Abstract
Breast cancer is the leading cause of cancer-related death among women in Saudi Arabia. Many studies have suggested a strong correlation between vitamin D and multiple types of cancer. This study included 100 female Saudi patients with early or locally advanced breast cancer. Patients were recruited from King Faisal Hospital in Taif City, Saudi Arabia, from January 2020 to September 2020. We aimed to study the association between serum vitamin D, calcium, interleukin-6 (IL-6), tumour necrosis factor-alpha (TNF-α) and chemerin and breast cancer progression. The control group consisted of 100 healthy individuals. Serum levels of vitamin D, calcium, IL-6, TNF-α and chemerin were measured in all participants. Vitamin D was significantly decreased in patients with high-grade tumours (p < 0.0001), obesity (p = 0.013), negative oestrogen receptors (p < 0.0001), negative progesterone receptors (p < 0.0001) and positive HER2 receptors (p < 0.0001). Vitamin D was also decreased in patients with large tumours (p < 0.0001), axillary lymph node involvement (p < 0.0001) and advanced-stage cancers (p < 0.0001). Moreover, higher levels of IL-6, TNF-α and chemerin were significantly associated with the presence of breast cancer, particularly in its advanced stages. Vitamin D deficiency and elevated levels of IL-6, TNF- α and chemerin were associated with adverse clinicopathological features of breast cancer. Vitamin D deficiency and elevated inflammatory cytokines (IL-6, TNF-α and chemerin) were associated with the clinicopathological features of breast cancer in female Saudi patients.
Introduction
Breast cancer has been recognised as the leading cause of cancer-related death in women worldwide and constitutes approximately one-third of all malignancies in females. 1 In 2018, nearly 2,088,000 new breast cancer cases were diagnosed, and 6,260,000 deaths from breast cancer were recorded. 2 In Saudi Arabia, it is not only the most common malignant tumour in women, with a prevalence of 21.8%, but also the ninth leading cause of death. 3
Vitamin D deficiency is considered a global public health issue. In Saudi Arabia, the prevalence of vitamin D deficiency is approximately 60% across all ages and both genders.4,5 Serum 25(OH)D level has been determined to be the best biomarker of vitamin D status due to its greater half-life and stability in circulation compared to the active form of vitamin D. 6 A person is considered to be vitamin D deficient with a serum 25(OH)D level of <20 ng/mL, vitamin D insufficient with 20–30 ng/mL and vitamin D sufficient with >30 ng/mL. 7
Substantial scientific evidence has indicated a strong relationship between vitamin D and calcium levels and cancer. 8 The findings of a recent study, which reported a significant relationship between low serum vitamin D and high breast cancer risk, indicate that vitamin D may serve in a protective capacity against breast cancer. 7 The relationship between calcium levels and breast cancer risk is thought to be predominantly triggered by the chemo-preventive action of the active form of vitamin D, which is the primary regulator of calcium homeostasis. 9
Vitamin D deficiency may lead to uncontrolled metabolic syndromes that result in carcinogenesis. 10 The biologically active form of vitamin D (1,25(OH)2D3) is produced in the kidney by the action of 1-alpha hydroxylase (CYP27B1). CYP27B1, in turn, is produced by other tissues and immune cells that regulate their local levels of active vitamin D through it. 11 The expression of CYP27B1 in immune cells is triggered either by the presence of cytokines directly 12 or through immune regulatory mechanisms associated with vitamin D receptor activation. 13
A deficiency in vitamin D may cause immune cell dysfunction 14 and cytokine variability.15,16 Research has indicated that adequate levels of 25(OH)D are associated with increased levels of interleukin (IL)-4 and IL-10 and decreased levels of pro-inflammatory cytokines, such as IL-1, IL-6, IL-8 and tumour necrosis factor-alpha (TNF-α). Vitamin D down-regulates the expression and production of these pro-inflammatory cytokines. 17
The effect of pleiotropic cytokines on cancer development has been well established. In fact, treatment efficacy and prognosis in malignant diseases are assessed by the disease stage and the function of immune system pathways mediated by IL-6, IL-8 and TNF-α.18,19
The inflammatory pathogenic environment present in breast cancer may provide a mechanism by which chemerin expression becomes elevated, leading to an association between breast cancer and heightened chemerin levels. 20 Chemerin is an adipocytokine responsible for regulating multiple immunological and biological functions, primarily acting through chemokine-like receptor 1, which is expressed by many cells.21,22 Chemerin is associated with inflammation, cell metabolism and cellular differentiation and its release is governed by numerous mediators, including vitamin D, retinoids, corticosteroids and the cytokines that regulate acute and chronic inflammation. 23 Chemerin is linked to the pro-inflammatory cytokines IL-6, IL-8 and TNF-α which mediate cytotoxic cellular immunity and are considered inflammatory promoters of breast carcinogenesis.24,25
According to current epidemiological evidence, vitamin D may play a protective role against breast cancer through its anti-inflammatory and anticancer actions and specific signalling pathways. Furthermore, a meta-analysis has revealed a significant dose–response association between circulating vitamin D and overall survival in breast cancer patients. 26
In the current study, we aimed to evaluate the association of vitamin D deficiency and certain inflammatory cytokines with the progression of breast cancer in female Saudi patients.
Patients and methods
Study design and population
Characteristics of patients and control.

Participants were included if they were female and had an early or locally advanced breast cancer diagnosis without metastasis. Patients diagnosed with other types of cancer or metastasis were not included. In addition, to ensure that true serum vitamin D and cytokine levels were measured, patients who received vitamin D or calcium supplementation and patients who were under anticancer (hormonal, chemotherapy or radiotherapy) or anti-inflammatory treatment were excluded from the study.
Before acceptance into the study, all patients underwent a thorough history, physical examination, complete blood count, liver and kidney function tests, chest X-ray, pelvic and abdominal ultrasonography, echocardiogram, magnetic resonance imaging of both breasts and isotopic bone scan. Oestrogen hormone receptors (ER), progesterone hormone receptors (PR) and HER2 receptors were determined by immunohistochemistry using SP1, 1E2 and 4B5 (Ventana) antibodies, respectively. Weight (kg) and height (m) were measured to calculate BMI using the equation BMI = kg/m2.
Blood samples
We obtained 5 mL of venous blood from all included participants. The samples remained at room temperature for 1 h to clot in the serum separator tubes before being centrifuged for 5 min at 3,000 r/min. The sera were collected and stored at −20°C until analysis. In the patient group, all samples were taken at the time of diagnosis, before surgery or administration of chemotherapy or radiotherapy treatment.
Methods
Estimation of serum 25(OH)D level by enzyme-linked immunosorbent assay (ELISA)
Estimation of 25(OH)D level in the serum samples of all included participants was performed using the Abcam human vitamin D enzyme-linked immunosorbent assay (ELISA) kit (USA; Cat No: ab213966) in collaboration with the manufacturer’s protocol. The range of detection was 0.5–1010 ng/mL, and the sensitivity of the assay was 1.98 ng/mL. A serum vitamin D level of less than 20 ng/mL was considered deficient.
Estimation of serum interleukin-6, tumour necrosis factor and chemerin levels (ELISA)
Serum levels of IL-6 were estimated in all participant samples using the ELISA kit (MyBioSource, USA; Cat No: MBS261259), with a detection range of 4.7–300 pg/mL and sensitivity of up to 1 pg/mL. Serum TNF-α levels were assayed using the Abcam human TNF-α ELISA kit (UK; Cat No: ab181421), with a detection range of 15.63–1000 pg/mL and sensitivity of 4.32 pg/mL. Serum chemerin concentration was quantified using the Abcam human chemerin ELISA kit (Cat No: ab155430), with a detection range of 0.51–50 ng/mL and sensitivity of 0.5 ng/mL according to the manufacturer’s guidelines.
Estimation of serum calcium level
Serum calcium was estimated using the Abcam calcium colourimetric assay kit (USA; Cat No: ab102505), with a detection range of 0.4–100 mg/dL according to the manufacturer’s protocol.
Statistical analysis
Data analysis was performed using SPSS 10.00 software (SPSS Inc. Chicago, IL, USA). One-way analysis of variance and student’s t-test were calculated to evaluate the statistical significance between variables. Pearson’s correlation coefficient was used to assess the association between vitamin D and the other studied parameters. p values were considered statistically significant at p < 0.05. The current study had more than 95% power, as computed by G*power 3.1. 28 Specific criteria developed by Miaoulis and Michener were used to determine the necessary number of subjects. 29 Cochran’s formula was employed to calculate the minimum sample size, 30 with a confidence value of 95% at a significant concentration of 5%.
Results
Pathological characteristics of patients.

Serum vitamin D, calcium, IL-6, TNF-α and chemerin levels in patients and healthy controls
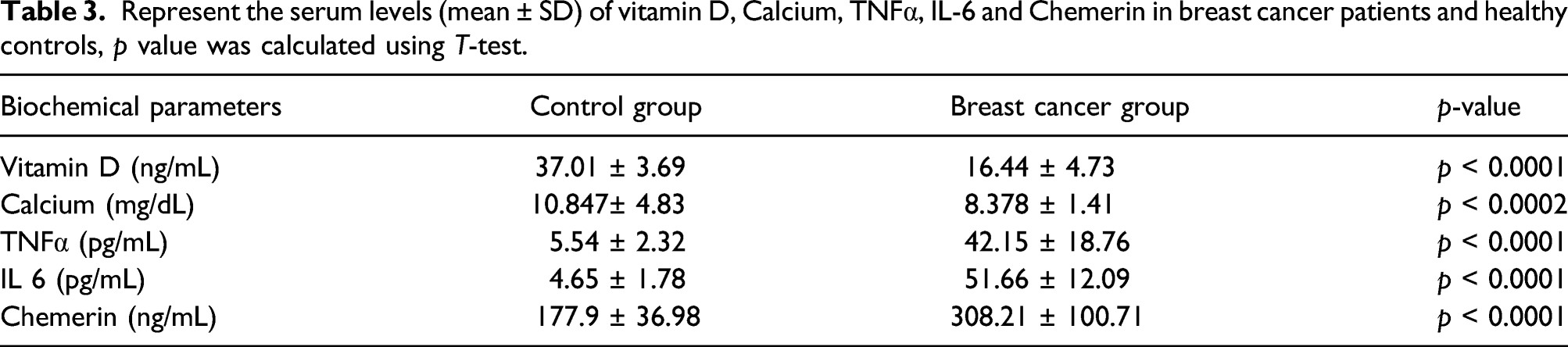
There was a statistically significant difference in serum vitamin D levels in the patient group compared with the control group, with the patient group demonstrating decreased vitamin D levels (student’s t-test = 25.88, p < 0.0001). Mean vitamin D levels in the patient group were 16.44 ± 4.73 ng/mL compared to 37.01 ± 3.69 ng/mL in the control group.
There was a statistically significant difference in serum calcium levels between patients and controls, with a mean calcium level of 8.91 ± 1.46 mg/dL in the patient group compared with 10.85 ± 83 mg/dL in the control group (t = 4.9, p < 0.0001).
Represent the serum levels (mean ± SD) of vitamin D, Calcium, TNFα, IL-6 and Chemerin in breast cancer patients and healthy controls, p value was calculated using T-test.
Relationship between serum vitamin D and calcium levels and clinicopathological features
The relations between the serum levels of vitamin D and calcium, and different clinical and pathological features.

*One way ANOVA. **Student t-test.
Relationship between IL-6, TNF-α and chemerin serum levels and clinicopathological features
Serum IL-6 in the breast cancer patient group was significantly elevated in patients with old age (p < 0.0001) and obesity (p < 0.0001). IL-6 levels were also increased in patients with infiltrative lobular carcinoma (ILC; p < 0.0001), tumours with a high histopathological grade (p < 0.0001), negative ER and PR tumours (p < 0.0001) and positive HER2 tumours (p < 0.0001). In addition, IL-6 was elevated in patients with large tumours (p < 0.0001), axillary lymph node involvement (p < 0.0001) and advanced-stage cancers (p < 0.0001).
Serum TNF-α was significantly increased in patients who were premenopausal (p = 0.029), obese (p = 0.0001) and diagnosed with ILC (p < 0.0001), high-grade tumours (p < 0.0001), negative ER and PR tumours (p < 0.0001) and positive HER2 tumours (p < 0.0001). Serum TNF-α levels were significantly increased in patients with large tumours (p < 0.0001), axillary lymph node involvement (p < 0.0001) and advanced-stage cancers (p < 0.0001).
The relations between the serum levels of IL-6, TNF-α, chemerin and different clinical and pathological features.

*One way ANOVA. **Student t-test.
Correlation between vitamin D, calcium, IL-6, TNF-α and chemerin in the patient group
Pearson’s correlation revealed a significant positive correlation between vitamin D deficiency and calcium level (r2 = 0.298, p < 0.0001), as well as a significant negative correlation between vitamin D deficiency and IL-6 (r2 = 0.537, p < 0.0001), TNF-α (r2 = 0.437, p < 0.0001) and chemerin (r2 = 0.484, p < 0.0001) levels (Figure 1). Pearson’s correlation between serum vitamin D levels and serum calcium (a), IL-6 (b), TNF-α (c) and chemerin (d) in breast cancer patients.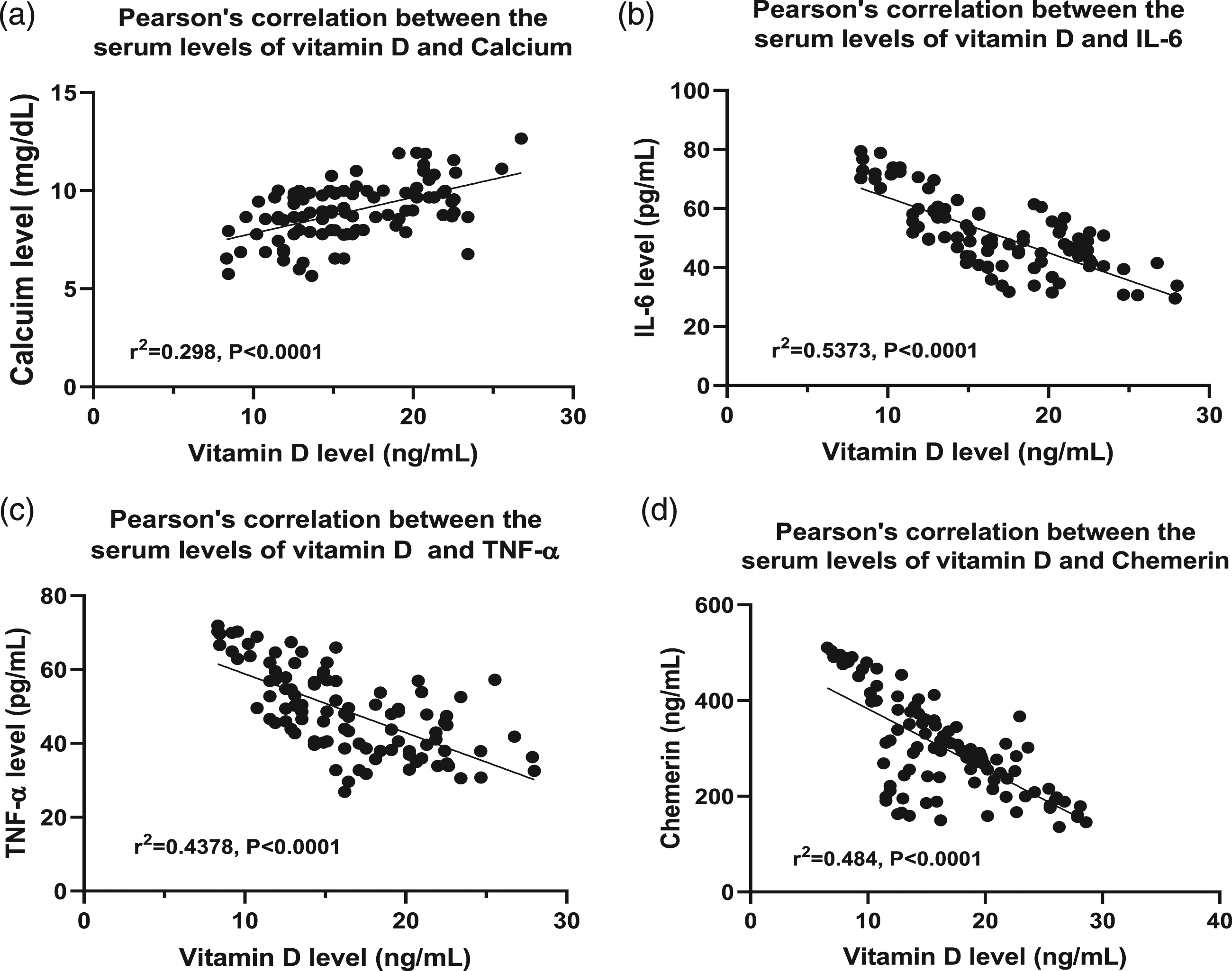
Correlation between vitamin D, calcium, IL-6, TNF-α and chemerin in the control group
Pearson’s correlation revealed a nonsignificant correlation between vitamin D and calcium level (r2 = 0.0017, p = 0.68), as well as a nonsignificant correlation between vitamin D and IL-6 (r2 = 0.026, p = 0.11), TNF-α (r2 = 0.00,072, p = 0.79) and chemerin (r2 = 0.0023, p = 0.63) levels (Figure 2). Pearson’s correlation between serum vitamin D levels and serum calcium (a), IL-6 (b), TNF-α (c) and chemerin d) in healthy controls.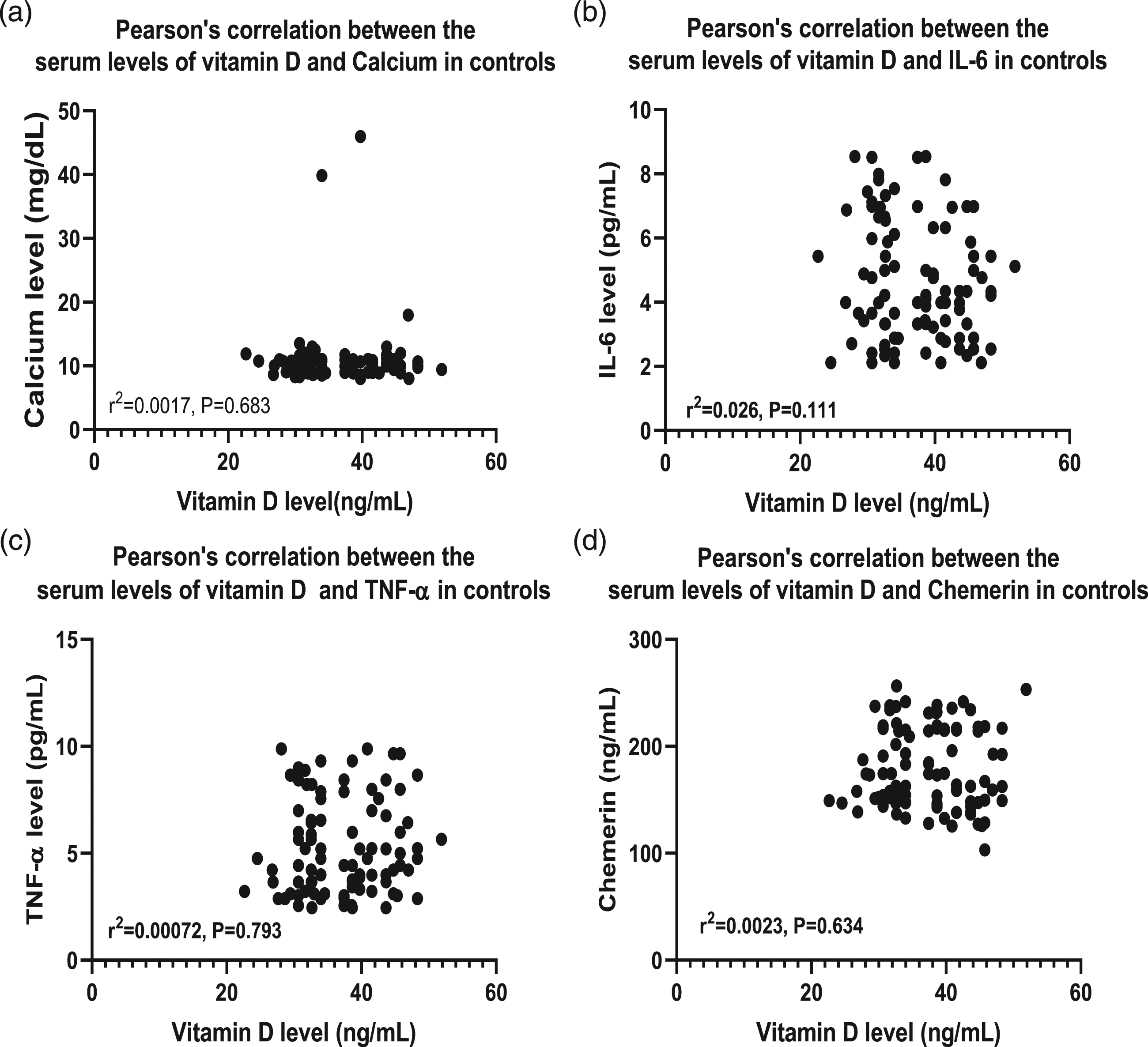
Discussion
Breast cancer is the most prevalent malignant tumour in women. 1 Many of the pro-inflammatory cytokines induced as a result of inflammation in tumours can accelerate tumour progression, enhance angiogenesis and suppress cell apoptosis. 1 Vitamin D, in contrast, has an anti-inflammatory effect on the microenvironment of malignant cells. 31
Improving the prediction of prognosis is one of the cornerstones of optimising outcomes in breast cancer patients. Individualised treatment adaptation is an essential component of increasing survival in patients with a poor prognosis. To that end, the current study explored the relationship between breast cancer and serum levels of 25(OH)D, calcium, IL-6, TNF-α and chemerin. In addition, we studied the relationship between these serum levels and patient-specific clinical and pathological parameters to evaluate their association with breast cancer progression.
The influences of vitamin D on the inflammatory processes of cancer have been studied, and several pathways by which vitamin D may have a protective effect against cancers have been identified. These pathways include the control and regulation of cytokine levels, inhibition of nuclear factor-kappa B (NF-κB) signalling pathways, up-regulation of mitogen-activated kinase protein 5, suppression of immune system cells and inhibition of prostaglandin-regulated mechanisms. 32
Our results revealed a significant reduction in serum levels of 25(OH)D and calcium in breast cancer patients compared to controls (p < 0.0001). Vitamin D deficiency was significantly related to poor prognostic features, such as high histopathological tumour grade, large tumour size, axillary lymph node involvement, negative ER and PR, positive HER2 and advanced clinical stage (p < 0.0001).
These results support the findings of Narvaez et al. 33 and Imtiaz and Siddiqui, 34 who reported on the presence of vitamin D deficiency in breast cancer patients. Moreover, de Sousa et al., 35 Colston and Hansen, 36 and Shaukat et al. 37 reported that low vitamin D levels were associated with an increased risk of breast malignancy. Quiroz et al., 38 furthermore, found that vitamin D inhibited breast carcinogenesis through its enhancement of apoptosis and suppression of angiogenesis and malignant cell division. Vitamin D has an established role in ER signalling pathways as an inhibitor of aromatase enzyme expression in mammary adipose tissue.39,40 It also decreases the expression of ER in breast cancer cells. 41 Interestingly, in ER-negative tumours, vitamin D can induce ER expression and tumour response to antioestrogen drugs. 42
Thus, the protective mechanism of vitamin D against breast cancer cells may be explained by its ability to provide an anti-inflammatory and antioxidant environment that prevents the initiation of malignant transformation and its capacity to repair damage to DNA.43,44
Our results also indicated a significant decrease in serum calcium levels in our patient group compared to controls. This observation is supported by Chen et al., who reported on the protective function of calcium against breast cancer, a relationship that is likely due to the fact that active vitamin D, which has an established chemopreventive mechanism, is responsible for the regulation of calcium homeostasis. 45
The findings of a previous study indicated that vitamin D and calcium supplementation in premenopausal women reduced their percent mammographic density (MD), which is considered a risk factor for breast cancer. 46 Premenopausal women and those with high serum insulin growth factor 1 (IGF-1) levels were shown to have a stronger link between vitamin D and MD, suggesting that vitamin D may affect MD via IGF-1 signalling. 47 Vitamin D supplementation lowered the overall incidence of advanced (metastatic or lethal) cancer in this randomised clinical study, with the highest risk reduction reported in people of normal weight. 48
Similarly, Colston and Hansen observed an inverse relationship between breast cancer risk and sunlight exposure, which increases the dermal synthesis of vitamin D. 36 In contrast, Chlebowski et al. 49 reported that there was no relationship between 25(OH)D levels and breast cancer risk and that vitamin D and calcium supplementation in postmenopausal women did not reduce breast cancer risk. Furthermore, Crew et al. 50 and Li et al. 51 revealed that MD was favourably linked to serum IGF-1 and IGF-1/IGFBP-3, though the ability of vitamin D supplementation to reduce the incidence of breast cancer was not proved.
Inflammation is considered a key component of the malignant microenvironment, as immune cells and cytokines produced as part of the inflammatory process have a direct influence on malignant cell growth and migration.52,53 In this study, a significant increase in serum levels of IL-6, TNF-α and chemerin was found in patients compared to controls (p < 0.0001). The results also indicated a statistically significant relationship between these elevated levels and adverse prognostic features, including ILC, high-grade tumours, negative hormone receptor (ER and PR) tumours, positive HER2 tumours, large tumours (T2 and T3 on the TNM scale), axillary lymph node involvement (N2 and N3 on the TNM scale) and advanced clinical stage cancer (IIB and IIIA on the TNM scale; p < 0.0001).
Shuchen et al. 54 similarly concluded that increased levels of serum IL-6 were significantly related to breast cancer and its advanced stages. Furthermore, elevated IL-6 levels were associated with a poor prognosis. On the other hand, a study by Ahmad et al. 55 found that high IL-6 and IL-10 levels were related to positive prognostic indicators, including small tumour size and low histopathological tumour grade. Some clinical studies have reported high levels of serum TNF-α in patients with breast cancer compared to healthy women. 56 High levels of TNF-α messenger RNA expression have also been found in primary breast cancer tissue compared to normal breast tissue. 57 Kesler et al. 58 reported that IL-6 and TNF-α levels were significantly increased in breast cancer patients compared to controls. Yunfeng et al. 59 also concluded that increased levels of serum IL-6, IL-8 and TNF-α were significantly associated with breast cancer clinical stage as well as ER and HER2 expression in tumours. These pro-inflammatory cytokines have thus been considered possible prognostic biomarkers of breast cancer.
The evidence is controversial regarding the relationship between chemerin and cancer. In agreement with the results of this study, Sarmadi et al. 60 found elevated chemerin in breast cancer tissue and reported that the pro-inflammatory cytokines IL-1β, TNF-α, IL-6 and interferon-γ up-regulated chemerin expression in cancer tissues. Wang et al. 61 additionally reported that elevated chemerin levels were associated with markers of aggressive squamous cell cancer of the tongue, such as advanced stage and increased tumorigenesis. In contrast, Serkan et al. 62 found that serum chemerin levels were not associated with breast cancer stage. Parolini et al. 63 suggested that chemerin may play a protective role against cancer through the enrolment of natural killer cells. According to their research, increasing chemerin levels enables the immune system to identify and combat cancer cells.
We analysed the correlation between serum levels of vitamin D and serum calcium, IL-6, TNF-α and chemerin in our patients. A significant positive correlation was found between serum 25(OH)D and calcium. Moreover, there was a significant negative correlation between serum 25(OH)D and IL-6, TNF-α and chemerin. Consistent with our findings, Benetti et al. 64 suggested that vitamin D may act as a negative regulator of pro-inflammatory cytokine release by suppressing the activation of NF-κB and consequently the transcription of its downstream pro-inflammatory cytokines.
This study had some limitations that warrant mentioning. The small number of patients may not be representative of the population, though it may provide guidance that aids researchers in performing larger studies in the future. In addition, the study was underpowered to examine the patients’ overall survival outcomes due to a lack of follow-up.
Conclusion
Based on our results, it can be concluded that vitamin D deficiency and elevated levels of IL-6, TNF-α and chemerin are associated with the clinicopathological features of breast cancer. In addition, the significant relationship between these parameters and adverse prognostic features may introduce them as predictors of poor prognosis of breast cancer in women. Further studies are needed to clarify whether the results described are the cause of the disease or simply a consequence.
Recommendation
Further studies with large samples of patients are required to investigate the clinical significance of using vitamin D supplementation in patients with early diagnosed or locally advanced breast cancer. We recommend adding articles as a preprint, and we acknowledge Research Square Company for adding our article as a preprint.
Footnotes
Acknowledgements
The authors would like to extend their sincere thanks to the Deanship of Scientific Research, Taif University, Taif, Saudi Arabia, for funding of this research through the Research Group Project number: (1-441-95).
Author contributions
The study was conceptualised and designed by authors AG, MA and WE. AE, MA and AA wrote the initial manuscript. Final data preparation was performed by authors AG, WE and MA. The statistical analysis was completed by author WE and the final data analysis and interpretation were assembled by authors WE, AG and AE. HA, LE and HA revised the manuscript. All contributing authors took part in editing and reviewing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors would like to extend their sincere thanks to the Deanship of Scientific Research, Taif University, Taif, Saudi Arabia, for funding of this research through the Research Group Project number: (1-441-95).
Data availability statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval
All procedures were performed in accordance with the ethics of the Declaration of Helsinki. The study was approved by the Directorate of Health Affairs in the Taif region in Saudi Arabia and the Institutional Review Board (IRB) of King Abdulaziz City for Science and Technology (KACST; IRB Registration number with KACST, KSA, HAP-02-T-067; approval number 294).
Consent to participate
Written informed consent was obtained from all participants before enrolment in the study.
