Abstract
Brugada phenocopies (BrP) are clinical commodities that are etiologically distinguishable from genuine congenital Brugada syndrome. BrP is marked by type-1 or type-2 Brugada electrocardiogram (ECG) patterns in precordial V1 to V3, which is provoked by various underlying clinical disorders. Hemodialysis may improve the BrP ECG image. This case report describes the occurrence of a transient Brugada pattern that was seen on the ECG of an asymptomatic 64-years-old woman after administering hemodialysis due to renal failure. Diagnosis of Brugada phenocopy and BrS is necessary to achieve appropriate further treatment. Hemodialysis may improve the BrP ECG image.
Introduction
Brugada syndrome (BrS) is the primary electrical heart disease diagnosed based on clinical circumstances and typical electrocardiographic features. 1 Brugada phenocopy (BrP) is a clinical aggregation differentiated from authentic congenital BrS fundamentally from its etiology. BrP represents type-1 or type-2 Brugada electrocardiogram (ECG) patterns in precordial V1 to V3. Nevertheless, BrP is stimulated by eclectic underlying clinical circumstances, such as myocardial ischemia, pulmonary embolism, electrolyte anomalies, or flawed ECG filters. 2 The type-1 pattern has a high take-off ST-segment elevation of ≥2 mm followed by a down-sloping concave or rectilinear ST-segment with a negative symmetric T-wave. The Brugada type-2 pattern has a high take-off (r′) ≥2 mm and a convex ST elevation ≥0.5 mm, with a variable T wave at V1 and a positive or flat T wave at V2. 2 The most serious complications of BrS are sudden cardiac arrest and loss of consciousness.3-5 Many other diseases and conditions can lead to a Brugada-like ECG pattern, including atypical right bundle branch block, hypothermia, myocardial infarction/ischemia, Prinzmetal angina, acute pericarditis, arrhythmogenic right ventricular cardiomyopathy, electrolyte disturbances, acute stroke, central and autonomic nervous system abnormalities, duchenne muscular dystrophy, and so on, but the vast majority of patients with BrS possess a structurally normal heart, which is consistent with the notion that this is a primary electrical heart disease. 6 The terminology used to describe the Brugada type-1 ECG pattern induced in patients without BrS is diverse and variable. BrP has a different etymology than true congenital BrS. ECG pattern abnormalities are expected to disappear within a few days of discontinuation of the trigger factors causing BrP. This case report describes the occurrence of BrP features on the ECG that improved after hemodialysis due to renal failure.
Case Report
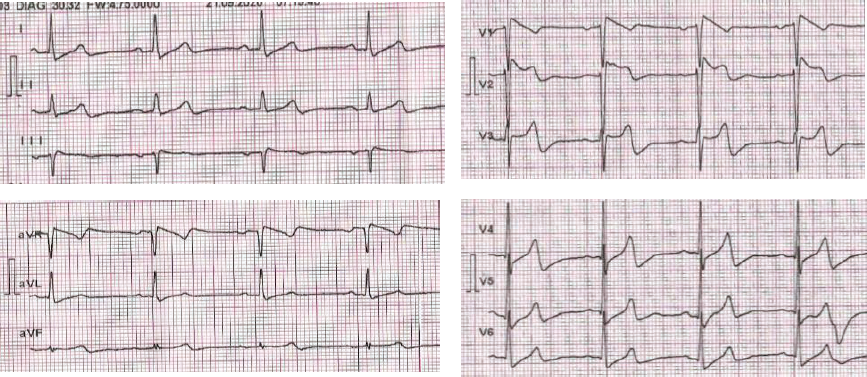
A 64-year-old woman came to the hospital for routine hemodialysis procedures. The patient did not have any complaints when he came for hemodialysis. The patient had a long history of hypertension of about 15 years. The patient has a history of shortness of breath and palpitations when doing activities such as going upstairs and downstairs. The patient denied having a history of stroke, cardiovascular disease, and arrythmia. Patients routinely consume daily total of 10 mg amlodipine, 16 mg candesartan, 2 mg folic acid, 1,500 mg sodium bicarbonate, and 1,500 mg CaCO3. There is no family history of sudden cardiac arrest in the patient’s family from the previous 2 generations. Examination of vital and physical signs indicated that the patients found were glasgow coma scale (GCS) 15, blood pressure 180/90 mmHg, respiratory rate 18 ×/min, pulse rate 56 ×/min, regular, temperature 36.7 Celsius degree, weight 60 kg, conjunctiva anemic, and the border of the left heart rests on 2 fingers lateral to the left mid-clavicle line. Significant investigations showed hemoglobin 10.2 g/dL and eGFR 4.6 mL/min/1.73 m2. The patient’s electrocardiogram examination showed sinus bradycardia with left ventricular hypertrophy and Brugada pattern type 1 (Figure 1). The patient was diagnosed with congestive heart failure grade II, chronic kidney disease on hemodialysis, and Brugada phenocopy class-B. The potassium level of patient before hemodialysis was 6.5 mEq/L.
Prehemodialysis ECG.
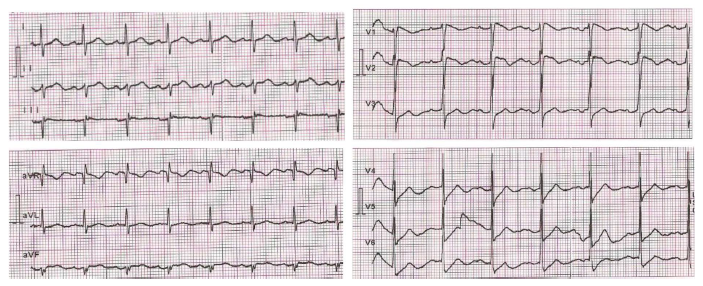
The patient was still on hemodialysis for 4 h with an ultrafiltration goal of 2,000 mL, quick blood 200 mL/min, and quick dialysis 500 mL for 4 h. Examination of vital and physical signs indicated that the patients found were GCS 15, blood pressure 140/86 mmHg, respiratory rate 18 ×/min, pulse rate 74 ×/min, regular, and temperature 36.6 Celsius degree. We performed another electrocardiographic evaluation after the patient had finished hemodialysis by the same physician. Electrocardiography results show an improvement in the appearance of Brugada pattern in the patient. On posthemodialysis ECG, ST-elevation in lead V1 decreased by 2 mm (0.2 mV). Decreased T-wave amplitude was found on the posthemodialysis ECG (Figure 2) by 1 mm (0.1 mV). Reexamination of the potassium level was carried out after hemodialysis, with a value of 5.7 mEq/L.
Posthemodialysis ECG.
Discussion
BrP are clinical commodities etiologically distinguishable from genuine congenital BrS. BrP is marked by Brugada ECG patterns in precordial leads, particularly V1 to V3, which is provoked by several disorders, such as myocardial ischemia, pulmonary embolism, electrolyte abnormalities, or flawed ECG filters. Upon resolution of the rousing underlying pathological disorder, the BrP ECG afterward regularizes. 2
Dendramis 6 studied 55 pieces of literature, which states that BrP has an underlying reversible disease, a low clinical pretest possibility of actual genetic BrS as objected to a high clinical pretest possibility in patients with genuine BrS, and an adverse provocative challenge with a sodium channel blocker (Table 1).
Criteria to Differentiate Brugada Phenocopy and True Congenital Brugada Syndrome. 6
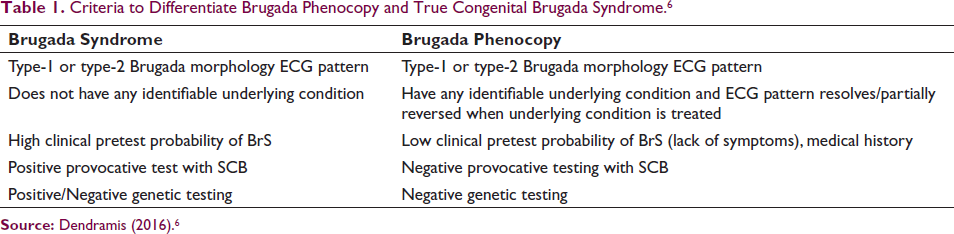
Source: Dendramis (2016). 6
There are 3 additional qualifications in each group to determine if each case of BrP meets the diagnostic criteria: class A includes cases of BrP that meet all mandatory diagnostic criteria including negative provocative drug challenge. Class B-included cases of BrP were highly suspected, but not all diagnostic criteria were met includes cases in which a provocative drug challenge is pending or is not possible to perform due to various factors. Class C includes highly suspected cases of BrP in which provocative testing is not justified or clinically indicated. Our case is included in the B class Brugada Phenomenon case as the provocative sodium channel blocker (SCB) test was not carried out. Our facilities are limited in the provision of medicines, and patients disagree with provocation testing.1, 7
Even though the Brugada pattern, which indicates a hereditary malformation, was detected in sudden unexplained death syndrome (SUDS) and near-SUDS episode (N-SUDS) patients, other triggering component may stimulate the SUDS. Those factors include genders, psychological stress, redundant work, and potassium depletion. Further characteristics, such as zinc and magnesium inequality, are documented in several cardiovascular disorders. The combination of electrolyte disorders and Brugada signs might provoke myocardial predisposition in SUDS. Magnesium, the second most sufficient intracellular cation, is a cofactor of several enzymes implicated in the biosynthesis and metabolism of nucleic acids and proteins, which are vital in order to maintain the accumulation of intracellular potassium. Magnesium administration through intravenous can increase the amounts of muscular magnesium and potassium and greatly reduce the recurrence of premature ventricular contractions. These premature rhythms have been related with the part of magnesium. Magnesium insufficiency is linked with cardiac-induced arrhythmias, when the magnesium intercepts potassium channels and inhibits the cellular efflux, and performs as a cofactor for the Na+-K+ ATPase enzyme, which then catalyzes the cellular influx of potassium in trade to performs sodium efflux. Devouring magnesium will conduct to exaggerated cellular efflux of potassium, defective potassium influx, and drained intracellular potassium content, directing to depolarization of the cardiomyocytes. Magnesium deficiency and Na+, K+ -ATPase pump disbanding may be incriminated as capable risk to cardiac arrhythmias. 8
A unique thing happened in this case compared to other cases, which revealed that transient Brugada-type electrocardiographic abnormalities generally returned spontaneously after dialysis in cases of chronic renal failure. The emergence of BrP before hemodialysis with spontaneous resolution after dialysis treatment may be due to corrections of electrolytes such as hyperkalemia (either due to chronic kidney failure or the use of certain drugs) after hemodialysis. 9
In conclusion, diagnosis of BrP and BrS is necessary to achieve appropriate further treatment. Chronic kidney disease can be a factor in the occurrence of BrP. Therefore, hemodialysis may improve the BrP ECG image. At this point, this suggest that the best treatment for BrP is to ameliorate the underlying condition.
Footnotes
Acknowledgements
We would like to thank Depati Hamzah Regional Public Hospital for their permission to carry out this research. We also thank all nurses, doctors, and research assistants who have worked together so that this research can be completed properly.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
