Abstract
Background:
Phosphaturic mesenchymal tumor (PMT) is a very rare tumor of bone and soft tissue that has no specific clinical manifestations. Here we present 2 cases of PMT in the right thigh, including comparatively adequate immunohistochemistry.
Case Presentation:
We described 2 cases of PMT in the right thigh with manifestations of hypophosphatemia. PET-CT examination showed that both patients had lesions with increased expression of somatostatin receptors in the right thigh. Bland cells and dirty calcified stroma were exhibited under the microscope. And immunohistochemical detection of FGF-23 was positive.
Conclusions:
PMT is a very uncommon tumor for which diagnosis and treatment are often delayed. Considering the importance of surgery for the treatment of this disease, a full understanding of its clinicopathological features will facilitate the diagnosis of this disease.
Introduction
Tumor-induced osteomalacia (TIO), also known as oncogenic osteomalacia, is a rare paraneoplastic syndrome. Its biochemical characteristics include hyperphosphaturia, hypophosphatemia and significant reduction of 1,25-dihydroxyvitamin D. This disease is caused by tumors that secrete fibroblast growth factor 23 (FGF23), especially phosphaturic mesenchymal tumor (PMT).1 -5 Patients often show nonspecific symptoms such as fatigue, bone pain, fracture, and muscle weakness.5,6 Due to the nonspecific clinical manifestations of the disease, the lack of doctors’ understanding of the disease and the difficulty in tumor location, the diagnosis and treatment of this disease are often delayed.7,8 Here, we report 2 cases of TIO caused by PMT in the right thigh.
Case presentation
Case 1
The first case was a 76-year-old man who was hospitalized with 11 years of bilateral lower extremity mobility limitation and 5 years of recurrent multiple fractures. In 2010, the patient began to suffer from fatigue of both legs after walking, inability to step on both legs, decreased height, hypophosphatemia, and other discomforts. Imaging in another hospital found local bone destruction in the upper segment of the right femur, and he underwent surgical resection of the lesion and right femoral head replacement. Postoperative pathological examination showed bone tissue degeneration, hyperemia, and a small amount of fibrosis. No other characteristic lesions were found. The patient’s condition gradually worsened after operation, with decreased activity and multiple rib fractures. In 2016, octreotide imaging was performed in a hospital in Beijing, which suggested a right femoral lesion. Surgery was performed again, and postoperative pathological examination revealed a phosphaturic mesenchymal tumor in the right femur.
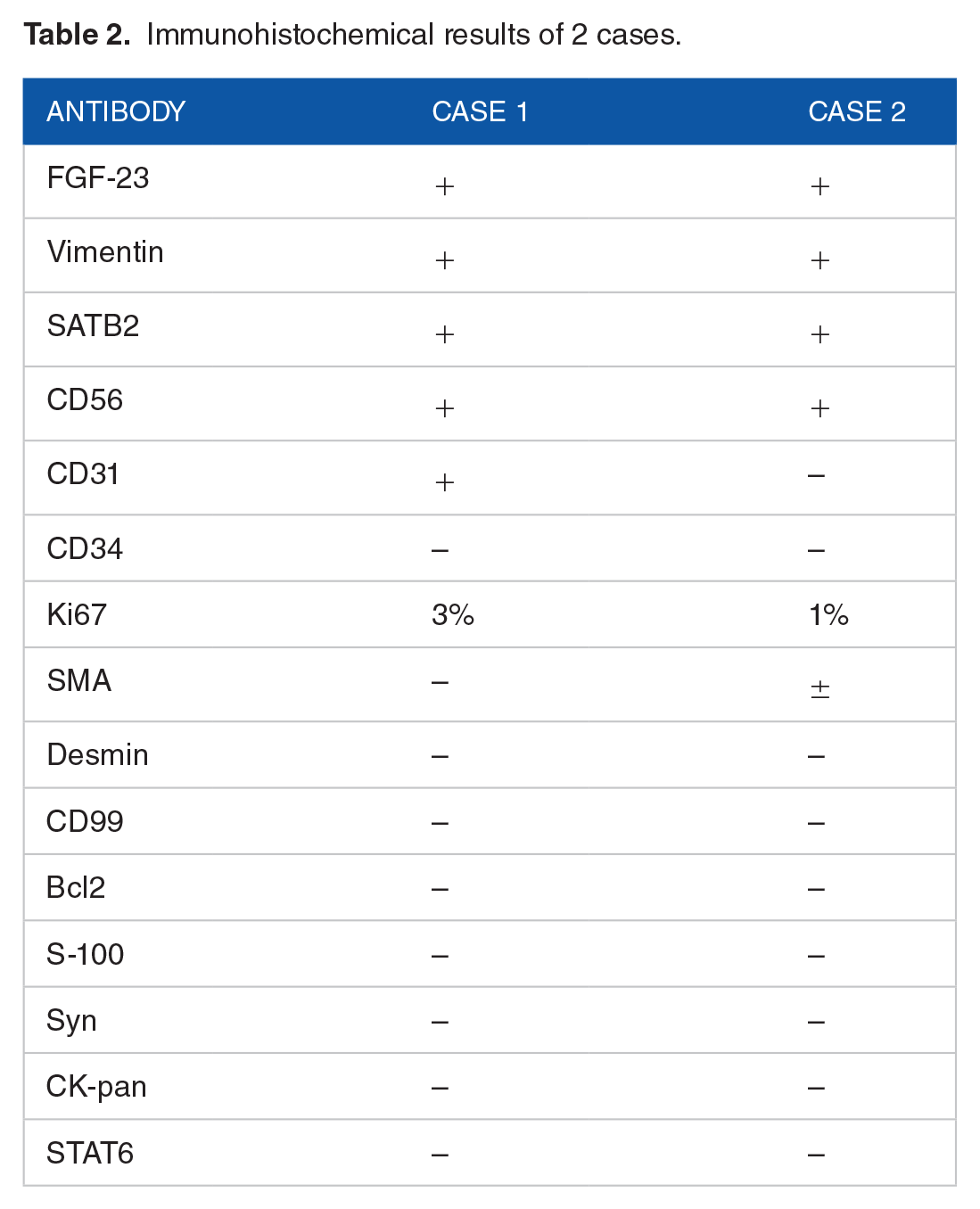
Postoperatively, the patient underwent outpatient reexamination, and the serum phosphorus was lower than before, so he was admitted to our hospital for treatment. The specific values of the patients’ preoperative blood phosphorus, blood calcium, blood magnesium and other indicators are shown in Table 1. Physical examination revealed no obvious abnormality. In addition, PET-CT examination showed soft tissue nodules in front of the right femur, about 0.7 cm in diameter, with increased expression of somatostatin receptors: it is likely to be a phosphaturic mesenchymal tumor (Figure 1). Then, surgical resection was performed. The histology of the postoperative specimens showed that the tumor was composed of bland short spindle cells, smudgy/grungy basophilic matrix, mature adipose tissue, hemorrhage, and focal ossification (Figure 2). Immunohistochemical staining showed that vimentin, SATB2, CD56 and CD31 were positive, while CD34, SMA, desmin, CD99, Bcl-2, S-100, Syn, CK-pan and STAT6 were negative. The proliferation index Ki-67 was about 3% (Table 2). Compared with preoperative, the patient’s serum phosphorus showed an overall upward trend, although it still did not return to normal half a year after the operation.
Laboratory workup results.

Immunohistochemical results of 2 cases.

A PET-CT scan showed a lesion about 0.7 cm in diameter above the right thigh.

Representative images of histology and immunohistochemistry of case 1. (A and B) H&E (10×and 4×) highlighted bland spindle cells, smudgy/grungy basophilic matrix and hemorrhage. (C) H&E (10×) showed focal ossification. (D) Immunohistochemistry (20×) demonstrated positive staining for FGF-23 in tumour cells.
Case 2
The second case was a 32-year-old man who was admitted to hospital with generalized pain for more than a year. One year before admission, the man had no obvious inducement of foot pain, which was aggravated after weight-bearing activities and exercise, and then gradually developed upward to both lower limbs, bilateral hip joints, ribs, and shoulder blades. The degree of pain gradually increased, accompanied by walking, turning over and bending difficulties, and morning stiffness. No obvious abnormality was found in physical examination. In addition, serological examination showed that blood phosphorus was 0.59 mmol/L (reference value range: 0.86-1.78 mmol/L), alkaline phosphatase was 471.8 u/L (reference value range: 45.0-125.0 u/L), and blood calcium was in the normal range (Table 1). And PET-CT showed a slightly low-density nodule in the inner muscle of the right middle thigh, about 1.5 cm × 1.8 cm in size, with increased expression of somatostatin receptors: the possibility of phosphaturic mesenchymal tumor (Figure 3). Then, surgical excision of the lesion was performed. The histology of the postoperative specimens showed that the tumor was composed of bland short spindle cells. Smudgy/grungy basophilic matrix, hemangiopericytomatous-like blood vessels, a small number of thick walled vessels, a little adipose tissue, hemorrhage, focal ossification, multinucleated giant cells, and focal myxoid degeneration could be seen (Figure 4).Immunohistochemical staining showed positive for vimentin, SATB2, and CD56, but negative for CD31, CD34, desmin, CD99, Bcl-2, S-100, Syn, CK-pan, and STAT6. And there was some nonspecific staining for SMA. The proliferation index Ki-67 was about 1% (Table 2). Compared with preoperative, the patient’s serum phosphorus showed an overall upward trend and completely returned to normal levels on the second postoperative day.

A PET-CT scan showed a lesion about 1.5 cm × 1.8 cm in size above the right thigh.

Representative images of histology and immunohistochemistry of case 2. (A and B) H&E (20× and 10×) highlighted bland spindle cells, multinucleated giant cells and focal myxoid changes. (C) H&E (4×) showed hemangiopericytomatous-like blood vessels and smudgy/grungy basophilic matrix. (D) Immunohistochemistry (20×) demonstrated positive staining for FGF-23 in tumor cells.
Materials and Methods
Review of the literature
In addition to our own cases, we also collected 41 cases diagnosed with PMT from literature in the PUBMED database. The patients in this group ranged in age from 26 to 70 years old. The male to female ratio is approximately 1:1. Among them, 13 cases occurred in the head and neck, 19 cases were found in the extremities, and 9 cases were detected in the spine and ribs. As for biological behavior, the majority of cases presented with a benign course, and only 5 cases developed metastasis. Bland spindle cells and smudgy/grungy basophilic matrix are seen in most cases. Different immunohistochemical items were performed in each case, with 21 expressing vimentin and 13 expressing FGF-23. The positive expression of CD68, SMA, CD56, S-100, Bcl-2, FGFR1, CD31, CD34, SATB2, CD99, and D2-40 can be observed in tumor cells. The immunohistochemical features of the above cases are summarized in Table 3.
Immunohistochemical results of reported 41 cases.

Discussion
PMT is a rare FGF23-producing tumor of bone and soft tissue and is one of the rare causes of osteomalacia. 9 FGF-23, a circulating factor normally secreted by osteocytes and osteoblasts, can act on proximal renal tubular epithelial cells to reduce phosphate reabsorption and promote its excretion, resulting in phosphate consumption.10,11 This process may require the involvement of Kloth, which has been shown to convert canonical FGF receptors to FGF23-specific receptors.12,13
PMT often occurs in middle-aged and elderly people, and can appear in various soft tissues and bones, with no obvious gender differences.14,15 It is most common in the lower extremities, followed by the head and neck. 16 The onset of the disease is insidious, and fractures, bone pain and weakness are the main clinical manifestations, and some patients may also show decreased height.17,18 In children, rickets and growth retardation are often present. 11 Furthermore, lesions tend to be small, making tumor localization difficult, and diagnosis delayed. 19
Patients often have low blood phosphorus, high urinary phosphorus, and high serum alkaline phosphatase, while calcium metabolism is often basically normal, and blood phosphorus levels will mostly return to normal after surgery.18,20 Some patients may be in the compensated or early stage of the disease without the manifestation of hyperphosphaturia.14,21,22 In addition, it has been reported that FGF23 can be significantly elevated in people with TIO and returned to normal after tumor resection. 23 A study found that whole-body sampling of FGF23 was helpful for tumor localization. 24 The measurement of FGF23 is also useful for postoperative follow-up. 25
Both CT and MRI can be used to find the site of PMT. Radionuclide scans such as FDG-PET, Tc 99 m sestamibi scintigraphy, and 68 Ga-DOTATOC-PET/CT are very valuable in identifying PMT.26 -28
As for histopathology, Weidner and Santa Cruz classified PMT into 4 subtypes based on histology: mixed connective tissue tumor (PMTMCT), osteoblastoma-like tumor, non-ossifying fibroma-like tumor, and ossifying fibroma-like tumor. 29 Among them, PMTMCT was the subtype with the highest proportion. 14 Histological features include low to moderate amount of bland spindle cells, small nuclei, inconspicuous nucleoli, few mitotic figures, dirty calcified stroma, hemangiopericytoma-like vessels, osteoclast-like giant cells, fat, microcysts, hemorrhage, metaplastic bone and membranous ossification with incomplete margins, infiltrative growth, etc.14,30,31 Therefore, PMT needs to be differentiated from solitary fibrous tumors, soft tissue giant cell tumors, and osteosarcomas.
Immunohistochemistry is not necessary for the diagnosis of PMT. It has been reported that 22 of 22 PMTs in the jaws are FGF-23 positive. 32 However, immunostaining for FGF-23 of PMTs without TIO tends to be negative. 33 In addition, tumor cells can also express D2-40, SSTR2A, ERG and FLI1, CD56, SMA and SATB2, etc.15,32,34 -36 Although ultrastructurally similar to neuroendocrine tumor cells, PMTs rarely express markers such as S-100, NSE, syn, and CgA. 11 Houang et al believed that FGF23 and SSTR2A were particularly sensitive but not specific for the diagnosis of PMT. 35
FN1-FGFR1 fusions can be detected in most PMTs, and FN1-FGF1 fusions can be detected in a minority of cases. Also, the 2 fusions appear to be mutually exclusive.37,38 It has been reported that the detection of FGFR1 by immunohistochemistry may be helpful in the diagnosis of PMT. Although FGFR1 is a known FGF23 receptor and FGFR1 signaling increases the expression of FGF23 in the autocrine/paracrine loop, FGF23 serum levels were independent of FGFR1 membrane expression (strong staining). This suggests that there may be important factors other than FGFR1 involved in the tumor biology of FGF23-overexpressing PMTs. 39 RT-PCR of FGF23 is a sensitive method for the diagnosis of PMT, but it should be noted that it is not uncommon to detect FGF-23 mRNA expression in aneurysmal bone cysts and chondromyxoid fibroma. The diagnosis of PMT requires a close combination of clinical and imaging. 40 Furthermore, the expression level of FGF23 mRNA was different in PMT with TIO and PMT without TIO, and FGF23 mRNA would be higher in PMT with TIO. 33 Compared with RT-PCR, chromogenic in situ hybridization (CISH) can reduce the occurrence of false positives to a certain extent. 41
Most PMTs are benign tumors, and a few malignant cases have been reported.42 -45 It is currently believed that changes in tumor size, morphology, and FGF-23 levels are biomarkers for malignant transformation of tumors. 43 Complete surgical treatment with negative margins is the most preferred treatment, which can significantly improve the patient’s symptoms.29,46,47 Persistence of symptoms may be a sign of tumor recurrence or incomplete resection. Radiation therapy is an option for unresectable or incompletely resected tumors. 5 It is said that oral supplementation with phosphate and 1-25-dihydroxyvitamin D may help relieve symptoms in patients with unresectable or poorly located tumors. 48 However, drug therapy is not a permanent solution and may lead to complications such as hyperparathyroidism, hypercalcemia, and kidney stones.49,50
Conclusions
The rarity of PMT and the nonspecificity of clinical manifestations pose a major challenge to the diagnosis of this disease. This article reported 2 cases of PMT in the right thigh and reviewed the relevant literature, which might help clinicians and pathologists improve their understanding of this disease. A combination of clinical, imaging and pathological features is helpful in the diagnosis of the disease.
Footnotes
Acknowledgements
We are grateful to the professors from the Department of Pathology, Xiangya Hospital, Central South University, Hunan, China, for their assistance with pathological diagnosis.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the National Natural Science Foundation of China (project no. 81602167), the Hunan Provincial Natural Science Foundation of China (project no. 2017JJ3494 and 2021JJ31100), and the Science and Technology Program Foundation of Changsha City (project no.kq2004085).
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors made contributions to the study conception and design. The first draft of the manuscript was completed by Ruifeng Wang. Material preparation, data arrangement and analysis were performed by Ruifeng Wang, Jiayu Zhou, Yupei Yu, Junqi Deng, Ze Wu, Yanhao Wu and Chunlin Ou. Confirmation of pathological analysis was carried out by Keda Yang. Study design and organization and writing—review and editing were undertaken by Keda Yang and Junpu Wang. The final manuscript was read and approved by all authors.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Consent for Publication
All authors consent to the publication of the manuscript in Clinical Pathology.
Ethics Approval and Consent to Participate
Not applicable.
