Abstract
The aim of this study was to evaluate whether the control of ocular symptoms with cyclosporine or with tacrolimus in eye drops allows to improve sun exposure and therefore serum level of vitamin D (VD; 25OHD), in the more severe forms of vernal keratoconjunctivitis (VKC). Out of 242 children followed for active VKC, 94 were treated with 1% cyclosporine or 0.1% tacrolimus eye drops, while the other 148 with mild VKC did not need to be treated with immunomodulators. VD serum levels were measured in spring and autumn in 71 children. In total, 60 of them were treated with cyclosporine eye drops (first group) and 11 (not responding to cyclosporine therapy previously) with 0.1% tacrolimus eye drops (second group) between March and November 2016. Pre-treatment median values of VD were 23.7 ng/mL in the first group and 23.8 in the second group, and post-treatment values increased up to 32.8 and 32.9 ng/mL, respectively. Before treatment, 33% presented a deficiency (25OHD < 20 ng/mL), and at the end of summer, only 4% were deficient. The overweight children had lower improvement in VD serum levels than children with a body mass index (BMI) lower than 85th percentile. Children in therapy with cyclosporine, but requiring the administration of local steroid therapy during the summer for control of the symptoms, showed a greater improvement in 25OHD serum levels in ng/mL (23–37 ng/mL) than children who did not require steroid therapy (24–35 ng/mL). Furthermore, there was a significant difference in change of 25OHD in children presenting limbal VKC (21–41 ng/mL) versus tarsal VKC (24–35 ng/mL) (P = 0.04). Our study suggests that ocular treatment carried out with immunomodulator eye drops could allow for an improvement in 25OHD serum levels. In children with active VKC and at risk of 25OHD deficiency, likely due to avoidance of sun exposure, the role of other risk factors (BMI, phototype and treatment) on 25OHD serum levels should be considered.
Introduction
Vernal keratoconjunctivitis (VKC) is a chronic keratoconjunctivitis, manifesting itself especially in males below 10 years. It is always bilateral, but often asymmetrical. Its prevalence is extremely variable and increases towards the equator. In fact, it is highest in Africa, with 6% reported in Ethiopia, much lower in Italy (0.27%) and close to 0% in Scandinavian country. 1 The causes of VKC are still under investigation, but the condition seems strongly related to sunlight exposure, responsible for the worsening of the disease.
VKC is characterised by itching, photophobia, white mucous discharge, lacrimation and foreign body sensation. 2 There are three forms of VKC: tarsal, limbal and mixed. The tarsal form is characterised by the presence of papillae in the upper tarsal lid, while the limbal form by gelatinous infiltrate in the limbus and Trantas’ dots.
Although VKC usually resolves after puberty, it can lead to severe visual impairments if not adequately treated, such as visual loss (reported in 5%–30% of cases), cataracts and glaucoma, caused by a prolonged use of steroid eye drops.
The VKC therapy aimed at the control of patient’s symptoms, at the objective improvement of the signs of VKC (tarsal large cobblestone papillae, Trantas’ dots and infiltrates in the limbus) and at the avoidance of complications, among which the most fearful are corneal ones (peri-corneal vascular infiltration, superficial punctate keratopathy and shield ulcer). 2 For this purpose, the main drug, which proved to be more effective, is the steroid eye drops used locally in short cycles. In case of more severe forms, in which steroid eye drops would be necessary for long periods, immunomodulators such as cyclosporine eye drops are used with good results. In the most severe and resistant forms to cyclosporine eye drops, that is, in the forms in which prolonged cycles of steroid eye drops are necessary to control the patient’s symptomatology and improve the clinical ocular objectivity, 0.1% tacrolimus eye drops have been used in recent years. 3
Vitamin D (VD) is a prohormone substance regulating a wide range of functions in the human body, including the mineral level of bones and the immune system response. The main source of VD is sunlight exposure. VD deficiency can lead to rickets, an increased risk of airway infections and autoimmune diseases such as diabetes mellitus type I and cancer. Serum levels of 25-hydroxyvitamin D (25OHD) below 20 ng/mL, reported in up to 50% of children, 4 indicate a deficiency in VD, and insufficient levels of 25OHD (<30 ng/mL) are found in 80% of paediatric patients. 4 Children affected by VKC tend to avoid sunlight exposure and outdoor activities during spring and summer, the hottest months of the year, 5 thereby possibly increasing the risk of VD deficiency.
The aim of this study was to evaluate VD serum levels in children affected by VKC and to verify whether adequate ocular treatment 6 can affect 25OHD levels.
Materials and methods
A total of 242 children were followed in 2016 for active VKC, classified according to Bonini’s grading. Of the 242 children, 94 were treated with 1% cyclosporine or 0.1% tacrolimus eye drops, while the other 148 with mild VKC did not need to be treated with immunomodulators. From these 94, we excluded children who took VD supplementation during 2016 (n = 2) and children who had their 25OHD level checked only in spring (n = 9) or in autumn (n = 12) in 2016. In total, 71 children were followed; 60 of them were treated with cyclosporine eye drops (first group) and 11 (not responding to cyclosporine therapy in the previous year) with 0.1% tacrolimus eye drops (second group) between March and November 2016. If the symptoms could not be controlled, the children were instructed to receive steroid (dexamethasone sodium phosphate) eye drops (one drop) three times daily for 3 days up to three cycles per month. VD levels were measured in spring and autumn in all enrolled patients, with an average time difference between the first and the second tests of 170 days (interquartile range (IQR) = 140–200).
The children’s ages spanned from 5 to 17 years, with a median age of 9.6 years, 72% were males, and 31% were overweight (>85th percentile).
The study was approved by the local Institutional Ethics Committee (no. 259_2018bis) and was carried out according to the Ethical Principles of the Declaration of Helsinki. All the patients’ legal guardians gave their informed consent.
Two treatment groups were obtained: group 1 included 60 children treated with 1% cyclosporine eye drops and group 2 included 11 children treated with 0.1% tacrolimus eye drops.
The following data of each patient have been recorded: age, gender, body mass index (BMI), phototype (Fitzpatrick’s scale), 7 atopy, type of VKC, dosage of inhalant allergen-specific IgE or positive skin prick test. The VD test was performed with Liaison© 25(OH) Vitamin D Total Assay.
25OHD serum levels are defined as follows: 8
Severe deficiency: 25OHD < 10 ng/mL;
Deficiency: 25OHD between 10 and 20 ng/mL;
Insufficiency: 25OHD between 20 and 30 ng/mL;
Normality: 25OHD between 30 and 100 ng/mL;
Toxicity: 25OHD > 100 ng/mL.
Results are presented as median, quartiles (Q1 and Q3) and ranges, or percentages for continuous variables or categorical variables, respectively. Differences between groups were first investigated with univariate analyses with Wilcoxon rank tests for differences in median values, chi-square and Fisher’s exact tests for differences among frequencies. Multivariate linear regression analysis of covariance (ANCOVA) models were applied to investigate the association between change in 25OHD after treatment and ocular treatment, type of VKC, phototype, BMI, atopy, adjusting for season, values at baseline and evaluating the role of age and gender. The normal distribution of residuals was graphically checked.
We reported two-sided P-values based on type III sum of squares. We set the criterion for statistical significance at 5%. The data were analysed using the SAS System Software for Windows, release 9.3. (SAS Institute, Cary, NC, USA).
Results
The median age at diagnosis was 6 (IQR = 5–8) years, and the age at first symptoms was 5 (IQR = 3–6) years. A total of 46 children had an active mixed VKC, 19 had a tarsal form and 6 had a limbal form; 52 children (72%) were males.
Of these, 61% had a normal weight (BMI between 10th and 85th percentile), 8% low weight (<10th percentile) and 31% overweight (>85th percentile); 70% had prick tests or allergen-specific IgE which were positive for birch, hazel, olive trees, grasses, pellitory, ambrosia, absinth, dermatophagoides farinae, dermatophagoides pteronyssinus, alternaria and cat and dog dander; and 14% of the children had phototype II, 68% phototype III, 17% phototype IV and 1% phototype VI.
No significant differences were observed between the two treatment groups in terms of age, gender, BMI and phototype. Pre-treatment median values of 25OHD were about 23.7 ng/mL in the first group and 23.8 in the second group, while post-treatment values increased up to 32.8 and 32.9 ng/mL, respectively. The median rise of 25OHD values was 10.2 (IQR = 2.25–16.25) ng/mL in the first group and 11.8 (IQR = 6.5–15.6) ng/mL in the second group.
In spring and summer, 81% of the children showed insufficient 25OHD serum levels (<30 ng/mL) and 33% presented a deficiency (25OHD < 20 ng/mL); at the end of summer, these percentages decreased to 39% and 4%, respectively (Figure 1). A severe deficiency of 25OHD (with 25OHD levels <10 ng/mL) was never observed. There was not a statistically significant difference between the two treatment groups. Children in group 1 who required the administration of local steroid therapy during summer (more than 10 days per month for at least two consecutive months) showed greater improvement in 25OHD serum levels than the children who did not require local steroid therapy. 25OHD serum levels increased from 24 (IQR = 18–29) ng/mL in spring to 32 (IQR = 27–39) ng/mL in autumn in children who did not require steroid treatment and from 23 (IQR = 20–27) ng/mL to 37 (IQR = 28–44) ng/mL in children who required steroid treatment, with a median improvement of 14 versus 8 ng/mL (Figure 1). The difference was significant in univariate analysis (P = 0.05) but not in multivariate analysis (P = 0.09).

Box plots of change in 25OHD by use of local steroid therapy.
Children affected by limbal VKC had a greater increase in 25OHD serum levels than children affected by tarsal VKC (Figure 2). This difference was statistically significant in univariate analyses (P = 0.02) and lost significance (P = 0.40) in multivariate analyses, probably due to the low number of patients with limbal VKC (N = 6). Children with limbal VKC had a 25OHD serum level of 21 (IQR = 17–23) ng/mL before summer and 41 (IQR = 33–42) ng/mL in autumn, while children with tarsal VKC showed levels of 24 (IQR = 19–30) ng/mL in spring and 35 (IQR = 27–39) ng/mL in autumn. Children with no allergic sensitivities had a greater increase in 25OHD compared to children with positive prick tests or IgE positivity for at least one inhalant allergen (median change = 12 vs 8 ng/mL, respectively), but the difference was not statistically significant (P = 0.23).

Box plots of change in 25OHD by type of vernal.
Children with phototypes II and III presented greater improvement in 25OHD serum levels than children with phototype IV (median values = 11 vs 7 ng/mL, respectively; P = 0.02, adjusted for the sampling month after treatment and 25OHD levels at baseline), even if the values after treatment were greater among children with phototype IV than those with phototypes I and II (median values = 38 vs 33 ng/mL, respectively; P = 0.02, adjusted for the sampling month after treatment and 25OHD levels at baseline) (Figure 3).
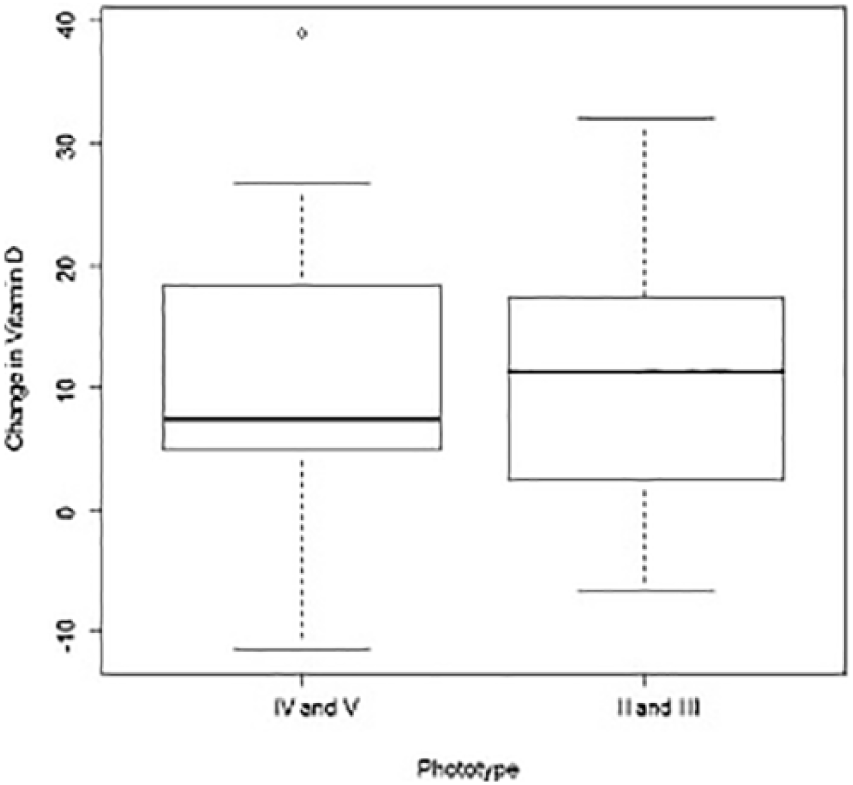
Box plots of change in 25OHD by phototype.
Overweight children had a less significant improvement in 25OHD serum levels than children with a BMI below 85th percentile. In fact, the median improvement was 7.3 ng/mL in the first group and 9.8 ng/mL in the second one. Before and after summer, median 25OHD serum levels were 25.4 and 33.2 ng/mL in the first group and 29.1 and 37.9 ng/mL in the second one.
Discussion
Our study suggests that ocular treatment carried out with 1% cyclosporine or with 0.1% tacrolimus eye drops could allow a clinical improvement of VKC signs and symptoms and a better tolerance to sun exposure, as evidenced by the increase in 25OHD serum levels. There was not a statistically significant difference in terms of 25OHD increase between group 1 (cyclosporine eye drops) and group 2 (tacrolimus eye drops), but the sample size did not provide adequate statistical confidence to be able to draw definitive conclusions on this aspect.
Others factors that could affect the reliability of our conclusions are the absence of a control group and of precise information about the patients’ sun exposure and well-being and about the bioavailability of the immunomodulator drops.
This study confirms the presence of hypovitaminosis D in children affected by VKC, as noted by Zicari et al. 9 and Bozkurt et al. 5
Children who required the administration of steroid ocular therapy during the treatment period with cyclosporine eye drops had greater improvement in 25OHD levels in summer when compared to those who did not require steroid treatment to fully control the disease.
Children affected by limbal VKC had a significantly higher post-treatment increase in 25OHD levels than children affected by the tarsal form. A possible explanation for this could be found in the fact that limbal VKC is less symptomatic than tarsal or mixed VKC forms, thus consenting greater sunlight exposure.
Even if the importance of BMI, phototype and sampling month is known to be a determinant in 25OHD serum levels, our study is the first to stress their importance in the VKC population as well.
A confirmation of our results through large-scale studies can lead to not only customised therapies but also a deeper insight into the pathogenesis of VKC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
