. ECG findings were consistent with Type I BrS, shown in Figure 1b. Cardiac biomarkers at presentation were elevated (Troponin T-0.275, CKMB-13.24 both in ng/mL). Arterial blood gas analysis at presentation showed metabolic acidosis (pH:7.27, pCO2:45, pO2:122 in mmHg, HCO3:20, BE-6.1 in mmol/L). Other investigations like complete blood count, renal, liver, thyroid function tests, serum electrolytes, calcium, magnesium, and parathormone levels were within normal limits. Computed tomography scan of the brain plain was unremarkable. Two-dimensional echocardiogram (2D ECHO) showed regional wall motion abnormality (RWMA) in anterior segments, mid and distal septum, and apical-lateral segments with features of apical ballooning and reduced LV function (EF of 35%) as shown in Figures 2a to 2d. and preserved RV function. Due to a lack of prior history of similar episodes, previously recorded ECG, or family history of SCD, we decided to rule out ACS by doing a coronary angiogram (CAG), which revealed normal epicardial coronary arteries. Six hours later her ECG changes started resolving with persistent type 1 Brugada pattern in anterior chest leads (Figures 1c and 1d). Post CAG, ACS protocol was withdrawn, with continued small dose aspirin and atorvastatin combination. The patient regained consciousness and was extubated on the second day. She was discharged on day 3 with guideline-directed medical therapy for heart failure, oral Quinidine, and instructions to avoid certain medications (a handout of drugs to be avoided in BrS was given) and precipitating factors. A provocative test to elicit BrS was planned during follow-up once the patient was stable and recovered from TCM. The patient had complete recovery of LV function at 6 weeks follow-up (Figures 3a–3d). The provocative test was done in the sixth week of follow-up with oral Flecainide following the prescribed protocol. 5 Serial ECGs did not show any significant changes from baseline to meet the criteria 6 for BrS and 2 of the ECGs are shown in Figures 4a and 4b.
(1a) Presentation ECG showing monomorphic ventricular tachycardia (red arrows), (1b) postcardioversion using 200 joules DC shock reverting to sinus rhythm with sinus tachycardia, incomplete RBBB, ST elevation/coving of ≥2mm in V1 to V3 (2 of 3) consistent with Brugada type I pattern, (1c) postcardioversion ECG at 6 hours showing resolving ST segments yet persisting Brugada pattern (maroon arrows), and (1d) postcardioversion ECG at 12 hours, persisting Brugada pattern although the amplitude of ST coving reduced (maroon arrows). There are ST depressions in V4 to V6, II, III, and AVF (blue arrows). DC: direct current; ECG: electrocardiogram; RBBB: right bundle branch block.
Discussion
BrS is an inherited autosomal dominant disorder with mutations in the SCN5A gene and prevalence of 0.5 to 1 per 1000 in Asian/South East Asian countries. 1 Proven treatment to prevent SCD due to VT or VF is the implantation of an automatic implantable cardiac defibrillator (ICD) 7 and radiofrequency catheter ablation in a small high-risk population. 8
2D ECHO on the day of admission showing features of TCM with RWMA, hypokinetic distal septum, apex, and apical-lateral segments. Apical ballooning (red arrows) and hypercontractile basal segments (green arrows) and reduced LV function are also seen. (2a) PLAX view, (2b) A4C view, (2c) A3C view, and (2d) PSAX view. 2D ECHO: 2-dimensional echocardiograms; TCM: takotsubo cardiomyopathy; RWMA: regional wall motion abnormality; LV: left ventricle; PLAX: parasternal long axis; A4C: apical 4 chamber; A3C: apical 3 chamber; PSAX: parasternal short axis.
2D ECHO postrecovery after 6 weeks from initial illness. Note the disappearance of RWMA, apical ballooning, and normalized LV function (green arrows). (3a) PLAX view, (3b) A4C view, (3c) PSAX view, and (3d) A3C view. 2D ECHO: 2-dimensional echocardiogram; RWMA: regional wall motion abnormality; TCM: takotsubo cardiomyopathy; LV: left ventricle; PLAX: parasternal long axis; A4C: apical 4 chamber; A3C: apical 3 chamber; PSAX: parasternal short axis.
BrP, on the other hand, has a reversible underlying condition such as adrenal insufficiency, hypokalemia, mechanical compression, pulmonary embolism, myocardial ischemia, pericardial diseases, and ECG modulation that elicits or induces the Brugada ECG pattern.9, 10 Once this underlying condition resolves there is prompt normalization of the ECG. This is contrary to true congenital BrS where the ECG manifestations are unmasked by sodium channel blockers, vagotonic agents, febrile states, and various metabolic conditions. Patients with BrP have a low clinical pretest probability of true congenital BrS as opposed to a high clinical pretest probability in patients with true congenital BrS who have a documented personal history of cardiac arrest, nonvagal syncope or a family history of SCD. 11 Patients with BrP have a negative provocative challenge with a sodium channel blocker, while those with true congenital BrS have a positive provocative challenge.
ECGs pre- and post-provocative testing using oral flecainide after 6 weeks of initial illness. (4a) Preprovocative test and (4b) postprovocative tests after 4 hours showing no significant changes meeting the criteria for the diagnosis of BrS. ECG: electrocardiogram; BrS: Brugada syndrome.
Stress cardiomyopathy or TCM commonly affects postmenopausal women with intense physical or emotional stress and has a favorable prognosis. 5 The condition is diagnosed based on modified Mayo Clinic criteria which include transient RWMA, ECG changes or positive cardiac biomarkers, no angiographic evidence of CAD or spasm, and absence of myocarditis or pheochromocytoma. 12 Potential ECG findings of TCM include ST elevation, ST depression, new-onset left bundle branch block (LBBB), or prolonged QT interval. 5
In this case, the first dilemma was initially with a possible diagnosis of ACS (due to symptoms of chest discomfort, VT on presentation, RWMA on ECHO with reduced LV function, increased cardiac biomarkers) and TCM (given the typical ECHO findings, age, onset, raised biomarkers), which was cleared by CAG. The second dilemma was between BrS (due to typical type I Br ECG findings, VT on presentation) vs BrP (due to absence of a prior history of syncope, resuscitated SCA, significant family history of SCD). This diagnostic dilemma was eventually cleared by a negative provocative test confirming the diagnosis of BrP triggered possibly by TCM, which is not reported in the literature. As TCM is associated with catecholamine excess, the possibility of BrS was low as BrS is potentiated by vagotonic conditions. Postema et al however have reported a case of true congenital BrS in the context of hyperkalemia and acidosis. 13 At this point, we are not clear whether the same conditions that trigger BrS also trigger BrP or there is a completely different pathophysiological mechanism for BrP. This rare case serves to broaden our understanding of this relatively new concept, which rather appears to occur frequently and triggered by many clinical events and TCM could be one of them.
Learning points:
BrP is relatively common and is still an evolving entity and needs to be further explored. The TCM as seen in this case could be a trigger for BrP. Differentiating BrS and BrP is crucial in avoiding expensive treatment strategies like ICD.
Conclusion
BrP are etiologically distinct clinical entities from true congenital BrS, which are triggered by various underlying clinical conditions. TCM could be one of those triggers in a susceptible individual that leads to a diagnostic and therapeutic dilemma. Appropriate diagnosis changes the management of these patients as the clinical implications of BrP are unknown. At this point, treatment remains focusing on the resolution of underlying clinical conditions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
