Abstract
Background:
Tumor budding (TB) has been defined as an independent prognostic factor in many carcinomas like colon adenocarcinoma, but its prognostic impact on gastric cancer patients remains not well established. In the present study, we aimed to highlight the correlation of tumor budding with clinicopathological features and predict its survival outcomes in gastric cancer patients for the first time in the Moroccan population.
Methods:
This study was conducted on 83 patients who underwent surgery for gastric adenocarcinoma from 2014 to 2020. The patient’s clinico-pathological characteristics were obtained from the pathological and clinical records of each patient. Tumor budding was assessed on HES slides, according to the 2016 International Tumor Budding Consensus Conference criteria. The association of tumor budding grades with categorical and continuous variables were respectively assessed by the χ2-test and the unpaired t-test. Survival analysis was performed by the Kaplan-Meier method, the log-rank test.
Results:
Patients consisted of 65.1% of men and 34.9% of women with a median age of 61.2 years. Histologically, the majority of the tumors were adenocarcinoma (65.1%). Among all cases, 18.1% were classified as Bud1 (15/83), (27/83) 32.5% as Bud 2, and 49.4% (41/83) as Bud 3 grades. High-grade tumor budding (BUD 3) was found to be significantly associated with special clinicopathological features including older age (P = .02), unradical resection (R1/R2) (P = .03), and the presence of vascular invasion (P = .05), and perineural invasion (P = .04). Furthermore, tumors with high-grade tumor budding were significantly associated with a low rate of resected lymph nodes (P = .04) and advanced TNM stage (P = .02). Among all stages, high-grade tumor budding was correlated with shorter overall survival in univariate and multivariate analysis (P = .04). Patients with high-tumor budding had worse relapse-free survival compared with patients with low-tumor budding grade (P = .01).
Conclusion:
According to our study, the high-tumor budding grade was correlated with unfavorable clinicopathological features and poorer survival. The present study findings suggest that tumor budding should be considered in the treatment and prognosis of gastric cancer patients.
Introduction
Gastric cancer is a real public health problem. Indeed, it’s the fifth most common cancer and the third leading cause of cancer-related deaths worldwide. 1 In Morocco, gastric cancer is the third cause of death and ranks seventh by incidence, according to the updated GLOBOCAN 2020 data. 1 Currently, the depth of tumor invasion, lymph node involvement, and the presence of distant metastases are well recognized as classical prognostic factors in GC, which makes the TNM classification the robust method to predict the prognosis of GC patients.2,3 However, the advances in diagnosis and treatment, clinical outcomes, and patient survival still vary remarkably among GC patients. Furthermore, many early-stage gastric cancer patients still die due to cancer. 4 For this reason, additional molecular and histological factors are needed to precisely estimate patient outcomes and to define the therapeutic option according to the specific characteristics of each patient. Tumor budding is one of the parameters that need to be introduced in the management of gastric cancer since it has been defined as an independent prognostic factor in gastrointestinal carcinomas like colon cancer. 5
Tumor budding is defined as the presence of single tumor cells or clusters of up to 4 cells at the tumor invasive front (peritumoral budding, PTB) or the tumor center (intratumoral budding, ITB).6 -8 Many recent studies, have demonstrated that high tumor budding correlates with unfavorable clinicopathological features (stage, lymph node metastases, distant metastases, and perineural invasion) in colorectal cancer.5,7 Moreover, TB is recognized as an independent prognostic factor in CRC by the International Tumor Budding Consensus Conference (ITBCC), and is officially defined as a prognostic hallmark by the Union for International Cancer Control (UICC).9 -11
Currently, the prognostic value of tumor budding in gastric cancer has not been well established. Furthermore, few studies have reported the correlation between the tumor budding grades and the clinicopathological features of tumors, especially the prognostic features. In Morocco, there is an essential need to improve the survival of GC patients, so it is very important to choose the best tools for risk stratification and make a suitable therapy decision after the surgery, and TB may be one of these tools. This study aimed to highlight the association between tumor budding with, clinic-pathological features and predict its survival outcomes in gastric cancer patients for the first time in Morocco and MENA region.
Methods
Ethics
The requirement of obtaining ethical approval was waived off by Hassan II University Hospital Ethics Committee of FEZ.
Patients
This study was a retrospective cohort study in a single institution including 83 patients with gastric carcinoma who underwent gastrectomy between the years 2014 and 2020 at the University Hospital Center of Fez, Morocco. Patients with incomplete clinical and survival data were excluded from the study. Clinicopathological data for each patient, including sex, age at diagnosis, tumor localization, tumor differentiation, histological subtype, depth of invasion, presence of vascular invasion, presence of perineural invasion, number of resected lymph nodes, number of lymph nodes with metastases and presence of distal metastases, were obtained from the pathologic database of the laboratory of anatomic pathology at the University Hospital Center of Fez, Morocco.
Histology
Tumors obtained from resected specimens at the department of surgery were transferred to the department of anatomic pathology for histological study. Tumors were routinely formalin-fixed in 10% neutral formalin for 24 hours and paraffin-embedded (FFPE). Deparaffinized sections were stained with hematoxylin and eosin (H&E). The HES slides were reviewed by experienced pathologist for pathological evaluation. The pTNM stage was determined according to the eighth edition of the UICC guidelines (2017). Histopathological subtypes were classified according to the Lauren and 2017 WHO classifications, and histological grading according to the 2010 WHO classification. 12
Tumor budding
Tumor budding was evaluated for each patient on HES-stained slides by experienced pathologist, according to the International Tumor Budding Consensus Conference (ITBCC) 2016 recommendations. 7 Firstly, a single hotspot area was defined at the invasive front using microscopy at 20×. Secondly, Bd was counted in the selected area (20 objectives).
TB was scored Bd1 (1-4 buds), Bd2 (5-9 buds) or Bd3 (⩾10 buds) (Figure 1A–C). Cases were divided into 2 groups, low budding group (<10 buds: Bud1 + Bud2), and high budding group (Bud3 ⩾ 10 buds) as reported in a Germanien study. 8

Tumor budding grades. Gastric Adenocarcinoma: (A) Bud0 (HESX100), (B) Bud2 (HESX100), and (C) Bud3 (HESX40).
The assessment of tumor budding might be challenging in the case of diffuse versus intestinal gastric cancer. However, we only counted tumor buds that escaped from the tumor mass.
Follow up and survival
All patients were followed-up at the department of Surgery and Oncology. The outcome of this study was overall survival (OS) and relapse-free survival (RFS). OS was defined as the period from the date of the first diagnosis to the day of death due to gastric cancer, or the date of the last available follow-up visit. Death was scored as an event. RFS was measured from the date of the first diagnosis to the date of the first distant or local disease recurrence or last follow-up/death.
Statistical analysis
Data analysis was performed using SPSS statistical software (Version 21; SPSS Inc., Chicago, IL). The correlation of tumor budding grades and several parameters was analyzed using the Chi-square test or Fisher’s exact test. Tests were statistically significant when P < .05. Survival curves were estimated by the Kaplan-Meier method and compared using the log-rank test and prognostic markers by COX regression. Differences were considered to be significant if the P-value was <.05.
Results
Clinicopathological features and tumor budding grades
In this study, we analyzed 83 gastric cancer patients, with 54 men (65.1%) and 29 women (34.9%). The mean age at diagnosis was 61.3 years (range: 21-87). Distal gastric cancer was the most common tumor location (51.2%; 42/83). Regarding histological types, 65.1% of the tumors were classified as adenocarcinoma. Tumors were mostly moderately and poorly differentiated (46.7% and 36.7%, respectively). According to Lauren’s classification, 47 (63.5%) of patients had intestinal tumors, 21 (28.4%) had diffuse, and 8 (8.1%) had mixed tumors. Only 23 (27.7%) and 24 (28.9%) of the patients presented perineural and vascular invasion respectively. The mean of harvested lymph nodes was 23.1 (range: 7-57). The majority of the patients had more than 15 lymph nodes (90.4%; n = 75), and 54.1% (n = 45) had metastasing adjacent lymph nodes. In this cohort, surgery was decided for 52 (65.0%) patients as the first therapy and periadjuvant chemotherapy for 28 (35.0%) patients. All data are summarized in Table 1.
Baseline of clinicopathological features in gastric cancer.

Regarding tumor budding, 15 (18.1%) of 83 GC patients had low (BUd1), 27 (32.5%) had intermediate (Bud2), and 41 (49.4%) had high TB (Bud3), Figure 1. After the combination of tumors with bud1 and those with bud2, 42 (50.6%) patients were classified as a low bud and 41 (49.4%) as high bud (Table 1).
Clinicopathological features and tumor budding grades
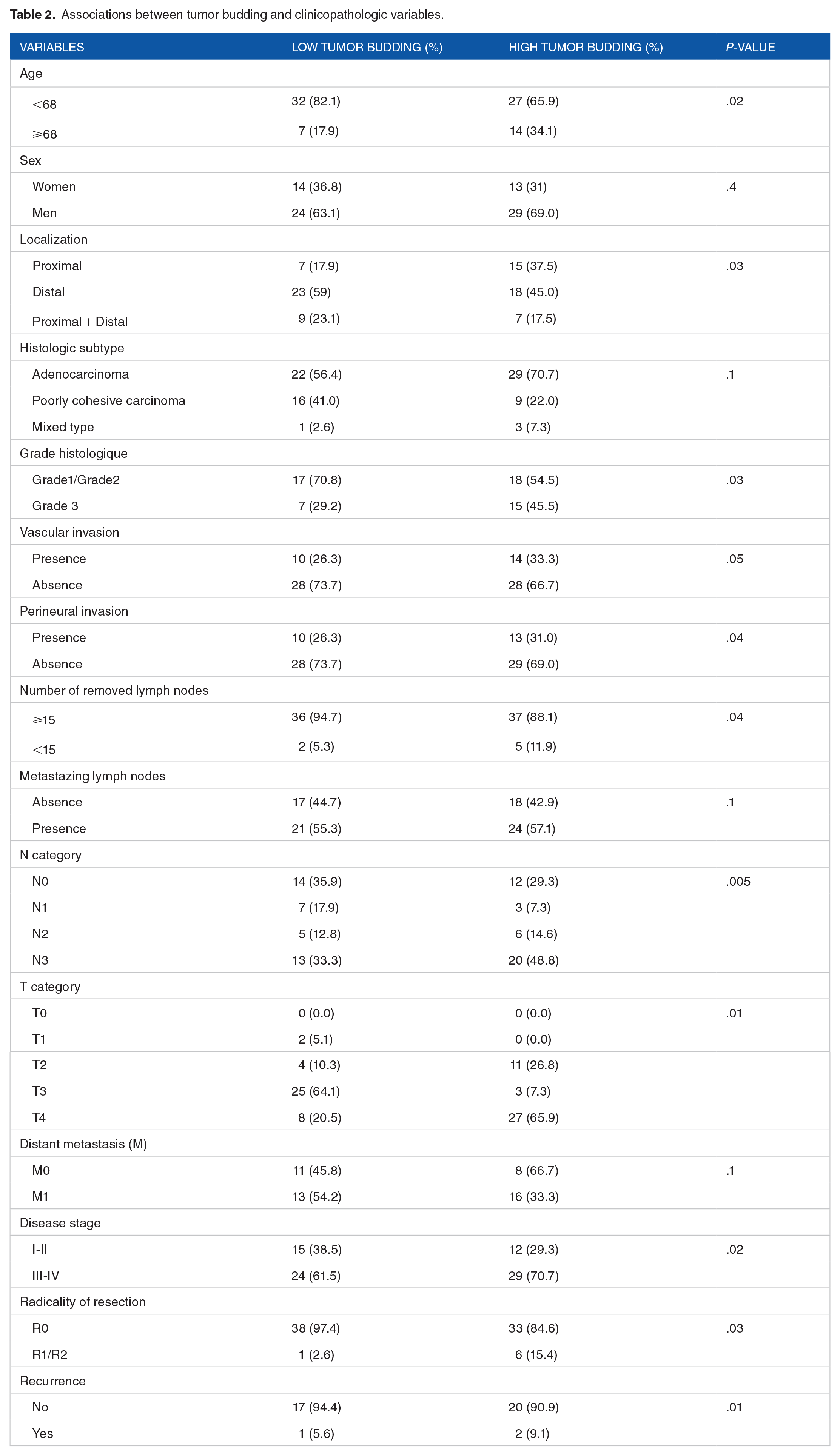
Table 2 displays the correlation between clinicopathological features and the TB grades for GC patients. In our study, the patients with high-budding were significantly older (aged ⩾68 years) than those with low budding (P = .02). Tumor localization was significantly associated with high-tumor budding tumors (P = .03). A significant difference was detected between the histopathological gradings and tumor budding groups. We found a significant correlation between the poorly differentiated tumors and the high-tumor budding grade (P = .03). Regarding invasive features of the tumor, patients with high-tumor budding grade showed the highest rates of perineural and vascular invasion (P = .04 and .05, respectively) compared to the other group. In addition, 88.1% of those patients had more than 15 removed lymph nodes, compared with 94.7% and 100% in patients with low-tumor budding, respectively. According to the pTNM classification, patients with high tumor-budding grade had more frequent N3 and T4 categories (P = .005 and .01, respectively) and were diagnosed in the advanced TNM stage (P = .02). Unradical resection (R1/R2) was found to significantly correlate with the high tumor budding grade (P = .03). Furthermore, the patients with high tumor budding exhibited a high rate of recurrence (P = .01). In contrast, no correlation was observed in the tumor budding grades according to sex, histological types, and distant metastasis.
Associations between tumor budding and clinicopathologic variables.
Survival analysis
The mean follow-up time was 25.9 months (range: 0-112 months) for the entire GC cohort. The mean overall survival time was 55.7 months (95% CI; 43.4-67.8), with 39 (49%) cases of death. Patient prognosis significantly depended on the Lauren classification; T categories; TNM stage; the presence of vascular and perineural invasion; and positive lymph node (Table 3).
Univariable and multivariable analysis of tumor budding and overall survival in patients with gastric cancer.
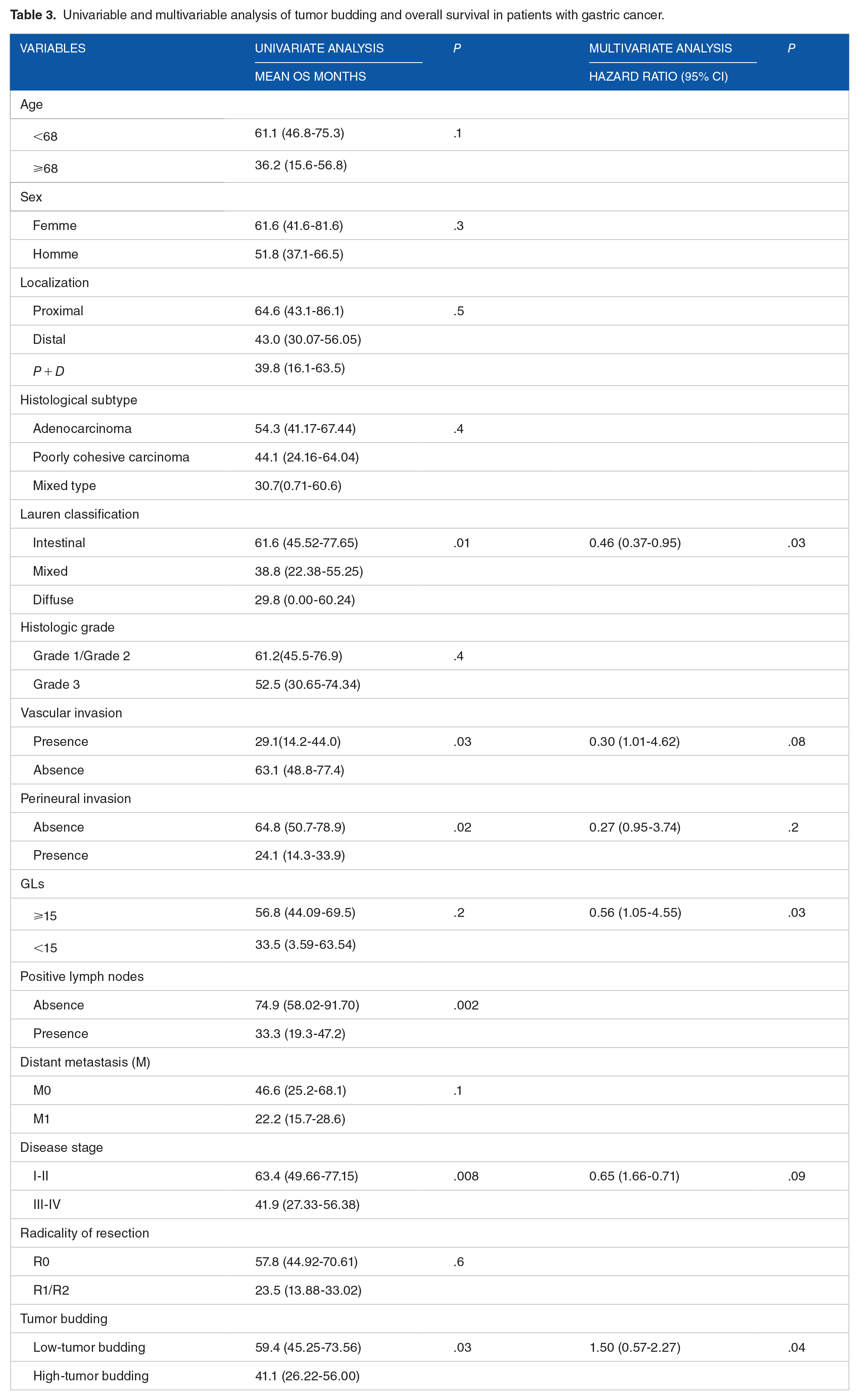
According to tumor-budding grades, Kaplan-Meier survival curves showed that the patients without tumor budding had the best prognosis (longer OS) (Figure 2). However, those with high-tumor budding had the worse prognosis. Indeed, the mean OS was 59.4 months in the low-tumor budding group, and 41.1 months in the high-tumor budding group (P = .03) (Table 3). In multivariate analysis, tumor budding was found to be an independent prognostic factor (P = .04) (Table 3).
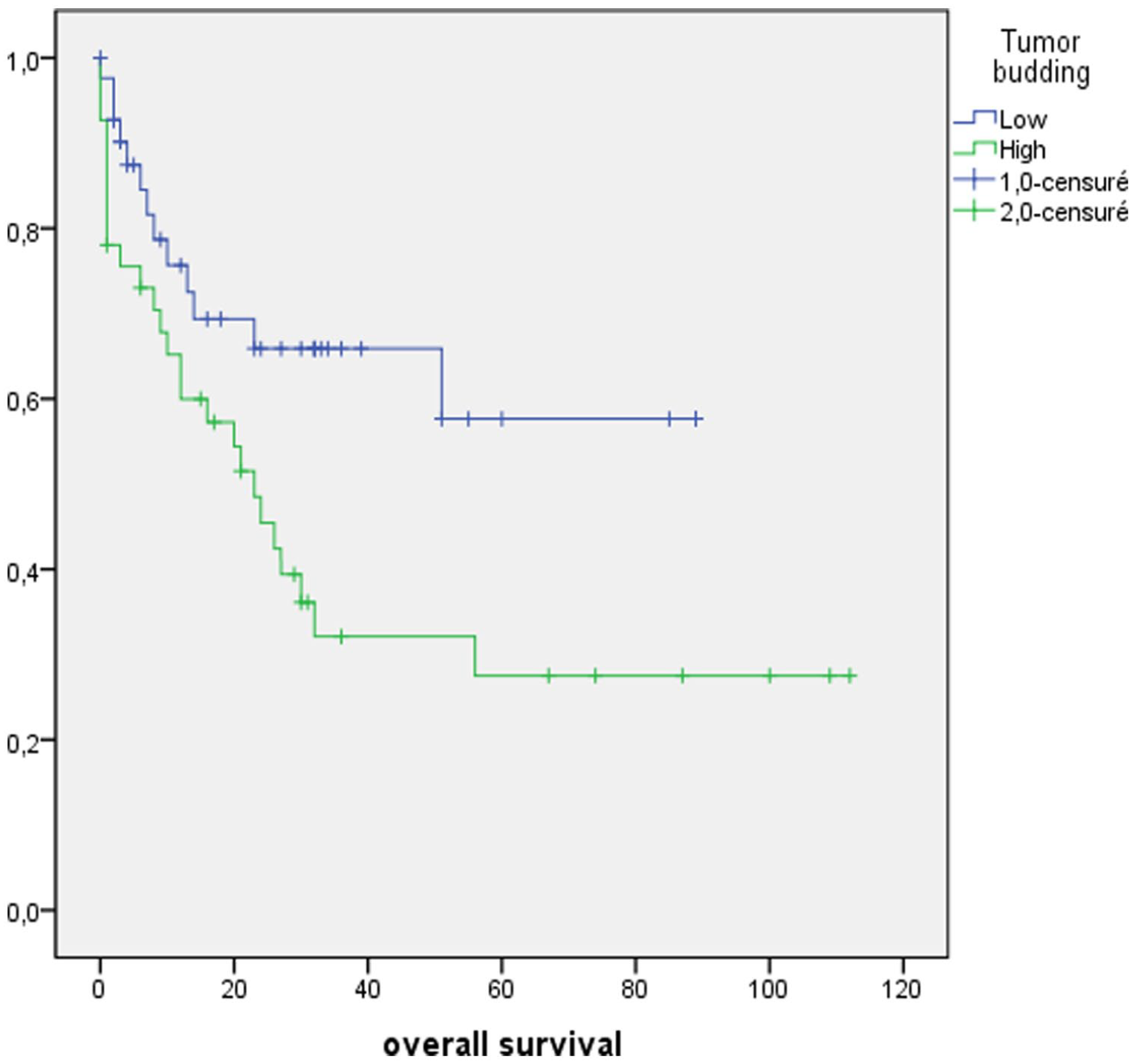
Kaplan-Meier curves demonstrating associations between tumor budding and overall survival (P = .03).
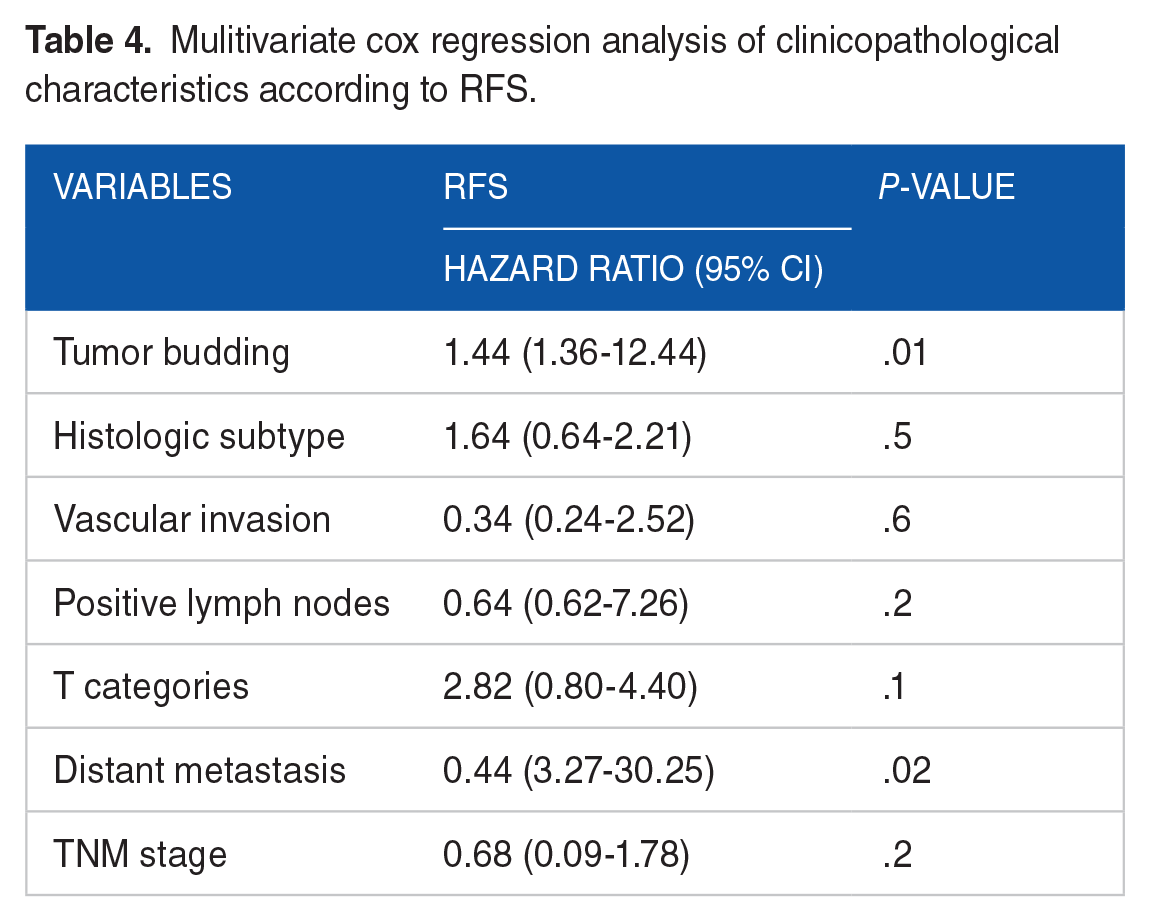
The mean relapse-free survival in our cohort was 36.1 months (range: 0-112 months). From 83 cases, recurrence was observed in 23 patients (27.7%). The most common recurrence sites were lung (n = 10; 43.5%), followed by liver (n = 7; 30.4%), and local recurrence (n = 4; 17.4%). The frequency of other sites (lymph nodes; os; peritoneum; gall bladder) was 4.3% (n = 1). The relapse-free survival was higher in the group low-tumor budding group (75.1) compared to the high-tumor budding group (60.4) with a difference close to significance (P = .08) (Figure 3). In multivariate analysis, the high-tumor budding grade was the independent poor prognostic factor for RFS in gastric cancer (Table 4).

Kaplan-Meier curves demonstrating associations between tumor budding grades and relapse-free survival (P = .08).
Mulitivariate cox regression analysis of clinicopathological characteristics according to RFS.
Discussion
Tumor budding is actually considered the histological basis for tumor invasion and metastasis since it is thought that the budding cells migrate through the extracellular matrix, infiltrate the lymphovascular constructs, and form metastatic cancer cell colonies in the lymph nodes and at distance locations.13,14 It prognostic impact has been evaluated in a large variety of malignant carcinomas from different organs like colorectal; esophageal; pancreatic; lung; gingival buccal complex squamous cell carcinoma; head and neck and breast cancer.15 -21 In gastric cancer, few studies have described the tumor budding and it’s implication on patient outcomes,22 -25 using different scoring methods.3,23,26 The current study is the one to highlight the relationship between tumor budding grades and the clinic-pathological features of gastric tumors, and to analyze the prognostic impact of this histologic parameter in patients with gastric cancer in the Moroccan population and MENA region.
In our study, the frequency of high tumor budding was 49.4%. This prevalence is consistent with findings noted in prior studies using the ITTBC 2016 scoring system.11,24 A lower frequency was documented by other studies, which reported that high tumor budding might be found in approximately 40% of gastric cancers.27,28
To date, the majority of studies conducted on tumor budding in gastric cancer have displayed that high tumor budding is related to various specific clinic-pathological features that indicated poor prognosis (poorly differentiated grade, vascular and perineural infiltration, positive lymph node, advanced T stage, and advanced TNM stage) despite the methodical variety of TB scoring.3,22,26,29,30 In this cohort, high tumor budding was correlated with older age. However, Dao et al 11 and Ulase et al 24 didn’t find any significant association between age and tumor budding. Che et al 30 showed the same result in a cohort from China. A significant correlation was observed between the distal tumor site and high tumor budding grade (P = .03). Similar results have been reported by Ulase et al. 24 Regarding to histological features, the results generated in the current study, support the findings that high tumor budding grade is significantly associated with poorly differentiated tumors in the previous studies.11,22,24,29 Similarly, we documented in our series that high tumor budding was significantly associated with invasive features (perineural and vascular invasion), as noted previously by Gabbert et al, 29 and Olsen et al. 22 Additionally, the results of our study demonstrated a significant relationship between T and N categories, and TNM stage with tumor budding grades, as reported in the literature. 24 In the present study, 15.4% of patients with high tumor budding exhibited non radical resection (P = .03). The findings of this study support those reported previously by Kemi et al 3 and Dao et al. 11
To date, 9 studies have been performed on tumor budding and prognosis in gastric cancer. A study from Germany, investigated by Gabbert et al 29 in 1992 years, was the first to report the association between tumor budding and overall survival. A Japanese study (n = 153) suggested that high budding is associated with poor prognosis (HR, 1.61; 95% CI, 1.12-2.41 in univariate analysis), but tumor budding was not an independent prognostic factor in multivariate analysis. 26 In addition, a Chinese study demonstrated that high tumor budding is associated with poor overall survival (HR, 1.57; 95% CI, 1.04-2.35, P < .001) in univariate and multivariate analysis. 30 Recently (2019), Kemi et al 3 found that patients with high tumor budding had significantly worse survival (HR, 1.46; 95% CI, 1.22-1.75) compared with those with low tumor budding in both univariate and multivariate analysis. The same results were reported in 2020, by Dao et al 11 and Ulase et al. 24 In the present study, we also confirmed the findings of the previous studies, and we noted that high tumor budding grade is an independent prognostic factor in gastric cancer (P = .04). Our results are the first in Morocco and MENA region.
In addition, the present study findings showed that high tumor budding grade had a prognostic significance for RFS in multivariate analyses (P = .01). Similar results were reported recently by Dao et al. 11 A Danish study (n = 52) including only patients with intestinal histology reported a lower RFS in the high-budding group, but the result was not statistically significant in multivariate analysis (P = .08). 22
There were some strengths and limitations to the present study. First is the size of the study cohort. Indeed, the number of cases included in our study is relatively small, in comparison with some previous reports since our country is not an endemic area for gastric cancer. Second, our study represents a retrospective single institution, which can limit generalizing our results to the overall population of our country. Third, it has been demonstrated that high-grade tumor budding is significantly associated with histological subtype. 3 However, we were unable to produce similar results. It’s could be likely attributable to the size and the characteristics of our sample. Above all, our study results are the first in Morocco and MENA region.
Conclusion
In summary, the findings of the present study show that high-tumor budding grade is correlated with unfavorable clinicopathological features, and it’s an independent prognostic factor in gastric adenocarcinoma. These results suggest that tumor budding status should be mentioned in the histopathological reports of patients with gastric adenocarcinoma.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
