Abstract
Objectives:
The study was carried out to assess the effect of zinc supplementation on changes in calcium homeostasis, and parathyroid gland, bone, and skeletal muscle histology in rats exposed to subchronic oral glyphosate-based herbicide (GBH, GOBARA®) toxicity.
Methods:
Sixty male Wistar rats in 6 equal groups (DW, Z, G1, G2, ZG1, ZG2) were used: DW and Z were given 2 mL/kg distilled water and 50 mg/kg of zinc chloride (2%), respectively; G1 and G2 received 187.5 mg/kg and 375 mg/kg of glyphosate (in GBH), respectively; ZG1 and ZG2 were pretreated with 50 mg/kg of zinc chloride before receiving glyphosate, 1 hour later, at 187.5 and 375 mg/kg, respectively. Treatments were by gavage once daily for 16 weeks. Serum calcium, vitamin D, and parathormone were estimated. Histopathological examination of parathyroid gland, femoral bone and biceps femoris muscle was done.
Results:
GBH exposure caused significant (P = .0038) decrease in serum calcium concentration in G1, significant (P = .0337) decrease in serum vitamin D concentration in G1, significant increases in parathormone in G1 (P = .0168) and G2 (P = .0079) compared to DW. Significant (P > .05) changes did not occur in the other parameters of G2 compared to DW. Dose-dependent effect in GBH exposure was not observed after comparing G1 and G2. Necrotic changes occurred in parathyroid gland cells, osteocytes, and muscle cells in G1 and G2. In ZG1 and ZG2, significant (P > .05) variations in the parameters were not observed and tissue lesions were absent.
Conclusion:
Subchronic GBH exposure impaired calcium homeostasis observed as hypocalcemia, hypovitaminemia D, and secondary hyperparathyroidism and caused tissue damage in parathyroid gland, bone, and muscle of rats and these were mitigated by zinc chloride pretreatment.
Keywords
Introduction
Glyphosate-based herbicide (GBH) is frequently used globally in agriculture and it acts through the inhibition of the synthesis of proteins in susceptible weed plants to prevent their survival.1,2 The widespread use of GBHs has increased interests in the evaluation of the associated toxicological risks among human and animal populations. 2 Traces of glyphosate have been found in foodstuffs and products, water for consumption, and biofluids like serum, urine, and breast milk of exposed individuals.3,4 Exposure to GBHs may lead to endocrine disruption and it may affect hormonal factors that regulate calcium homeostasis with alterations in the activation of ion channels.5,6
Calcium homeostasis is tightly regulated by parathormone, calcitriol (1,25-dihydroxyvitamin D), calcitonin, and fibroblast growth factor 23 through physiological actions in the parathyroid gland, bone, kidney, and intestine to maintain calcium balance in blood circulation. 7 Our previous studies in rats reported decreased serum calcium concentration after subchronic GBH exposure 8 and increased serum calcium concentration after chronic GBH exposure. 9 There are evidence to suggest that GBH exposure influenced intracellular calcium distribution by reducing calcium level in proliferating skin keratocytes to prevent apoptotic events, 10 increasing calcium levels in renal epithelium to induce apoptosis, 11 and causing loading of calcium in cardiac myocyte leading to necrosis. 12 GBH activates calcium channels of exposed cells to induce calcium influx 13 and it is linked to the promotion of oxidative stress that characterizes GBH toxicity. 6 This effect may be responsible for various tissue damages in organs of GBH exposed individual. Effect of GBH on skeletal muscle via calcium signaling may be responsible for reduced muscle strength and biochemical indications of muscle lesion.14 -16 No report is available on the possible effect of GBH exposure on parathyroid gland and parathormone secretion, despite report of evidence of endocrine disruptions in the hypothalamic-pituitary-gonadal axis. 17 When bone calcium is mobilized by parathormone secretion, bone mineral content may be affected. The urinary glyphosate concentration in human population exposed to GBH was negatively associated with total bone mineral density, 18 implying that increasing systemic GBH exposure could deplete bone calcium and cause bone resorption.
The disruption of calcium signaling in tissues of GBH exposed individual is associated with oxidative stress 6 which is the major mechanism of GBH toxicogenesis and zinc supplementation during exposure provides antioxidant effect to counteract the GBH toxic mechanism.19,20 We have reported previously on the mitigating role of zinc supplementation on the toxicity of GBH (Bushfire® and GOBARA®) in rats.21,22 Therefore the objective of the study was to evaluate the toxic effect of subchronic oral exposure to glyphosate-based herbicide (GOBARA®) on calcium homeostasis, parathyroid gland, bone and skeletal muscle in rats and to determine the presence of mitigating effect of zinc supplementation in the toxicity.
Materials and Methods
The animals, chemicals and experimental design of this study were previously reported in the study on the effect of zinc supplementation on immunotoxicity induced by subchronic oral exposure to GBH (GOBARA®) in rats 22 and are presented hereafter as both studies were concurrent.
Animals
Ethical approval was obtained from the ethics committee of the Institutional Animal Care and Use, University of Jos (reference number: F17-00379; approval date: 3 September 2019). Sixty adult male Wistar rats weighing 140 to 150 g were purchased from the National Veterinary Research Institute Vom and allowed to acclimatize for 2 weeks prior to commencement of the research. The rats were fed standard rat chow (Livestock Feed, Jos, Nigeria), and water was provided ad libitum.
Chemicals
GBH (GOBARA®, Saro Agrosciences, Ibadan, Nigeria) containing 360 g/L glyphosate isopropylamine salt in 100% surfactant and anhydrous zinc chloride (LOBA Chemie Pvt Ltd, Mumbai, India) were used for the research.
Experimental design
The rats were randomly selected and assigned to 6 groups (n = 10/group) as follows: group DW (control), each rat was orally administered 2 mL/kg of distilled water daily; group Z, each rat was orally administered zinc chloride (2%) at 50 mg/kg 9 ; group G1, each rat was orally administered 187.5 mg/kg glyphosate in GBH) (5% of the LD50) 23 ; group G2, each rat was orally administered 375 mg/kg glyphosate in GBH (10% of the LD50) 23 ; group ZG1, each rat was orally pretreated with 50 mg/kg zinc chloride 1 hour before the oral administration of 187.5 mg/kg GBH; and group ZG2, each rat was orally pretreated 50 mg/kg zinc chloride 1 hour before the oral administration of 375 mg/kg GBH.
The treatments were administered by gavage once daily for 16 weeks. 22 The rats were weighed weekly using an electronic weighing balance (OHAUS, Shanghai, China) to monitor the weight changes and ensure appropriate dosing.
Estimation of serum calcium, total vitamin D, and parathormone concentrations
Blood samples (3 mL) were collected by jugular venipuncture under ketamine anesthesia from each rat into clean test tubes without anti-coagulant. The blood samples were kept at room temperature for 30 minutes and then centrifuged at 80g for 10 minutes to obtain the sera.
Serum calcium was estimated using commercial kit (Agappe Diagnostics, Switzerland GmbH) on semi-automatic chemistry analyzer (Erba Chem 5V, Transasia Bio-Medicals Ltd, Mumbai; India). Total vitamin D was assayed using the electro-chemiluminescence binding assay method on automated immunochemistry analyzer (COBAS© E411, Roche Diagnostic, Germany). Serum parathormone concentration was determined using the enzyme-linked immunosorbent assay (ELISA) kits (Bioassay Technology Laboratory, Shanghai China).
Pathological examination
Necropsy was conducted after euthanasia by cervical exsanguination under deep ketamine anesthesia to grossly examine the carcass. 24 Tissue samples from the parathyroid gland, femoral bone, and biceps femoris were collected and fixed in 10% neutral buffered formalin. The bone samples were decalcified using 5% nitric acid before the routine processing. The tissues were processed, embedded in paraffin wax, and cut into sections that were stained with hematoxylin and eosin and examined using a light microscope. 25
Data analysis
Data were summarized as the mean ± standard error of the mean, and variations in means were analyzed by 1-way analysis of variance followed by Tukey’s post-hoc test using GraphPad Prism version 5.0 for Windows (GraphPad, San Diego, CA, USA). Significant variations were accepted at P < .05.
Results
Effect of treatment on serum calcium, vitamin D, and parathormone concentrations
Serum calcium (Figure 1): There was a significant decrease (P = .0038) in serum calcium concentration in G1 compared to DW. The mean value of G1 and G2 were comparable, but the G2 value was not significantly different from DW. Significant increases of the value in ZG1 (P = .0067) and ZG2 (P = .0008) compared to the G1 indicated reversal of depressing effect of GBH on the value.
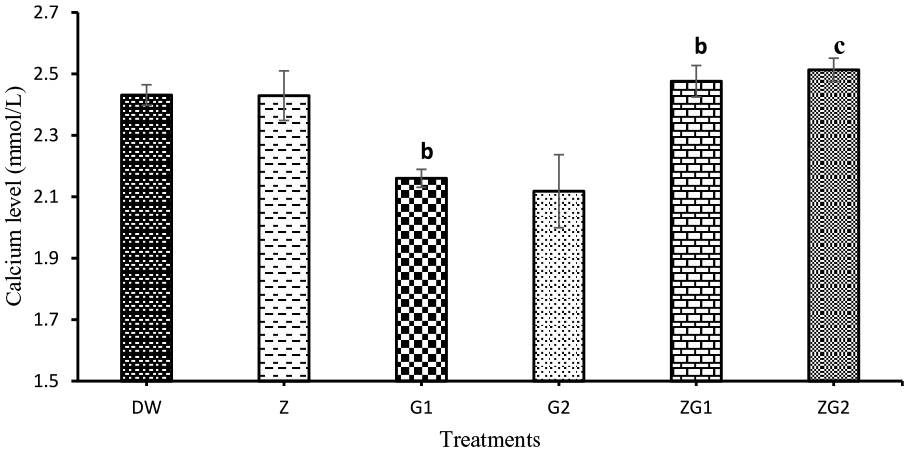
Serum calcium concentrations in Wistar rats treated with 2 mL/kg distilled water (DW), 50 mg/kg zinc (Z), 187.5 mg/kg glyphosate (G1), 375 mg/kg glyphosate (G2), 50 mg/kg zinc + 187.5 mg/kg glyphosate (ZG1), and 50 mg/kg zinc + 375 mg/kg glyphosate (ZG2) for 16 weeks. b, P = .0038 in G1 compared to DW and b, P = .0067 in ZG1, c, P = .0008 in ZG2 compared to G1.
Serum vitamin D (Figure 2): There was a significant decrease (P = .0337) in concentration in G1 group compared to the DW group. Conversely, there was a significant increase in concentration in ZG1 (P = .0474) compared to the G1. Differences were not observed in concentrations between G1 and G2. Also, there were significant increases in ZG1 (P = .0226) and ZG2 (.0468) compared to the G2.

Serum Vitamin D concentrations in Wistar rats treated with 2 mL/kg distilled water (DW), 50 mg/kg zinc (Z), 187.5 mg/kg glyphosate (G1), 375 mg/kg glyphosate (G2), 50 mg/kg zinc + 187.5 mg/kg glyphosate (ZG1) and 50 mg/kg zinc + 375 mg/kg glyphosate (ZG2) for 16 weeks. a, P = .0337 in G1 compared to DW, a, P = .0474 in ZG1 compared to G1, a, P = .0226 in ZG1 and 0.0468 in ZG2 respectively compared to G2.
Serum parathormone (Figure 3): The level significantly increased in the G1 (P = .0168) and G2 (P = .0079) groups compared to the DW. The levels in G1 and G2 did not differ from each other, and were not significantly different compared to ZG1 and ZG2. The other groups (Z, ZG1, and ZG2) did not have significant variations from one another and when compared to DW, G1, and G2.

Serum parathormone concentrations in Wistar rats treated with 2 mL/kg distilled water (DW), 50 mg/kg zinc (Z), 187.5 mg/kg glyphosate (G1), 375 mg/kg glyphosate (G2), 50 mg/kg zinc + 187.5 mg/kg glyphosate (ZG1), and 50 mg/kg zinc + 375 mg/kg glyphosate (ZG2) for 16 weeks. a, P = .0168 in G1 and b, P = .0079 in G2 compared to DW.
Effect of treatment on histopathology of parathyroid gland, femoral bone, and biceps femoris
Parathyroid gland (Figure 4): There was no observable lesion in the parathyroid glands of the control rats in DW (Figure 4a) and in Z (Figure 4b). Necrosis of the parathyroid gland was observed in G1 (Figure 4c). The parathyroid glands of rats in G2 also showed necrosis with vascular congestions (Figure 4d), but in ZG1 and ZG2 no observable lesion (Figures 4e and f) was identified.

(a) Photomicrograph of parathyroid gland of rat administered distilled water (DW) at 2 mL/kg for 16 weeks showing no observable lesion (H&E). (b) Photomicrograph of parathyroid gland of rat administered zinc (Z) at 50 mg/kg for 16 weeks showing no observable lesion (H&E). (c) Photomicrograph of parathyroid gland of rat administered glyphosate at 187.5 (G1) showing necrosis of the parathyroid gland cells (N) (H&E). (d) Photomicrograph of parathyroid gland of rat administered glyphosate at 375 (G1) showing necrosis of the parathyroid gland cells (N) (H&E). (e) Photomicrograph of parathyroid gland of a rat administered zinc at 50 mg/kg and glyphosate at 187.5 mg/kg (ZG1) for 16 weeks showing no observable lesion (H&E). (f) Photomicrograph of parathyroid gland of a rat administered zinc at 50 mg/kg and glyphosate at 375 mg/kg (ZG2) for 16 weeks showing no observable lesion (H&E).
Bone (Figure 5): The femoral bones of rats in DW and Z showed no observable lesion as shown in Figures 5a and b respectively. Necrosis of the femoral bone was observed in the bones of rats in G1 (Figure 5c) and G2 (Figure 5d). The femoral bone of rats in groups ZG1 and group ZG2 showed no observable microscopic lesion similar to those of DW group as shown in Figures 5e and f respectively.

(a) Photomicrograph of femur of rat administered distilled water (DW) at 2 mL/kg for 16 weeks showing no observable lesion (H&E). (b) Photomicrograph of femur of rat administered zinc (Z) at 50 mg/kg for 16 weeks showing no observable lesion (H&E). (c) Photomicrograph of femur of rat administered glyphosate at 187.5 mg/kg (G1) showing necrosis of the osteocytes and osteoid (N) (H&E). (d) Photomicrograph of femur of rat administered glyphosate at 375 mg/kg (G2) showing necrosis of osteocytes (N) (H&E). (e) Photomicrograph of femur of rat administered zinc at 50 mg/kg and glyphosate at 187.5 mg/kg (ZG1) for 16 weeks showing no observable lesion (H&E). (f) Photomicrograph of femur of rat administered zinc at 50 mg/kg and glyphosate at 375 mg/kg (ZG2) for 16 weeks showing no observable lesion (H&E).
Skeletal muscle (Figure 6): The biceps femoris (BF) muscle of the rats in groups DW and Z showed no observable lesion in Figures 6a and b respectively. The BF muscle of rats in G1 showed myofibre necrosis in Figure 6c. Myofibre necrosis with fragmentation of the myofibres and hemorrhages were observed in the BF muscle of rats in G2 (Figure 6d). The BF muscles of rats in ZG1 and ZG2 showed no observable lesion as shown in Figures 6e and f respectively.

(a) Photomicrograph of skeletal muscle (biceps femoris) of rat administered distilled water (DW) at 2 mL/kg for 16 weeks showing no observable lesion (H&E). (b) Photomicrograph of skeletal muscle of rat administered zinc (Z) at 50 mg/kg for 16 weeks showing no observable lesion (H&E). (c) Photomicrogaph of skeletal muscle of rat administered glyphosate at 187.5 mg/kg (G1) for 16 weeks showing necrosis of the myofibres (N) (H&E). (d) Photomicrogaph of skeletal muscle of rat administered glyphosate at 375 mg/kg (G2) for 16 weeks showing necrosis of the myofibres (N) with fragmentation of the muscle fibers (H&E). (e) Photomicrogaph of skeletal muscle of rat administered zinc at 50 mg/kg and glyphosate at 187.5 mg/kg (ZG1) for 16 weeks showing no observable microscopic lesion (H&E). (f) Photomicrogaph of skeletal muscle of rat administered zinc at 50 mg/kg and glyphosate at 375 mg/kg (ZG2) for 16 weeks showing no observable microscopic lesion (H&E).
Discussion
Investigation of the role of GBH exposure in disturbance of calcium homeostasis requires experimental assessment of the physiological components controlling plasma calcium concentrations to prevent hypocalcemia and hypercalcemia. In this study, we demonstrated that subchronic oral GBH exposure decreased in serum calcium and vitamin D concentrations, and increased serum parathormone concentration without apparent dose effect. These events were also accompanied by necrotic changes in the tissues of parathyroid gland, bone and skeletal muscle, which could suggest the occurrence of abnormal calcium influx in these calcium-sensing tissues. GBH-induced pathological conditions were mitigated by zinc chloride pretreatment, indicating significant role for zinc supplementation under GBH toxicological risk. Therefore, this study provides evidence of the need to pursue the toxicogenic dimensions of calcium dyshomeostasis in GBH toxicity as earlier proposed. 6
The decrease in serum calcium concentration induced by GBH exposure may arise from the following abnormalities: reduced intestinal absorption when calcium is chelated by glyphosate, 26 decreased calcium absorption when calcitriol level is low due to decreased serum vitamin D, and loss of calcium in renal tubules due to tubular lesions.11,27 The decrease in the serum vitamin D concentration in GBH-exposed groups may be due to reduced absorption of the vitamin from deranged digestive processes when exocrine pancreas is damaged by GBH exposure.21,28 Enzymes for fat digestion – lipase and amylase – were reported to be decreased in the intestinal tract of the crab exposed to GBH. 29 Vitamin D absorption is improved by dietary fat and its association with cholesterol, and decreased fat digestion may likely impact on vitamin D absorption. 30 Apart from altered calcitriol-induced calcium absorption through paracellular and transcellular pathways, 31 reports indicate that GBH induces oxidative stress in the intestinal tract causing inflammatory responses and villous atrophy capable of reducing micronutrient absoption.31,32 Calcium absorption is inhibited by oxidative stress and intestinal inflammation. 31 Therefore, the tendency toward hypocalcemia is possibly caused by a combination of factors involving enteropathy, 31 nephropathy, 9 exocrine pancreatic disorder,21,33 impaired luminal calcium concentration, and bioavailability in the intestine26,34 and inadequate active vitamin D. Persistent hypocalcemia usually elicits a secondary hyperparathyroidism associated with increased serum parathormone concentration. 35
Our observation of decreased serum calcium concentration indicated disturbance of calcium homeostasis. Serum concentration of parathormone had increased due to hypocalcemia and hypovitaminemia D in order to mobilize calcium from the bone and restore calcium balance in the peripheral blood.35,36 As the calcium is moved from the bone, the phenomenon causes bone resorption and decreases bone mineral density. 18 When the GBH exposure was extended from subchronic to chronic duration, it was observed in our earlier report that the hypocalcemia transitioned to hypercalcemia because of endocrine response in secondary hyperparathyroidism. 9 It was the lower dose of GBH exposure in G1 that caused significant hypocalcemia, but the higher dose in G2 did not reveal hypocalcemia because the increasing level of parathormone released might have adjusted the blood calcium to the normal level.
Necrosis of parathyroid gland, bone, and skeletal muscle cells was observed in the GBH-exposed groups (G1 and G2). Cell death in GBH toxicity might have been caused by toxicant-induced oxidative stress followed by activation of calcium influx into the mitochondria and cytosol of the dying cells.37,38 In the parathyroid gland, hormone-producing cell that grow and proliferate are sensitive to death by lipid peroxidation. 39 The oxidative stress by GBH is expected to affect calcium homeostasis and calcium dependent signaling pathways that could lead to cell death. 40 Damage-induced release of hormone by endocrine cells after necrosis has been reported. 39 In areas of parathyroid necrosis, fewer cells were observed because of loss of endocrine cells. Dead cells and debris were probably scavenged out by phagocytes with the capacity to control inflammation in the organ.41,42 In the bone, osteocyte death by necrosis would be associated with osteoclastic bone resorption and osteoid demineralization to release calcium to the blood.43,44 The implication is that GBH could induce osteoporosis due to rarefaction of the bone matrix and reduction of total mineral density. 18 This could be the direct preliminary evidence incriminating GBH in bone damage, as none exists at present despite the debate and controversy over the matter.45,46 The bone, as part of the musculoskeletal system, is closely linked to the skeletal muscle for ambulation of the individual. The involvement of the muscle in cell death, thus, heralds the possibility of the GBH exposure being a cause of frailty in exposed population. 16 The myofibres were necrotic and fragmented without obvious indication of inflammation in the observed foci of the lesion, suggesting a form of toxic necrotizing non-inflammatory myopathy 47 due to oxidative damage. GBH-induced oxidative stress damages contractile myofilaments by abnormal contractility and calcium signal pathway, resulting in muscle fatigue and weakness16,48 exacerbated by oxidant-induced depressed bioenergetics arising from insulin resistance. 49
The pretreatment with zinc before GBH exposure in groups ZG1 and ZG2 mitigated the toxic effects observed on serum calcium, vitamin D and parathormone concentrations, and eliminated the necrotic lesions in the parathyroid gland, bone tissue and skeletal muscle. This could have been possible because of the antioxidant property of zinc19,20 which counteracted the oxidative stress imposed by GBH exposure. Our earlier studies also reported that zinc supplementation in GBH exposed rats could reverse, prevent or reduce the toxic effect of the herbicide.8,9,21 -23 Another vital characteristic of zinc is that its oral intake does not affect normal intestinal calcium absorption from dietary sources. 50 Physiological functions of vitamin D require zinc as a cofactor and zinc supplementation increases vitamin D levels and induces calcitriol release. 51 Zinc is required for growth and metabolism of the bone because of its link with calcium absorption. 52 Thus, zinc supplementation will promote bone health and protect against the adverse effect of GBH exposure.
Calcitonin is a hormone released in response to hypercalcemia in calcium homeostasis. We could not include calcitonin among our parameters because of resource inadequacy. It could have been additional information to know how calcitonin responded in relation to other parameters measured. With increasing blood calcium concentration, calcitonin is released to decrease osteoclast activity and bone resorption, and promote bone formation. 53 The study would have benefited by elucidating the feedback mechanism as it relates to calcitonin response to osteocyte damage induced by GBH.
Conclusion
Subchronic oral exposure of rats to GBH caused a disturbance of calcium homeostasis by decreasing serum calcium and vitamin D concentrations and increasing serum parathormone concentration, with necrotic changes in endocrine-producing cells of the parathyroid gland, osteocytes in the femoral bone and skeletal muscle cells in the biceps femoris; but these toxic actions of GBH were prevented when zinc supplementation was done during the exposure.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Emmanuel Vandi Tizhe: Conceptualization, literature review,experimental data collection, data analysis, writing draft manuscript, and funding. Ikechukwu Onyebuchi IGBOKWE: Conceptualization, supervision, literature review, data analysis, critical review and writing of manuscript. Celestine Onwu-Ibe NJOKU: conceptualization, supervision and critical review. Mohammed Yakasai FATIHU: Conceptualization, supervision and critical review. Ussa Delia TIZHE: literature review, data collection and analysis. Najume Dogon-Giginya IBRAHIM: Conceptualization, supervision, critical review.
