Abstract
Adamantinoma (AD) is a rare, slow-growing primary malignant bone tumor characterized by a biphasic morphology of clusters of epithelial cells and spindle cell osteofibrous components. A strong relationship between AD and osteofibrous dysplasia (OFD) has been proposed, while fibrous dysplasia (FD) has been rarely associated with AD. We present an AD case that was followed and histologically evaluated 3 times over 6 years with different morphological patterns. The tumor in the primary biopsy and after complete resection showed classical features of AD and osteofibrous-like pattern, while the recurrent lesion presented with exclusively spindle cell morphology and was thus diagnosed as FD. However, the extensive immunohistochemical analysis in all 3 lesions revealed strong reactivity for pancytokeratin, vimentin, p63, and podoplanin, which are characteristic for AD. Although, in the FD-like section of the tumor from the first recurrence the positivity of podoplanin was stronger than pancitokeratin, which was variably positive on spindle cells. The present case highlights the problem of diagnosing AD based on a single biopsy with one tumor’s component predominating over the other, and at the same time emphasizes the importance of using immunohistochemical staining for keratin and podoplanin when the histopathological features of (osteo)fibrous lesion can be linked to AD.
Introduction
According to the WHO definition, the adamantinoma (AD) is a biphasic locally aggressive or malignant tumor characterized by a variety of morphological patterns, with a variable epithelial component within a bland osteofibrous component. 1 The tumor is classified into 3 distinct subtypes: classic, osteofibrous-like, and dedifferentiated. The main component in classic AD is the epithelial, with 4 growth patterns (basaloid, tubular, spindle-cell and squamous), in the osteofibrous-like AD (OFD-AD), it is the osteofibrous tissue with small clusters of epithelial cells, positive for cytokeratin, 2 while in the dedifferentiated subtype the areas of classic AD gradually combine with a diffuse growing proliferation in which the epithelial differentiation is lost.3 -5 Case reports suggest the possibility of progression from OFD-AD to AD.6,7 Other studies have also reported Ewing-like type 8 and rhabdoid variants of AD. 9 Since the listed components can be differently represented in the lesion, difficulties in distinguishing some cases of AD, especially from osteofibrous dysplasia (OFD) or spindle cell sarcomas, can emerge.
The real histogenesis and pathogenesis of AD is still being debated. In the past, several hypotheses have been made regarding the origin of the tumor. Some authors supported the hypothesis of congenital epithelial cell implantation, others argued for traumatic implantation or articular origin, while recent studies indicate the role of mesenchymal-to-epithelial transformation, leading to the origin of the epithelial component.10 -13
Similar clinical, radiological, histological and molecular features of AD and OFD suggest a relationship between the two.14 -19 Despite these similarities, the treatment and prognosis differ significantly; therefore, the establishment of diagnosis should be performed precisely. On the contrary, fibrous dysplasia (FD) is not believed to be linked to AD.11,20
Adamantinoma is frequently localized in long tubular bones, mostly in the tibia, 2 as it was in our patient who developed recurrent lesions, whose histological features ranged from a classic subtype of AD with a mostly tubular growth pattern in the primary lesion to lesions histologically indistinguishable from FD, and furthermore to a classic subtype with a basaloid growth pattern and foci of osteofibrous-like growth in the resected bone, thus representing a spectrum of epithelial-to-mesenchymal and mesenchymal-to epithelial transformation.
Case Report
A 70-year-old female patient sought medical attention due to a progressive tumorous lesion on her left tibia that was causing pain and swelling, which she had been attempting to treat for the past 6 years at different institutions. The older medical documentation, including X-rays, revealed that the lesion was previously treated by excochleation and spongioplasty, followed by preventive osteosynthesis. Radiological studies of the lesion initially demonstrated an irregularly expanding osteolytic lesion in the diaphysis of the distal tibia, with well-defined transition zones and solid periosteal reactions, without destruction. Six years after, radiographs show distal extension of the osteolytic lesions to the subarticular zone (Figure 1a and b). The first histological analysis demonstrates a classic AD (features of tubular epithelial cells), positive for pancytokeratin and vimentin, in a well-vascularized stroma (Figure 2). The recurrent lesion was characterized by spindle, fibroblast-like cells with uniform, oval nuclei, along with small and disconnected bone trabecule without osteoblastic rimming (Figures 2 and 3a). No further immunohistochemical evaluation was performed, and the diagnosis of FD was made.
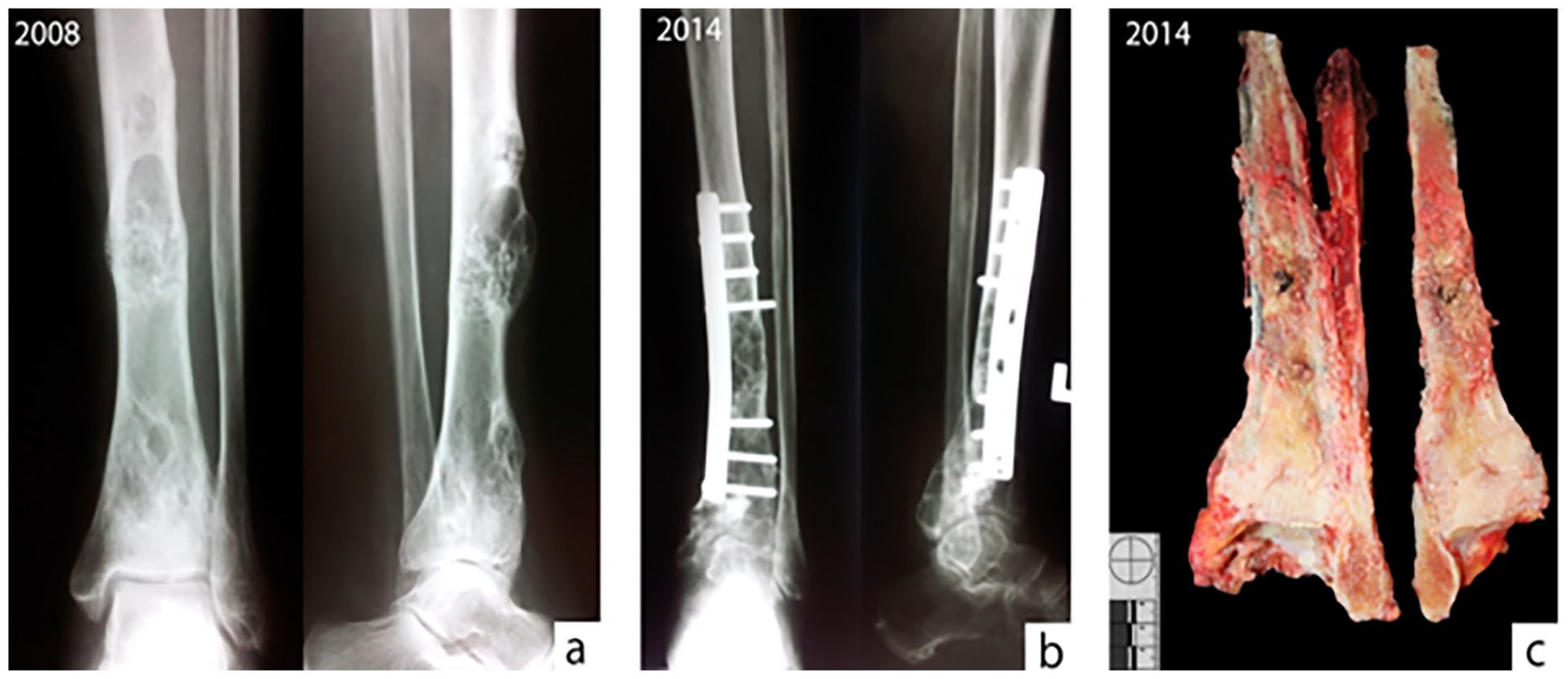
(a) Preoperative plain radiographs reveal multiple eccentric, predominantly lytic lesions of the tibia shaft with well-defined transition zones and solid periosteal reactions, without destruction, (b) postoperative radiographs 6 years after show distal extension of osteolytic lesions to the epiphysis and subarticular zone, and (c) gross appearance of the tumorous lesion after amputation. The tumor presents as a yellowish-gray, cheesy and gritty mass with areas of necrosis and hemorrhage that involve the cortex and expand into the medulla. The lesion reaches the distal portion of the tibia.
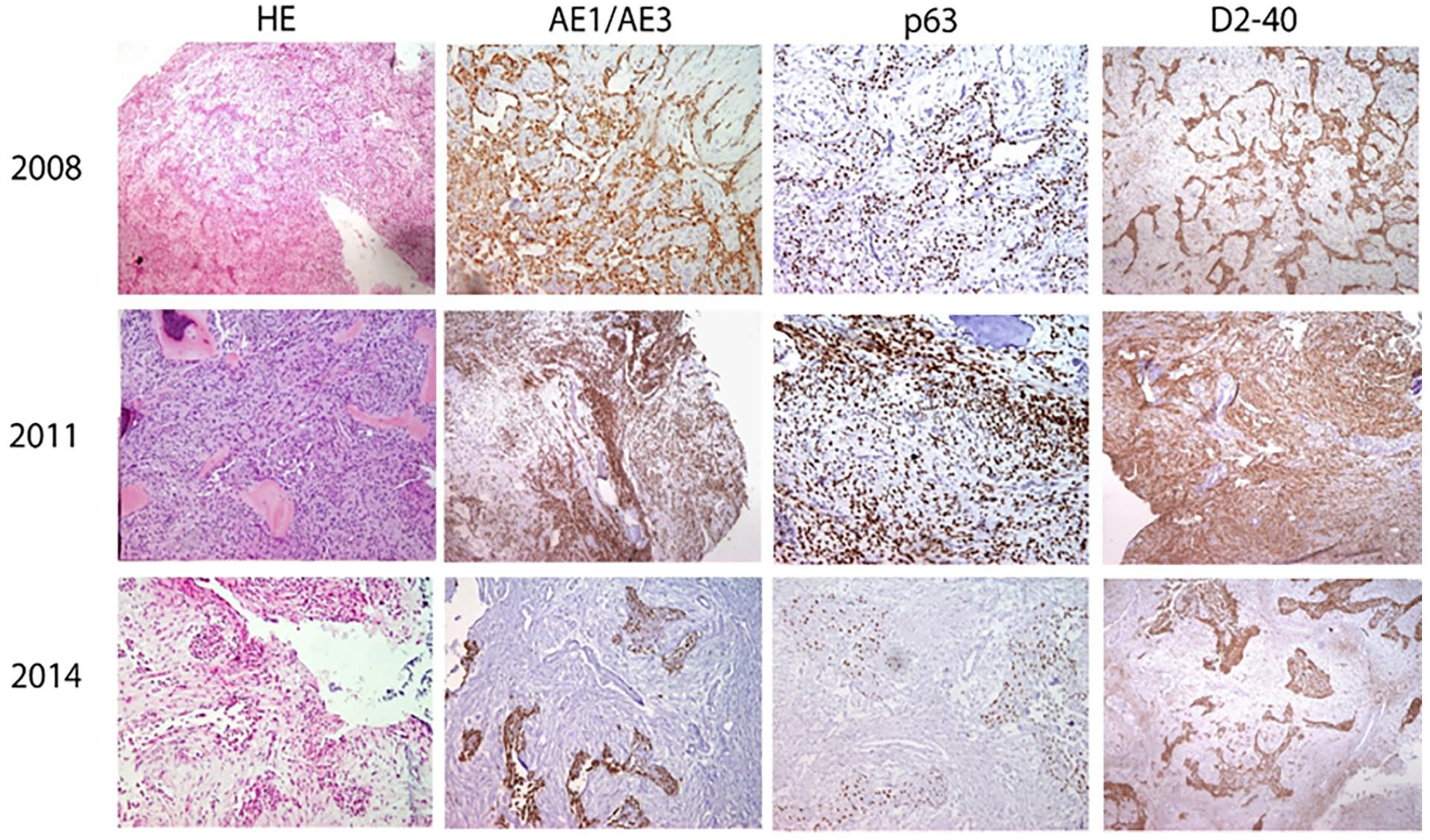
Histological and immunohistochemical (pancytokeratin (AE1/AE3), p63, podoplanin (D2-40)) results of sections of the same lesion taken at different time intervals (magnification 100×). Classical adamantinoma histomorphology was detected in the primary and last specimens, while the same lesion in the first recurrence showed fibrous-dysplasia like features. Independently of morphology, all sections were positive for adamantinoma markers.

(a) Fibrous dysplasia-like features of the tumor section from the first recurrence and (b) osteofibrous dysplasia-like features (with clear osteoblastic rimming) detected in the tumor section from the resected tumor (magnification 200×).
After the amputation of the leg, the specimen demonstrated the gross appearance of the tumorous lesion that involves the cortex and expands into the medulla, reaching the distal portion of the tibia (Figure 1c), while the histological analysis performed at our institution confirmed the diagnosis of AD, mostly of the classic subtype with basaloid epithelial cells, but also featuring some OFD-like and no FD-like zones (Figures 2 and 3b).
Since the aim of this study was to investigate the 6-year histological progression of the lesion, immunohistochemistry was performed on sections from all 3 biopsies. Based on literature data on the expression of different immunomarkers in AD, osteofibrous-like AD, OFD, and FD, histological sections were stained for pancytokeratin, vimentin, p63 and podoplanin20 -22 (Table 1). Immunohistochemistry confirmed the diagnosis of AD in all 3 biopsies. The results of the analysis are shown in Table 2 and Figure 2.
Antigen expression in stromal and epithelial components of adamantinoma, osteofibrous dysplasia, and fibrous dysplasia.

Abbreviations: ALB, adamantinoma (of long bones); ALB-OFD, osteofibrous dysplasia like adamantinoma; FD, fibrous dysplasia; na, non applicable (not tested on this group of lesions); OFD, osteofibrous dysplasia.
Antigen expression in the sections of the same lesion in the patient based on the diagnosis and time-period.
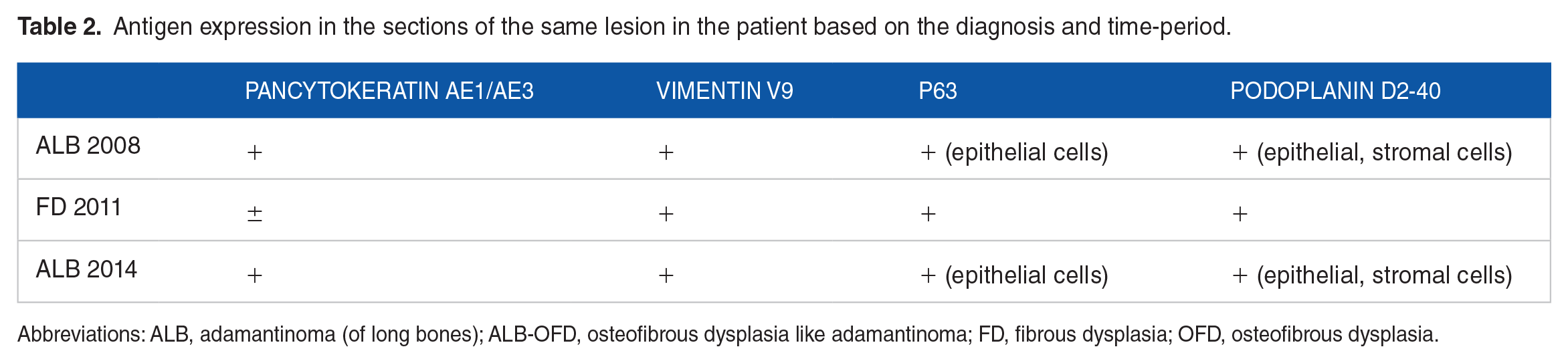
Abbreviations: ALB, adamantinoma (of long bones); ALB-OFD, osteofibrous dysplasia like adamantinoma; FD, fibrous dysplasia; OFD, osteofibrous dysplasia.
We evaluated the patient for 9 years after surgery, and there were no complaints. The patient could walk normally without aid since last year when she passed away, but not as a result of a bone tumor.
Discussion
The incidence of recurrences of AD is frequent, regardless of the surgical strategy, 22 while metastases have been reported in 15% to 30% of cases and included only the epithelial component of the tumor. 2 In the present case, we were able to histologically follow and compare the appearance of AD in a patient who developed 2 recurrences in the tibia over the course of 6 years. The first biopsy and the last examination showed features attributable to the classic subtype of AD, without any changes in immunoreactivity of the markers used. The only exception was the FD-like section of the tumor analyzed in the first recurrence (3 years after the first biopsy), which showed stronger podoplanin positivity than the samples from first and last examination, in contrast to cytokeratin, which was variably positive on spindle cells, as previously observed in the work of Kashima et al 11 In the last biopsy, there were no FD-like sections, but only small patches of scattered OFD-like areas (fibrous stroma with bone trabecule rimmed with active osteoblasts) were found. Although some authors have suggested that the predominance of spindle cells is a step forward to the last extreme end of the sarcomatoid dedifferentiation of adamantinoma,4,5 it seems that in our case the spindle-cell pattern did not progress (the final lesion maintained the same morphology as 6 years prior). It is possible that the FD-like section was just a transient form of the tumor cell population. Considering the differences in epithelial components of the tumor, it would be interesting to study a larger AD sample and observe the possible progression and regression of different epithelial variants and stromal components, and their relationship with diagnosis and clinical course. In our case, the patient had a long survival, which aligns with research showing that OFD- like AD has a better outcome than classic AD. 23
Adamantinoma is today recognized as an osteofibrous neoplasm, which, despite the possible de novo occurrence, is often seen as a continuum of OFD. Contrary to a series of earlier studies that linked FD to AD,24 -27 it is now generally accepted that FD remains a distinct entity and cannot be a precursor of AD or OFD.11,28 However, a case study by Nouri et al 5 has suggested that AD could develop from a malignant transformation of FD. They based their conclusion on the mesenchymal-to-epithelial transition theory and the simultaneous detection of FD and dedifferentiated adamantinoma in opposite legs of the same patient.
Since our patient presented with AD of the left tibia and the diagnosis of FD in the same leg in her medical history, it was hypothesized that this would be the first case demonstrating the co-existence and possible developmental relationship between the 2 lesions speculated before. However, immunohistochemistry proved that the FD, which was morphologically diagnosed in the first recurrence, was just a FD-like portion of a pre-existing AD, showing strong positivity for the AD epithelial markers (Table 1). The spindle cells were proven not to be fibroblastic stromal cells but fibroblast-like epithelial cells, confirming how the epithelial component can be easily hindered in cases with a marked spindle-cell pattern. 26 This confirmed the difficulties encountered when diagnosing AD, especially when relying on single biopsies, atypical clinical features, or medical history, as it was pointed out in the earlier works.29 -31 Since the spindle cell variant of the tumor produces the highest number of misdiagnoses, it is possible that some older cases describing FD lesions within AD were also just spindle-cell variants of the tumor not subjected to immunohistochemistry.24 -27 On the other hand, Nouri et al 5 presented a case of immunohistochemically proven FD in a patient with AD. However, since bilateral occurrence of AD is quite rare 32 and in this patient the FD affected a different extremity, their finding may be just an incidental finding and not proof of FD progression into AD.
Our study showed that the classical type of AD could present regions of spindle cells mimicking FD, and not only OFD. This supports the suggestion to rely on multiple biopsies for diagnosing AD, since a single biopsy with one tumor’s component predominating over the other can hinder the accurate diagnosis. Any (osteo)fibrous lesion accompanied by symptoms that can be linked to AD should be evaluated immunohistochemically, despite the apparently clear histology, 33 and not only with epithelial markers but also with podoplanin, since cytokeratins and EMA can show variable staining in the spindle cells of FD-like AD. This is supported by the fact that AD is a biphasic tumors that contain epithelial and mesenchymal component, such as synovial sarcoma, mesothelioma and ameloblastoma that also express podoplanin. 11
Footnotes
Acknowledgements
There are no acknowledgments.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AP and NJ: wrote the paper.
VŠ: concept and design of the paper, critical treatment of the article.
ASV: reviewed the literature and text.
PP: reviewed the text, collected clinical data.
All authors: final approval of the manuscript.
Ethical approval
Ethical approval was not required for this case report.
Consent
The authors certify that they have obtained all appropriate written informed consent forms from the patient’s family for the publication of this case report and accompanying images. The family understands that patient’s name and initials will not be published.
