Abstract
Introduction:
Tumor-induced osteomalacia (TIO) is a rare paraneoplastic syndrome that is characterized by hypophosphatemia resulting from the leakage of renal phosphate. Due to its diverse and nonspecific clinical manifestations, the diagnosis is often delayed.
Case Report:
We present the rare case of a 33-year-old man with TIO who was presented with chronic bilateral hip joint pain and was initially misdiagnosed with osteonecrosis of the femoral head. He suffered from gradually increasing pain and weakness, culminating in walking only with the aid of crutches and severe limitation in daily activities. He was admitted to our hospital for a planned total hip arthroplasty. 68Ga-DOTA-TATE positron emission tomography/computed tomography and pathology results confirmed a paranasal mesenchymal tumor. The patient was eventually diagnosed with TIO. The tumor removal led to a complete reversal of this patient’s health condition, restoring his ability to walk, and no signs of recurrence after the follow-up of 1 year.
Conclusion:
TIO due to a tumor of the nasal cavities and paranasal sinuses is very rare. Because of high rates of misdiagnosis and delayed diagnosis, nasal endoscopy can also be commended as a screening method for osteoporosis and hypophosphatemia in young people.
Keywords
Introduction
Tumor-induced osteomalacia (TIO), also known as oncogenic or oncogenous osteomalacia, is a rare paraneoplastic syndrome.1,2 The pathophysiology involves tumor-secreted “phosphatonins,” factors causing renal phosphate wasting, first described in 1994. 3 Fibroblast growth factor 23 (FGF23) is the primary pathogenic phosphatonin implicated in TIO and related hypophosphatemic disorders. 4 FGF23 downregulates sodium-phosphate cotransporters in the proximal renal tubule (inducing phosphaturia) and suppresses 1,25-dihydroxyvitamin D (1,25(OH)2D) production. 5 This leads to chronic hypophosphatemia, impaired intestinal phosphate absorption, inadequate bone mineralization, and osteoblastic dysfunction. Patients typically present with bone pain, muscle weakness, pathological fractures, and height loss. Biochemical hallmarks include elevated serum FGF23, low or inappropriately normal 1,25(OH)2D, hypophosphatemia, elevated alkaline phosphatase (ALP), and normocalcemia. Due to its rarity, nonspecific symptoms, and insidious onset, TIO is frequently misdiagnosed, with diagnostic delays exceeding 2 years in over 80% of cases. 6
We report a young man with chronic bilateral hip pain initially misdiagnosed as osteonecrosis of the femoral head (ONFH) and scheduled for total hip arthroplasty. Multidisciplinary team (MDT) collaboration enabled rapid diagnosis of TIO and curative resection of the causative tumor within 1 week, avoiding unnecessary joint replacement. This study adheres to the CARE guidelines. 7
Case Report
A 33 year-old Chinese man presented to West China Hospital, Sichuan University, in September 2023 with a 3 year history of progressive bilateral hip pain, predominantly left-sided, initially misdiagnosed as ONFH at a local hospital. One year before admission, pain became severe, limiting walking distance to 100 m and requiring crutches for ambulation. He reported no fracture history or relevant family history. Careful history revealed occasional mild nasal obstruction. Musculoskeletal exam showed deep tenderness over both hips (right > left), greater trochanteric percussion pain, and grade 4 muscle strength bilaterally in the lower limbs. Flexion and extension elicited pain. Patrick’s test was positive bilaterally.
Initial laboratory investigations showed normal calcium concentration of 2.21 mmol/L and parathyroid hormone of 5.6 pmol/L, decreased phosphate concentration of 0.48 mmol/L, decreased 25-oxyhydroxy vitamin D levels of 43.6 nmol/L, and full blood count, renal function, liver function, and fasting blood glucose tests were normal (Table 1). X-rays of the pelvis, femoral neck, shoulder joint, and knee imaging examinations indicated bilateral femoral head necrosis and osteoporosis.
Laboratory Test Results.
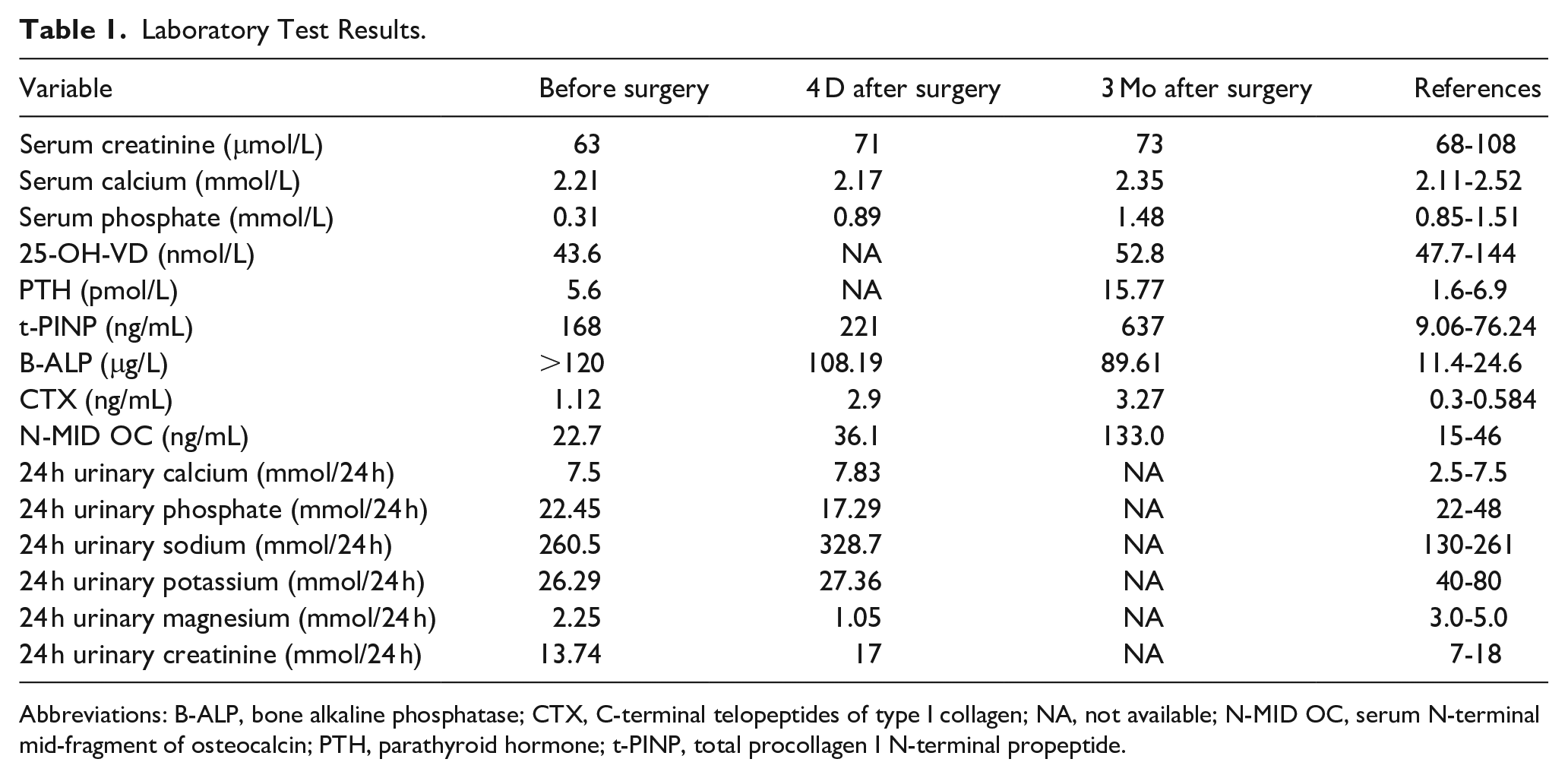
Abbreviations: B-ALP, bone alkaline phosphatase; CTX, C-terminal telopeptides of type I collagen; NA, not available; N-MID OC, serum N-terminal mid-fragment of osteocalcin; PTH, parathyroid hormone; t-PINP, total procollagen I N-terminal propeptide.
Since the cause of osteoporosis and hypophosphatemia in this young patient was unclear, bone metabolism and urine protein excretion were evaluated. The serum levels of bone ALP (B-ALP), total procollagen I N-terminal propeptide, and C-terminal crosslinked telopeptide of type I collagen were high, but N-terminal mid-fragment of osteocalcin was normal. The serum protein electrophoresis and serum immunofixation electrophoresis showed no M-protein or monoclonal immunoglobulin. The 24 hour urine protein and urinary microalbumin were 0.1 g/L (normal <0.15 g/L) and 2.41 mg/L (normal <19 mg/L), respectively. The urinary β2-microglobulin was 0.441 mg/L (normal <0.25 mg/L), and β1-microglobulin, urinary transferrin, and immunoglobulin G levels were all within the normal range (data not shown). Pelvic/femoral neck radiographs indicated bilateral femoral head necrosis and osteopenia. Bone mineral density (BMD, iDXA; GE Lunar) was significantly reduced at all sites compared to age-/sex-matched norms (Table 2). Whole-body bone single-photon emission computed tomography (SPECT) revealed increased uptake in both femoral heads and the right knee joint (Figure 1). Following MDT discussion, hypophosphatemic osteomalacia was suspected based on the young age, severe osteoporosis, bilateral ONFH, and profound hypophosphatemia. Further evaluation confirmed renal phosphate wasting: reduced Tubular reabsorption of phosphate (TRP; 73.9%; normal 85%-95%) and low tubular maximum reabsorption of phosphate (TMP)/glomerular filtration rate (GFR) (0.23 mmol/L; normal 0.84-1.23). Bone marrow biopsy showed acceptable hematopoietic cellularity with a focal mild increase in reticulin fibers. Immunophenotyping revealed no abnormal cell populations. Hematologic malignancy was excluded. 68Ga-DOTA-TATE positron emission tomography/CT (PET/CT) identified a somatostatin receptor-avid lesion in the right nasal cavity/paranasal sinuses, suggestive of a neuroendocrine tumor (Figure 2a–d). Dedicated sinus CT confirmed a 4.6 × 3.1 cm soft tissue mass involving the right inferior nasal meatus, maxillary sinus, and ethmoid sinus (Figure 2e–g).
The BMD Measurements of the Patient Before Surgery and 9 Mo After Surgery.
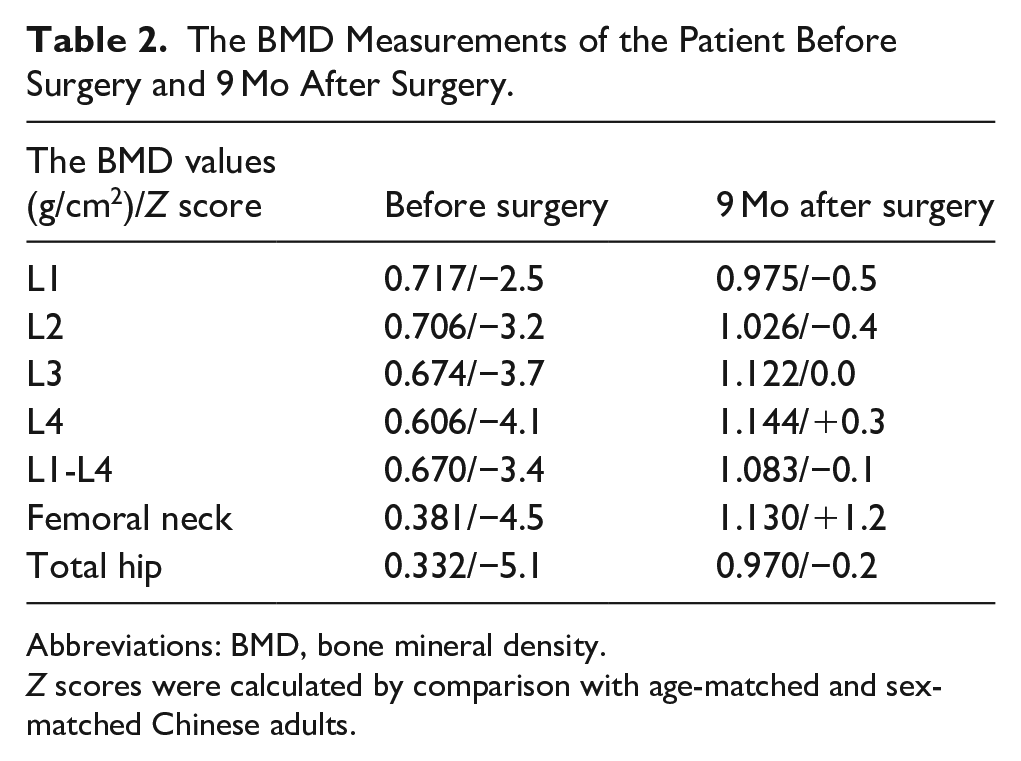
Abbreviations: BMD, bone mineral density.
Z scores were calculated by comparison with age-matched and sex-matched Chinese adults.
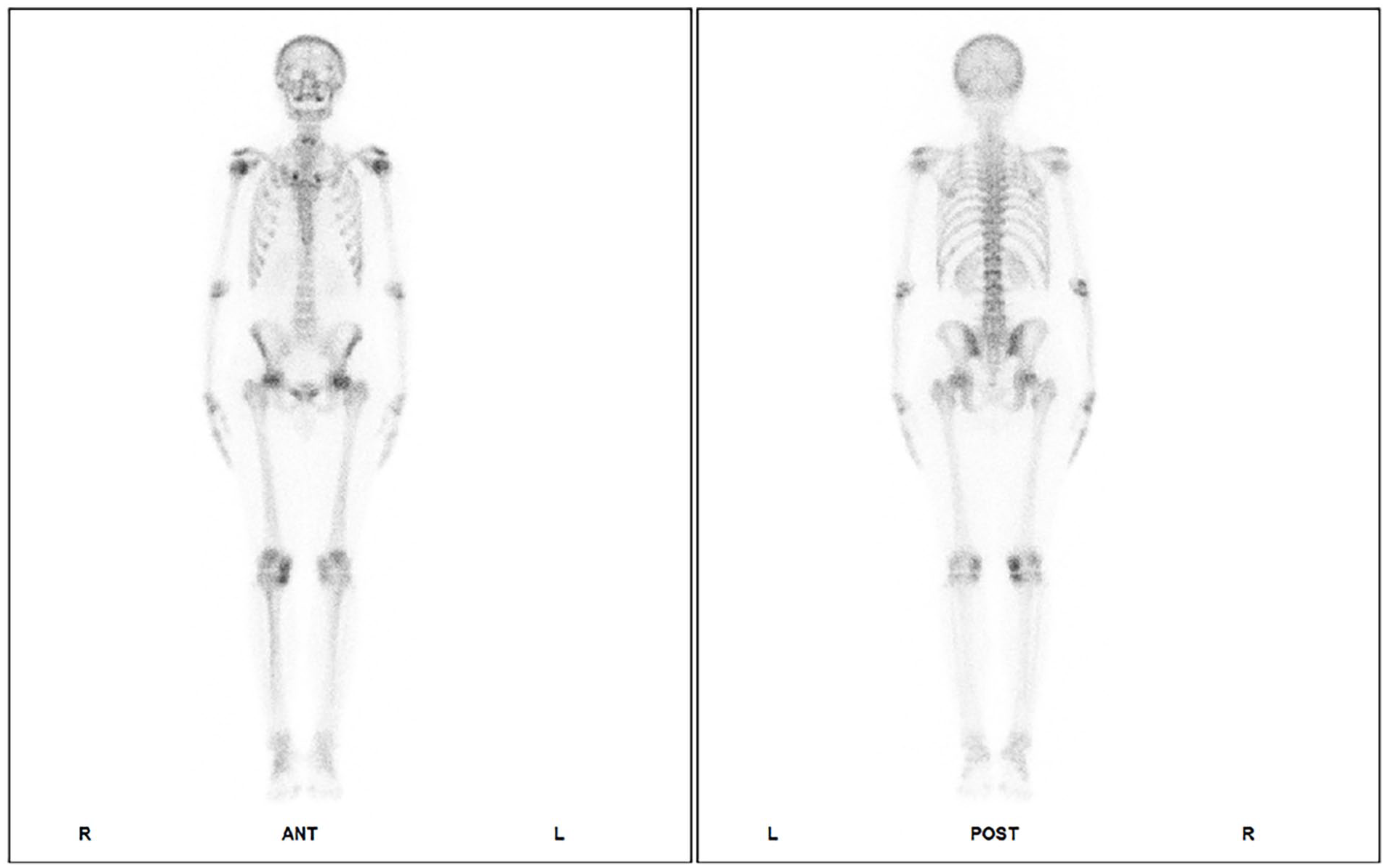
The whole-body bone SPECT of the patient. SPECT, single-photon emission computed tomography.
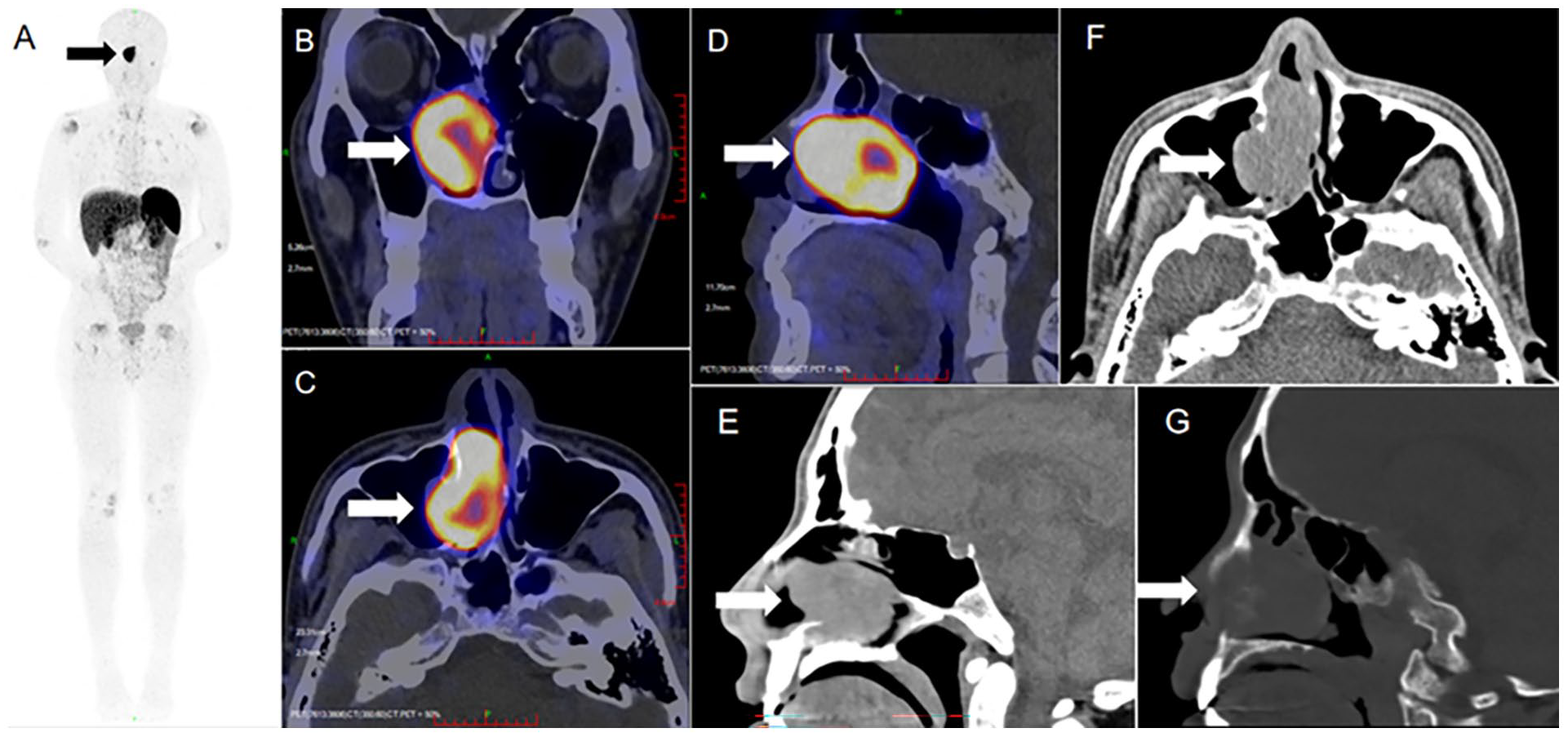
The tumor was detected by 68Ga-pentixafor PET/CT in this patient ((A) whole-body, (B) coronal, (C) axial, and (D) sagittal planes) and sinus thin-slice CT scans ((E) sagittal view in soft-tissue window, (F) sagittal view in bone window, and (G) axial plane). PET/CT, positron emission tomography/computed tomography. Each arrow pointed a tumor mass.
The mass, originating from the right lateral nasal wall, was completely resected endoscopically. The tumor measured 32 × 21 × 8 mm, appeared gray-white to taupe, and had a solid cut surface. Histopathology confirmed a phosphaturic mesenchymal tumor (PMT; Supplementary Figure 1). Immunohistochemical staining showed tumor cell CK (Pan) (−), EMA (part+, weak), HMB-45 (−), Desmin (−), SMA (little+, weak), TFE3 (−), CD34 (−), S100 (−), MART-1 (−), collagen type IV (−), Ki-67 (MIB-1; little+, 5%-10%), CgA (−), Syn (−), CD56 (+), CD31 (−), ERG (part+), SSTR2 (+), SATB2 (+), INSM1 (−). Postoperatively, the patient received calcitriol (0.5 µg/day), calcium carbonate (500 mg/day), and high-dose vitamin D3 (5000 IU/day initially). Serum phosphate normalized (0.89 mmol/L) within 5 days, accompanied by improved TMP/GFR (0.66 mmol/L) and normalized TRP (89.9%). Hip, shoulder, waist, and knee pain, along with upper extremity weakness, partially resolved. He was discharged on postoperative day 7.
At 1 month follow-up, supplementation was adjusted to calcitriol 0.5 µg/day, calcium carbonate 500 mg/day, and vitamin D3 5000 IU/week. By 3 months, he walked stairs unaided with normal serum phosphate. Quantitative CT of the lumbar spine (December 2023) and serial Dual-energy X-ray absorptiometry (DXA) scans at 3 and 9 months, respectively after the operation (Table 2 and Supplementary Table 1) showed progressive BMD improvement. After 1 year, he reported unrestricted jogging and squatting.
Discussion
TIO is primarily caused by FGF23-secreting PMTs. 8 While PMTs most commonly involve the soft tissues of extremities and appendicular skeleton, craniofacial, and sinonasal locations (as in our case) are well documented but less frequent.9,10 The mean age at diagnosis is ~38 to 55 years,11,12 with tumors predominantly occurring in the lower limbs (49.5%), head/neck (27.0%), and upper extremities (7.4%). 13 Sinonasal PMTs often lack specific local symptoms and are easily overlooked. Our patient reported only mild, intermittent nasal obstruction, significantly overshadowed by debilitating bone pain. This underscores the need for meticulous history-taking, including subtle upper respiratory symptoms, in patients with unexplained musculoskeletal complaints.
Diagnostic delays are common due to nonspecific presentations and difficulties in tumor localization. Initial presentation to orthopedics or rheumatology often delays endocrine and otolaryngologic evaluation. Key differential diagnoses for acquired hypophosphatemic osteomalacia in adults include TIO, repeated intravenous iron administration, and Fanconi syndrome.14,15 Hereditary FGF23-mediated disorders (eg, X-linked hypophosphatemic rickets) must also be excluded, particularly in younger patients or those with a family history. 16 Elevated FGF23 correlates with disease burden in plasma cell dyscrasias. 17 Thus, bone marrow biopsy is valuable to exclude hematologic malignancies. Persistent hypophosphatemia after vitamin D repletion should raise suspicion for TIO. 18 TMP/GFR measurement is crucial in hypophosphatemic patients with bone pain, gait disturbance, or muscle weakness. Once TIO is suspected, tumor localization is imperative, as complete resection is curative. Notably, 4.4% of TIO patients are misdiagnosed with ONFH, 12 but prompt TIO treatment can induce ONFH remission, avoiding arthroplasty. 19
TIO patients are frequently misdiagnosed with osteoporosis due to reduced BMD on DXA scans, which cannot reliably differentiate osteomalacia. Anti-resorptive therapy (eg, bisphosphonates) for presumed osteoporosis can be detrimental in osteomalacia by inhibiting bone repair. 15 Our case was atypical as whole-body SPECT showed focal increased uptake only in symptomatic joints (hips, knee), lacking the diffuse pattern sometimes seen in severe osteomalacia. 20 This highlights SPECT’s limitations as a screening tool for osteomalacia. 68Ga-DOTA-TATE PET/CT is the recommended first-line imaging modality for TIO tumor localization, boasting high detection rates (97.7%-100%) and superior accuracy compared to other techniques.21,22 Our findings corroborate reports of significant BMD recovery after TIO cure. 23
Given the risk of recurrence, we recommend regular monitoring: serum PTH, 25-OHD, creatinine, calcium, phosphate, albumin, magnesium, and 24 hour urinary calcium, creatinine, and phosphate—initially quarterly, then annually. 24 For unresectable tumors, medical management (calcitriol, phosphate) is mainstay; radiofrequency ablation or cryoablation are alternative options.25,26 Burosumab, an anti-FGF23 monoclonal antibody, effectively restores phosphate homeostasis, improves osteomalacia, promotes fracture healing, and enhances mobility in patients with unresectable tumors or contraindications to surgery. 27
In conclusion, we present a case of TIO initially misdiagnosed as ONFH, while avoided hip-replacement surgery. The cooperation of a MDT is essential to produce the right diagnosis and implement appropriate treatment in this case within a week, enriching the clinical characterisation. Clinicians should improve the recognition of this disease to achieve early diagnosis and administer the appropriate treatment.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251371415 – Supplemental material for A Sinonasal Paranasal Mesenchymal Tumor Associated With Tumor-Induced Osteomalacia: A Case Report and Literature Review
Supplemental material, sj-docx-1-ear-10.1177_01455613251371415 for A Sinonasal Paranasal Mesenchymal Tumor Associated With Tumor-Induced Osteomalacia: A Case Report and Literature Review by Hongping Gong, Fangyu Shi, Hongyu Deng, Zongke Zhou, Junming Xian, Yan Huang, Qin Wang, Yuhan Li and Bo Yuan in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
Our most sincere gratitude is extended to the patient because he allowed us to share our experience to provide new insights to the scientific community.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and according to the policy of the Ethics Committee of the Ethics Committee of the West China Hospital, Sichuan University for case reports’ publishing.
Consent to Participate
Signed consent to treatment from the patient was obtained and personal data were anonymized.
Consent for Publication
Signed consent to publish from the patient was obtained.
Author Contributions
Conceptualization: Hongping Gong and Fangyu Shi. Investigation resources: Zongke Zhou, Qin Wang, Junming Xian, Yan Huang, and Yuhan Li. Data curation: Hongping Gong, Fangyu Shi, and Hongyu Deng. Writing—original draft preparation: Hongping Gong, Fangyu Shi, and Bo Yuan. Supervision: Yan Huang, Yuhan Li, and Bo Yuan. All authors contributed to the interpretation of the data and critically revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Scientific and Technological Research and Development Projects (HX-H2409369) and National Science and Technology Major Project (2023ZD0506103).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original research data in this case report are available on request from the corresponding authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
