Abstract
Purpose:
Research examining the psychological and physical outcomes of hormone treatment in transgender adolescents have yielded inconsistent findings. Given these uncertainties and potential for adverse health events, it is critical to evaluate the long-term outcomes among this population. This systematic review aimed to summarize available evidence from the published literature investigating the long-term effects of hormone treatment of psychological (i.e., anxiety, depression, and gender dysphoria [GD]) and physical (i.e., cardiovascular and skeletal) outcomes within transgender adolescents.
Methods:
A total of 1,239 longitudinal clinical articles published in English between 2015 and 2024 were retrieved from PubMed, SCOPUS, EBSCOHost, and Science Direct. After extensive screening, 17 longitudinal studies were included in the systematic review (N = 12,090) with the treatment follow-up period ranging from six months to 15 years.
Results:
Hormone therapy was associated with improvements in anxiety, depression, and GD, but results were mixed among studies. In addition, changes were observed in cardiovascular measures (i.e., blood pressure, hemoglobin, hematocrit, and cholesterol) and skeletal measures (i.e., bone mineral density [BMD]). However, there was insufficient evidence to conclude whether these changes from hormone treatment increased the risk of developing pathogenesis, particularly adverse cardiovascular events, and osteoporosis.
Conclusion:
Hormone therapy may improve psychological outcomes, but study conclusions should be interpreted with caution, given the short treatment duration, limited controls, and homogenous samples. Future studies should investigate the psychological benefits and physical changes as transgender individuals progress from adolescence into later adulthood and include control groups.
Keywords
Abbreviations and Definitions Used
BMD: Bone Mineral Density
BMAD: Bone Mass Apparent Density
aBMD: Areal Bone Mineral Density
LS: Lumbar Spine
FN: Femoral Neck
TGD: Transgender Diverse
Transgender: An individual whose gender identity is different from the sex assigned at birth.
Hormone Therapy: A medical treatment that adds, blocks, or removes hormones in the body to treat various conditions, including gender dysphoria.
Introduction
The number of adolescents identifying as transgender and seeking gender-affirming care has increased significantly over the past few decades. 1 Transgender individuals may experience persistent and impairing psychological distress due to this incongruence, known as gender dysphoria (GD). To alleviate this distress, transgender adolescents may undergo treatment to better align their physical characteristics with their gender identity.2–4 Medical options include gender-affirming hormone treatment (GAHT), also referred to as cross-sex hormone treatment, and puberty blockers. 5 GAHT serves to induce the development of secondary sex characteristics that align with one’s gender identity and can include estrogen, testosterone, progesterOne, and anti-androgens (i.e., spironolactone, cyproterone).1,2 Gonadotropin-releasing hormone analogs (GnRHa) are commonly used as a puberty blocker to suppress puberty by delaying the development of secondary sexual characteristics.2,5
However, hormone treatment for transgender adolescents has been subject to debate and ethical discussion among healthcare professionals. 5 Recent concerns have pointed to long-term health outcomes and treatment irreversibility during adolescence, which is a critical period for physical, psychological, and social development. 6 Organizations, such as the World Professional Association for Transgender Health (WPATH) and the Endocrine Society, provide clinical practice guidelines based on patient eligibility.1,2 According to the WPATH standards, hormone therapy can have reversible and partially reversible changes. Fully reversible interventions may include GnRHa, which delays changes in puberty and decreases the effects of androgens secreted. 3 In contrast, feminizing and masculinizing hormone therapy may lead to irreversible changes and poor health outcomes, such as decreased fertility, which can have long-term implications for development.2,3,6
Previous literature presents conflicting results regarding the effects of hormone treatment on psychological, cardiovascular, and skeletal health. Transgender individuals are disproportionately affected by stigma, harassment, and discrimination, and face an increased risk for anxiety and depression.2,7 While there is growing evidence to support the improvement of mental health among transgender individuals taking hormone treatment, the existing data is inconclusive. 8
Studies in adolescents have also shown inconsistent findings for cardiovascular health outcomes. These include variations in high-density lipoprotein (HDL), low-density lipoprotein (LDL), hemoglobin, hematocrit, and blood pressure.1,9–11 Given these inconsistencies, it is unclear how an individual’s medical history and specific risk factors might contribute to adverse cardiovascular events or whether patients can make fully informed decisions to undergo hormonal treatment.
Similarly, there is a lack of research on the long-term effects of hormone treatment on skeletal health, such as bone mineral density (BMD), and the risk for osteoporosis, or whether these effects on bone health are reversible with treatment cessation.1,12,13 Since adolescence is a critical developmental period, these inconsistent findings highlight the need for more longitudinal studies to better train healthcare providers in assessing and administering treatment to patients presenting with GD.
The objective of this systematic review was to summarize existing literature investigating the long-term psychological and physical outcomes of hormone treatment within transgender adolescents. Clinically significant levels of the cardiovascular and skeletal outcomes measured may increase the risk of developing chronic life conditions, such as cardiovascular disease, stroke, hypertension, and osteoporosis.13,14 Given the limited and inconsistent data on treatment outcomes, this study sought to provide a comprehensive review to inform the decision-making of transgender individuals and their health practitioners. Previous reviews have narrowly focused on either psychological or physical outcomes. This systematic review aimed to examine both the long-term psychological (i.e., anxiety, depression, and GD) and physical (i.e., cardiovascular and skeletal health) changes that occur after hormone treatment within transgender adolescents.
Materials and Methods
Protocol and Registration
This systematic review was prepared according to the Preferred Reporting Items for Systematic Reviews guidelines 15 and followed the methodology recommendations outlined by Boland, Cherry, and Dickson (2017). 16 The protocol was established a priori but was not published. The methods were developed with and reviewed by Anna J. Dreyer (AJD), a postdoctoral researcher at the University of Cambridge. Since the researcher did not interact or collect data directly from human subjects, Institutional Review Board (IRB) review or approval was not required.
Search Strategy
The lead researcher searched four electronic databases: PubMed (including MEDLINE), Scopus (excluding MEDLINE), ScienceDirect (excluding MEDLINE), and EBSCO electronic databases for relevant published literature. Articles published within the last 10 years (2015–2025) were included to ensure the review reflects the most recent and relevant data. A final set of search terms was developed after several scoping reviews and included keywords such as “hormone,” “longitudinal,” and “gender identity,” and combined the Medical Subject Headings term “transgender” to account for variation in scientific articles. The search went from May 2024 to August 2024. See Appendix SA1 for the PubMed search strategy, which was modified as needed for other electronic databases.
Inclusion and Exclusion Criteria
Table 1 presents the eligibility criteria for studies included in the review. Studies were included if they were longitudinal, published in English, and focused on healthy transgender adolescents undergoing hormone therapy to better align their physical appearance with their gender identity. Adolescent age was based on each study’s definition. Psychological outcomes of interest included symptoms that were strongly associated with GD, such as anxiety and depression, from prior scientific articles.2,17 Physical outcomes included anatomical and physiological changes that present risk factors for developing long-term pathogenesis, given their level of clinical significance.1,3 Exclusion criteria included reviews, conference abstracts, case reports, chapters, cross-sectional studies, and non-longitudinal studies. Studies involving transgender adolescents with pre-existing medical conditions or those undergoing surgical interventions were also excluded.
Population, Intervention, Comparators, Outcomes, Study design, and Setting. 16
Study Selection
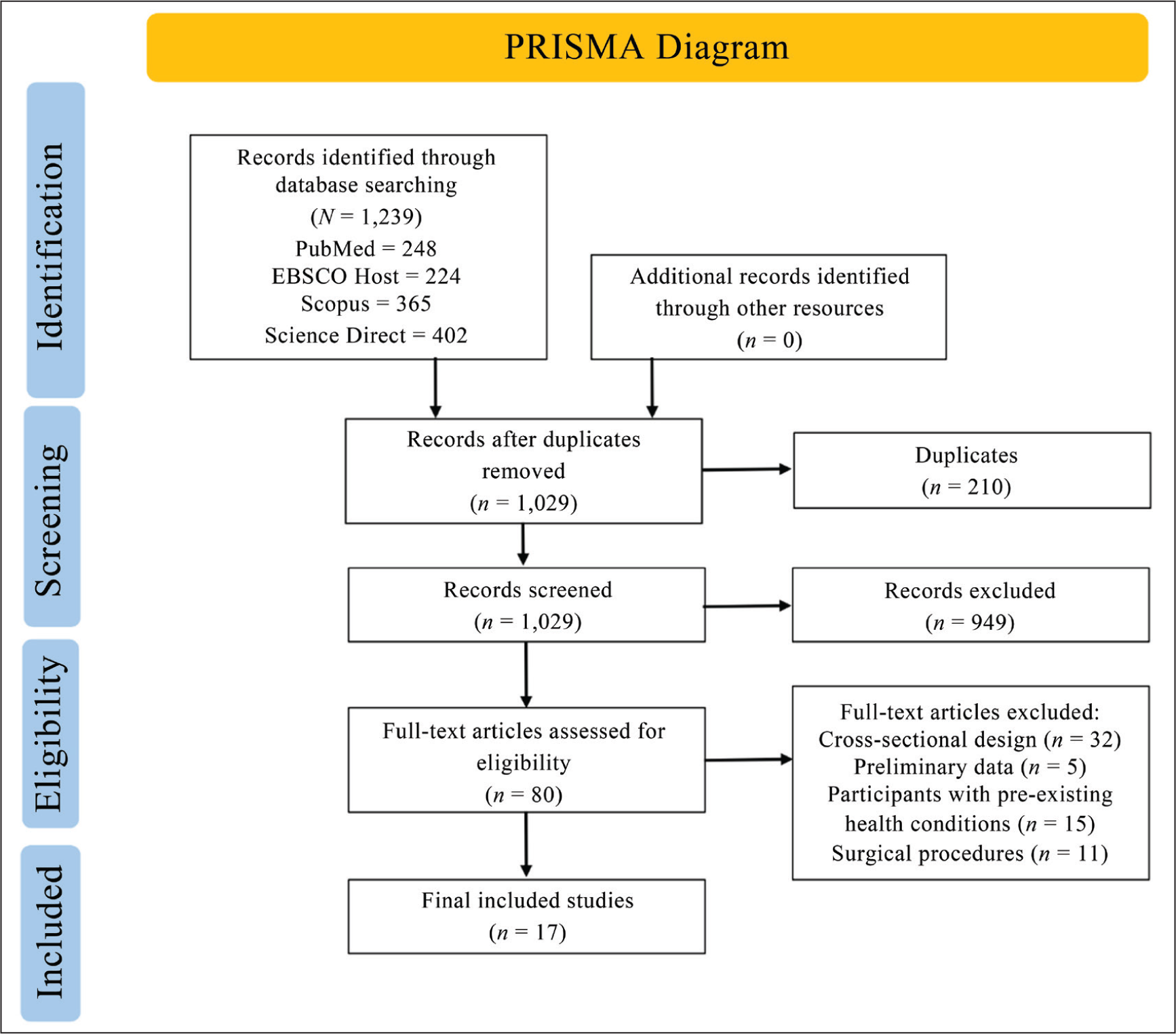
References were managed using Zotero (Version 6.0.37). A screening tool was developed by AJD and the lead researcher based on several scoping searches. This tool was based on the inclusion and exclusion criteria outlined in Table 1 to help raters make decisions on whether a study should be included in the systematic review. The researchers tested the tool by screening 20 abstracts to ensure reliability and consistency. Next, 1,024 titles and abstracts were manually screened from the combined search results, and any uncertainty about whether a study should be included or not was discussed by AJD and Hailey G. Prokop (HGP) until a consensus was reached. Full-text copies were then obtained for all potentially relevant articles and screened with the same criteria, resulting in the final sample of 17. AJD independently screened 10% of a random sample to ensure cross-check fidelity. Figure 1 illustrates the full search and screening process with reasons for exclusion.
Data Extraction
Data extraction was conducted by HPG using an Excel spreadsheet that listed fields for data that needed to be extracted from each article and provided a space for any relevant information. This tool was used by HGP to extract data from 17 studies. The data included sample demographic characteristics (i.e., age, sample size, gender, and ethnicity), clinical characteristics (i.e., interventions, dosage, tests used, intervention groups, and outcomes), and study design and quality information (i.e., country and date of publication). A meta-analysis was not conducted due to substantial heterogeneity in study interventions, measures, and designs.
Risk of Bias in Individual Studies
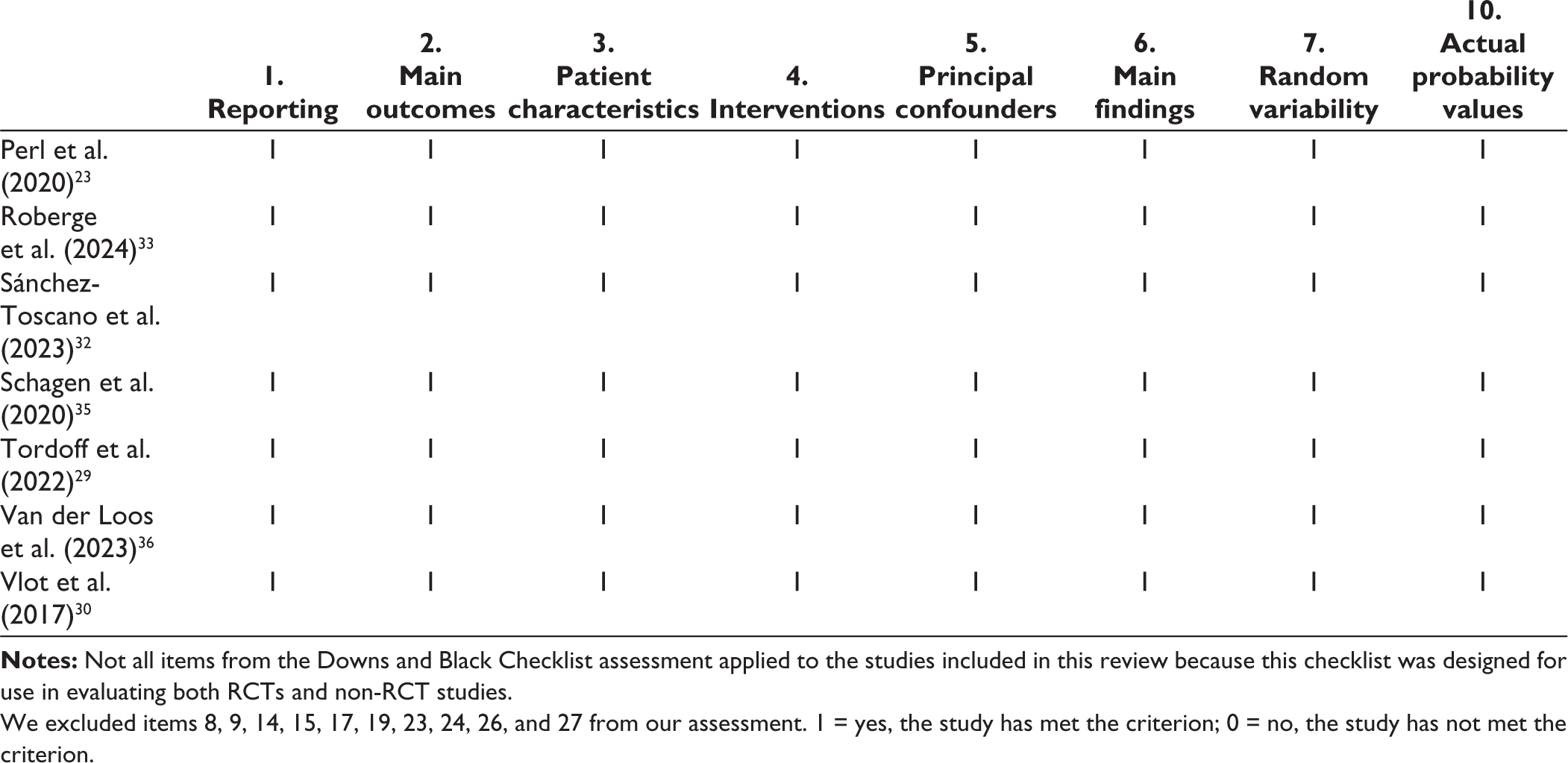
The methodological quality of the 17 studies was evaluated using the Downs and Black Checklist (1998). 18 Because this 27-item checklist was designed for evaluating both randomized controlled trials (RCTs) and non-RCT studies, only a subset of 16 items applied to the studies included in this review. Specifically, the quality of reporting, external validity, internal validity, and statistical power were assessed. Items 8, 9, 14, 15, 17, 19, 23, 24, 26, and 27 were excluded from the assessment because they were not relevant to the majority of studies included in the review.
Results
Study Characteristics
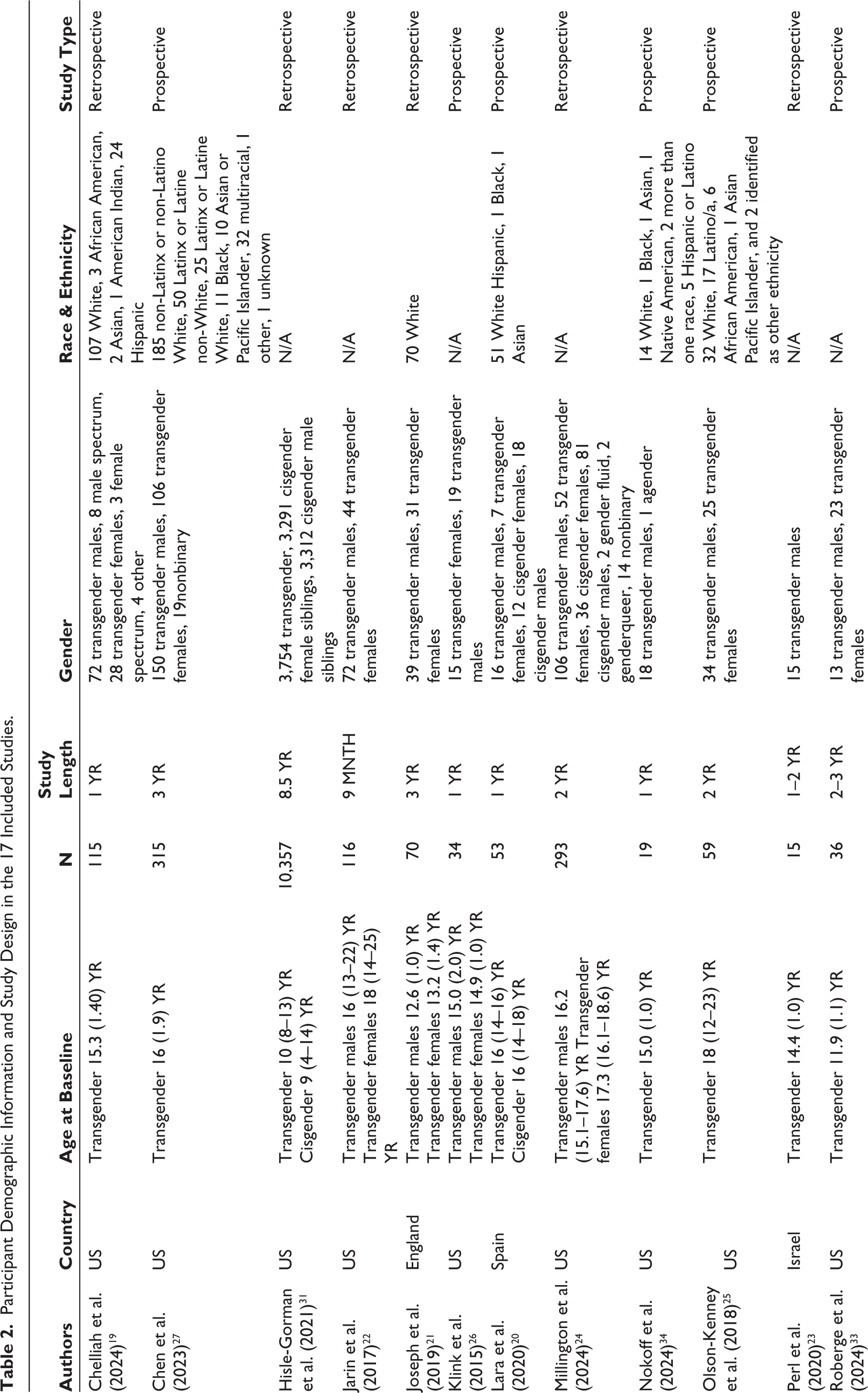
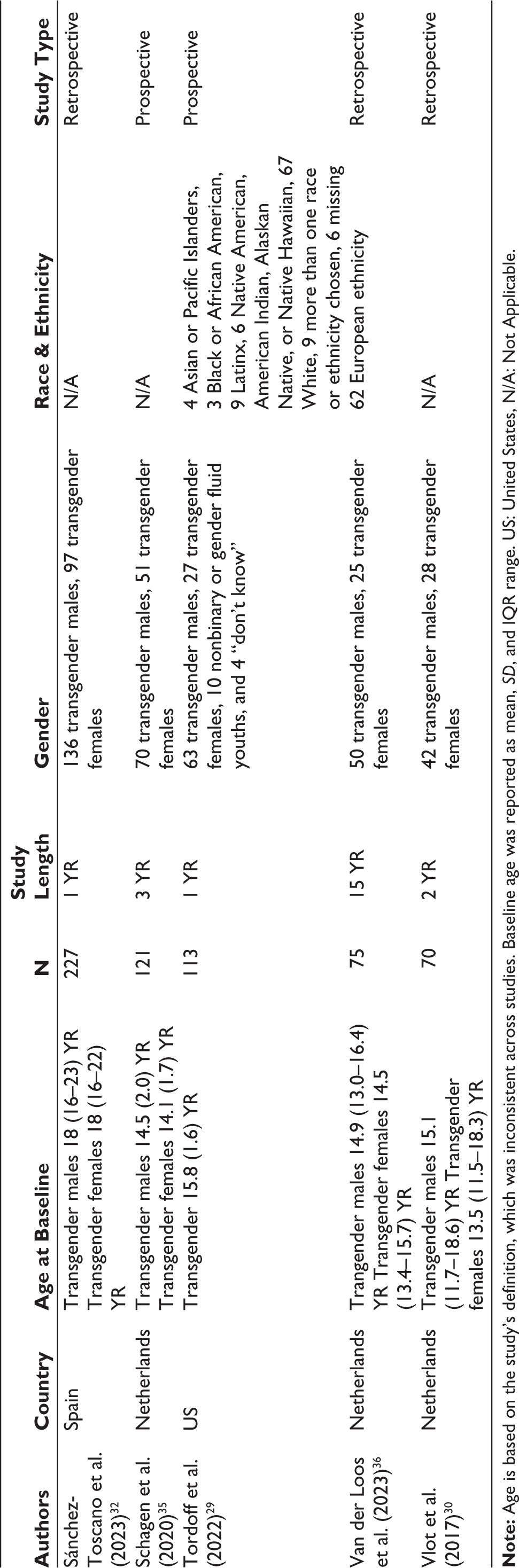
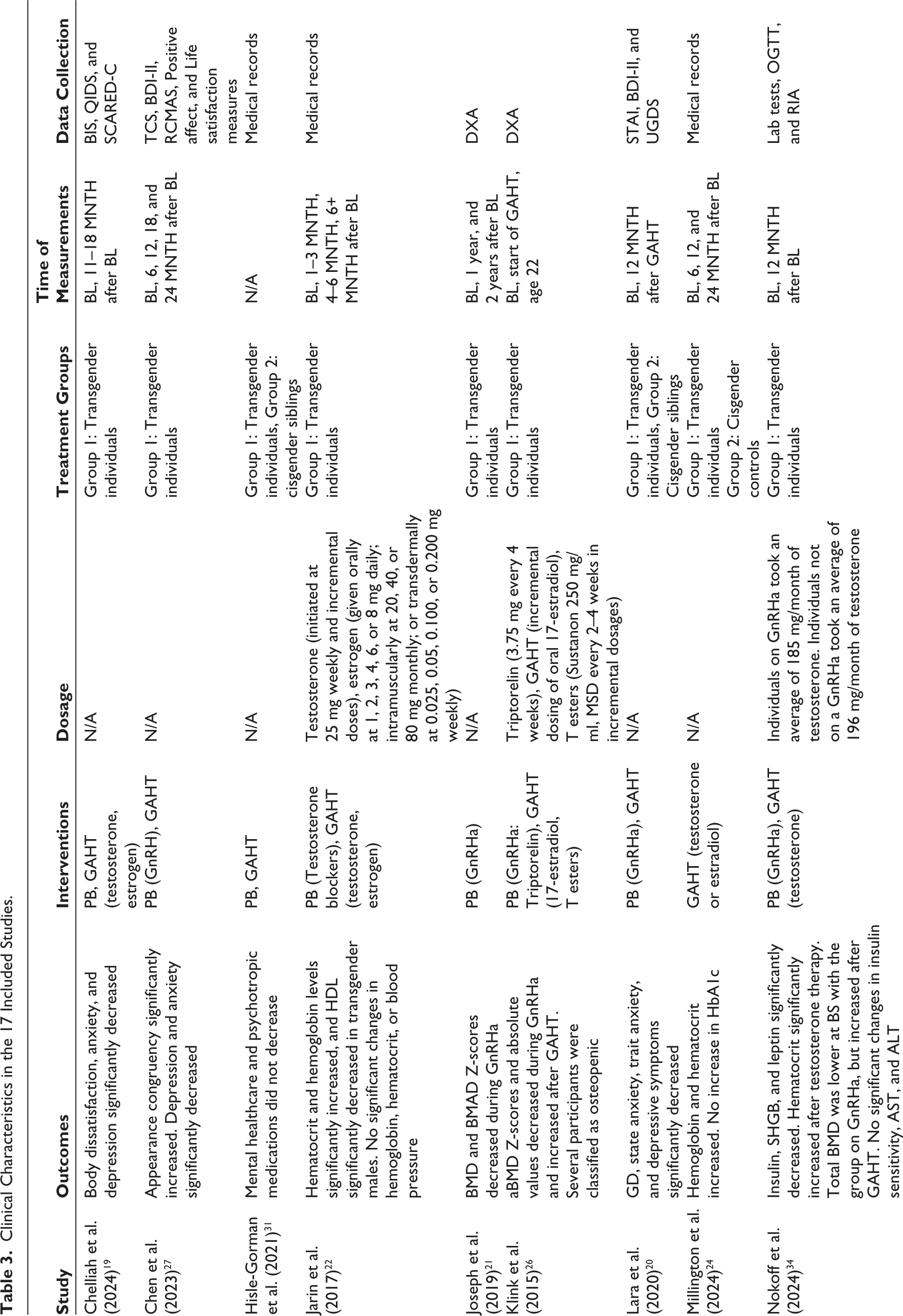
Table 2 displays the demographic information and study design, while Table 3 displays the clinical information for the 17 included studies.19–35 Of these, nine studies19,21–24,30–32,36 were retrospective and eight20,25–27,29,33–35 were prospective in design. The sample groups of interest consisted of transgender males (n = 3,454) and transgender females (n = 1,702). Three studies included cisgender controls (cisgender males: n = 3,411; cisgender females: n = 3,339),20,24,31 while 14 did not have control groups. None of the selected articles were qualitative studies; rather, they contained quantitative data. Study samples ranged from 15 to 10,357 participants, with most including more than 70 participants. Summary statistics were calculated for the average length of study in years (Mean [M] ± standard deviation [SD] = 2.96 ± 3.60) and the age of participants before treatment initiation (M = 15.02) in studies reporting mean age at baseline and (interquartile range = 8–25) within all included studies. Ten studies reported the Tanner stages at which treatment was initiated.20,23,24,26,27,30,33–36 Of these, the majority of participants started puberty suppressors during Tanner stages 2–3, and started GAHT in Tanner stages 4–5.
Participant Demographic Information and Study Design in the 17 Included Studies.
Clinical Characteristics in the 17 Included Studies.


Studies were published between 2015 and 2024, with the majority published in the last five years. Ten were conducted in the United States,19,22,24–27,29,31,33,34 six in Europe,20,21,30,32,35,36 and one in Israel. 23 While 10 studies did not report ethnicity, participants from these studies were predominantly composed of White individuals.22–24,26,30–33,35,36 The length of study varied from six months to 15 years, although most lasted two years or less. Regarding interventions, two studies used only puberty suppressors (i.e., GnRHa),21,33 one study used only GAHT (i.e., estrogen, progesterone, and testosterone), 24 and 14 used both puberty suppressors and GAHT, where a majority started with puberty suppression and took GAHT concurrently or sequentially.19,20,22,23,25–27,29–32,34–36 Of these 14 studies, eight took sequential measurements after each intervention,19,20,22,25,27,29,31,32 while six studies reported values only after both interventions were taken.23,26,30,35–37
Review of Included Studies
The results of the 17 studies are integrated and summarized below. Each outcome was reviewed separately and was split into psychological, cardiovascular, and skeletal outcomes.
Psychological Outcomes
Five studies assessed psychological outcomes, including anxiety, depression, and GD.19,20,27,29,31 These comprised two retrospective studies19,31 and three prospective studies.20,27,29
Anxiety. One retrospective study (n = 115) and two prospective (n = 315; n = 23) reported significant reductions (P < .01) in anxiety among transgender individuals following 12–24 months of treatment.19,27 Of these, one found that transgender individuals continued to report worse anxiety scores post-treatment compared to cisgender controls. 20 In contrast, another retrospective study (n = 3,754) found no significant reduction in mental healthcare utilization and psychotropic medication after hormone initiation compared to their cisgender siblings (n = 6,603) over 8.5 years. 31 A second prospective study (n = 113) found no association between hormone treatment and moderate to severe levels of anxiety after 12 months of treatment. 29 Overall, three out of five studies showed a decrease in anxiety symptoms, while two found no improvement in anxiety. Hormone therapy may decrease anxiety among transgender individuals. However, evidence remains low to moderate due to inconsistent findings, lack of controls, and variability in treatment duration.
Depression. One retrospective study (n = 115) and two prospective studies (n = 315; n = 53) reported significant reductions (P < .001) in depression following 12–24 months of hormone treatment.19,27 However, transgender individuals reported more depressive symptoms before and after treatment compared to the cisgender controls. 20 In contrast, a third prospective study (n = 113) found an increase in moderate to severe depression after the three-month follow-up, which returned to baseline at six and 12 months. However, the study reported a 60% lower odds of moderate to severe depression in individuals on puberty blockers and hormones. 29 Three studies found a decrease in depressive symptoms after treatment initiation, and one reported an increase after three months that returned to baseline by six to 12 months. Hormone treatment appears to reduce depressive symptoms among transgender individuals. The evidence for this conclusion is moderate to high, considering the consistent findings and short follow-up periods.
GD. A retrospective study (n = 115) and two prospective studies (n = 315; n = 53) found a decrease in GD among transgender individuals following 12–24 months of treatment. Participants who experienced less improvement in body dissatisfaction exhibited more depression and anxiety at follow-up.19,20,27 There is strong evidence that hormone therapy improves GD, given consistent findings among the three studies. However, treatment duration and control groups serve as limiting factors.
Cardiovascular Outcomes
Six studies assessed cardiovascular outcomes, including hemoglobin, hematocrit, insulin, systolic and diastolic blood pressure, LDL, and HDL.22–25,32,34 Changes in these biomarkers and parameters may reflect potential risks that may undermine cardiovascular health. Clinically significant levels of cardiovascular measures examined in this review have been found to increase the risk for venous and arterial thromboembolism, hypertension, cardiovascular disease, insulin resistance, diabetes, hypothyroidism, and hyperthyroidism.1,5,38 However, it is important to recognize that other factors, such as hormones, diet, and physical activity, can influence these measures and risk for developing adverse cardiovascular events. 39
Hemoglobin and Hematocrit. Among transgender females (n = 52), one retrospective study found a decrease in hematocrit after estradiol, 24 while another retrospective study (n = 44) found no significant changes. 22 Two studies reported significant decreases in hemoglobin (P < .001) among transgender females following estradiol,24,25 while one study found no significant changes. 22 One prospective study reported borderline anemia in one transgender female patient, 25 and another retrospective study observed hemoglobin and hematocrit levels above the typical adult female range for 20% of participants. 24
Among transgender males, three studies found a significant increase (P < .001) in hematocrit following testosterone therapy,22,24,34 while two participants in one study experienced supraphysiologic hematocrit levels. 22 Similarly, three studies found increases in hemoglobin (P < .001; P < .100; P = .040) among transgender males taking testosterone.24,25,32 One study noted that 27% of transgender males continued to have hemoglobin and hematocrit levels below the typical adult male range throughout treatment. 24
Among transgender females, one study found a decrease in hematocrit, one found no significant changes in hematocrit, two studies found a significant decrease in hemoglobin, and one found no significant changes in hemoglobin. Among transgender males, three studies found a significant increase in hematocrit, and three found a significant increase in hemoglobin. Hormone therapy more consistently affects hematocrit and hemoglobin levels in transgender males than in transgender females. Among transgender males, hematocrit and hemoglobin may increase, while transgender females may experience a decrease in hematocrit and hemoglobin levels. The evidence is low to moderate, given the inconsistent findings, small samples, and limited controls.
Insulin and Hemoglobin A1C (HbA1c). A prospective cohort of transgender males (n = 19) showed a significant decrease in fasted insulin values (P = .044) after 12 months of testosterone therapy but found no significant changes in insulin sensitivity. 34 Two retrospective studies found no increases in HbA1c in transgender individuals (n = 293; n = 72 ) after 24 months of hormone therapy. However, two transgender females and five transgender males had elevated HbA1c levels during treatment in one study.22,24
Among transgender females and transgender males, two studies found no changes in HbA1c, one study found a significant decrease in fasted insulin levels but no significant changes in insulin sensitivity. Hormone treatment may decrease insulin values, but the evidence is low given the small sample size. In addition, there is little evidence to support a change in HbA1c levels after hormone treatment, given the consistent findings and limited data.
Systolic and Diastolic Blood Pressure. Among transgender females, one retrospective study found a significant increase in systolic blood pressure (SBP) (P = .026) and diastolic blood pressure (DBP) (P = .017) following one year of treatment, 32 while a second retrospective study (n = 44) found no significant changes in blood pressure. 22
Among transgender males, three studies found significant increases in SBP,22,32 one of these observed one mildly clinical patient. 25 One retrospective (n = 136) found a significant increase in DBP (P = .015), 32 while a second retrospective study found a significant decrease in DBP percentiles (P = .019) after GnRHa among transgender males (n = 15). 23
Among transgender females, one study found significant increases in SBP and DBP, while another found no significant changes. Among transgender males, three studies found a significant increase in SBP, one found a significant increase in DBP, while another found a significant decrease in DBP. There is moderate evidence that hormone treatment affects blood pressure, particularly an increase in SBP among transgender males, given the consistent findings, limited controls, and small sample sizes.
LDL and HDL. Among transgender females, one prospective study (n = 25) showed a significant increase (P < .001) in HDL, 25 and one retrospective study (n = 97) found a significant increase (P = .008) in total cholesterol after one year of treatment, 32 while a second retrospective study (n = 44) found no significant changes in LDL or HDL cholesterol. 22
Among transgender males, one study (n = 34) found a significant increase (P = .001) in HDL, 25 two studies (n = 72; n = 136) found a significant decrease (P < 0.05; P < .001) in HDL levels after treatment,22,32 and one found no significant changes in HDL after GnRHa treatment. 34 Of these, one study found an increase in total cholesterol (HDL and LDL) that leveled off after three months, 22 while one study (n = 19) found no significant changes in total cholesterol after GnRHa initiation. 34
Among transgender females, one study found an increase in HDL, one found an increase in total cholesterol, while two studies found no changes in LDL and HDL. Among transgender males, three studies found a significant decrease in HDL, one found an increase in total cholesterol, and one study found no significant change in HDL or LDL. There is moderate evidence that hormone therapy changes LDL and HDL levels, particularly a decrease in HDL among transgender males, based on consistent findings, lack of controls, and sample size.
Skeletal Health Outcomes
Seven studies assessed skeletal health outcomes, including three retrospective and four prospective studies.21,26,30,33–36 Outcomes of interest are BMD, which includes bone mineral apparent density (BMAD), areal bone mineral density (aBMD), and bone mass accrual. Low levels of BMD have been shown to increase the risk of osteoporosis and osteopenia.3,5 The analysis reviews absolute BMD values and their respective Z-scores. The Z-score is used to compare an individual’s BMD to the average BMD of a reference population that is matched for age, sex, and ethnicity. 36
BMD. Six studies reported lower BMD values following GnRHa treatment in transgender participants.21,26,33,34,36 Of these, one retrospective study (n = 70) observed an immediate decline in hip and lumbar spine (LS) BMD and BMAD Z-scores, but BMD and BMAD values did not change substantially during the three years following GnRHa. 21 A prospective study (n = 34) observed a decrease in the LS aBMD Z-scores and absolute values, which increased after GAHT initiation. This study also classified several participants as osteopenic and noted that LS aBMD Z-scores were below the population mean at the start of treatment. 26 A second prospective study (n = 121) found that BMAD values stabilized or showed a decrease (for transgender females whose mean Z-scores were below the population mean), whereas Z-scores decreased for all groups. Although BMAD Z-scores increased during GAHT, the study noted that transgender females had relatively low Z-scores at baseline and after three years of estrogen. 35 Another prospective study (n = 36) also observed below-average aBMD Z-scores for transgender participants undergoing GnRHa. 33
Following GAHT, four studies found an improvement in BMD values and Z-scores among transgender participants.26,30,34,36 Of these, one prospective study (n = 19) observed that participants on GnRHa had greater gains in bone mineral content, but had lower total BMD values at baseline relative to the control. Moreover, the Z-scores were average or slightly below-average after treatment. 34 A retrospective study (n = 70) observed a decrease in LS BMAD Z-scores, which returned to normal after GAHT as BMAD values increased. 30 A second retrospective study (n = 75) reported decreased BMD Z-scores during puberty suppression, which “caught up” with pre-treatment levels after long-term GAHT, except for the LS region in transgender females. 36 Last, two studies (n = 70; n = 36) found a delay in bone mineral accrual during GnRHa treatment that was not fully reversed after the initiation of GAHT. 30,33
Overall, six studies showed a decrease in BMD values and Z-scores after GnRHa,21,26,33,34,36 four studies found an improvement in BMD values and Z-scores after GAHT,26,30,34,36 and two studies found a delay in bone mass accrual following GnRHa and GAHT.30,33 There is high evidence that GnRHa decreases BMD values and Z-scores, and moderate evidence that GAHT increases BMD.
Risk of Bias Within Studies
All 17 studies were assessed using the Downs and Black (1998) 18 and received moderate to fair-quality scores as shown in Tables 4 and 5. Studies implemented accurate outcome measures and reporting of results. However, there were consistent limitations, including the absence of control groups and the need for more representative samples. Three out of the 17 studies included a control group with cisgender individuals who were matched with cisgender siblings and age, ethnicity, and socioeconomic status, respectively.20,24,31 There was also a lack of representation based on gender23,28,34 as studies had a disproportionately higher sampling of transgender males. In addition, White participants were overrepresented and made up the majority of the sample sizes.19,21,25,27,29,34 Studies with the lowest score had the highest risk for bias due to a lack of sample representation, control groups, and mention of confounders in each group.19,27
We excluded items 8, 9, 14, 15, 17, 19, 23, 24, 26, and 27 from our assessment. 1 = yes, the study has met the criterion; 0 = no, the study has not met the criterion.
The Methodological Quality of Studies Included in the Sample Based on the Downs and Black Checklist. 18

Discussion
This review synthesized findings from 17 studies examining the psychological and physical outcomes of transgender adolescents taking hormone therapy. No RCTs were identified, and a summary of the outcomes is discussed below. Overall, the findings indicate an improvement in psychological outcomes, including reductions in anxiety, depression, and GD following hormone treatment. However, transgender individuals continued to report more depressive and anxiety symptoms post-treatment compared to cisgender controls. 20 This aligns with previous research that found significant decreases in psychological symptoms and functional impairment following treatment. 17 However, evidence for this conclusion remains moderate, given the small sample size, the lack of controls, confounding variables, and short treatment duration. Of the three outcomes, improvements in GD were the most consistent finding, while anxiety was the least consistent, as two of the five studies found no improvements following treatment.
The cardiovascular outcomes suggest that hormone therapy changes several markers, including hematocrit, hemoglobin, blood pressure, LDL, HDL, and insulin, within transgender individuals. However, the study findings were often inconsistent in the direction of these changes. Although the majority of participants had clinically insignificant changes, a few studies noted clinically meaningful alterations and worse cardiovascular profiles following hormone therapy.22,24,32 For instance, a retrospective study reported hyponatremia and mild hyperkalemia in one transgender female and supraphysiologic hematocrit in two transgender males. 22 However, these studies mentioned concurrent medications and medical comorbidities as potential confounders. These findings highlight the importance of ongoing monitoring following treatment, especially in the presence of comorbidities and concurrent medication use.
For skeletal outcomes, there were decreases in BMD values, Z-scores, and bone accrual turnover rate during GnRHa that partially recovered after GAHT. Moreover, several studies reported below-average BMD Z-scores among transgender participants, particularly transgender females.33–36 One study classified patients as osteopenic, 26 while a few studies suggested suboptimal bone mineral accrual for transgender adolescents. 33
In summary, the review identified improvements in psychological outcomes along with changes in cardiovascular and skeletal measures. The majority of participants showed non-significant changes in several cardiovascular markers (i.e., hematocrit, hemoglobin, blood pressure, LDL, HDL, and insulin), although a few participants reported worsened cardiovascular profiles after treatment. Last, the majority of studies reported lower BMAD values and Z-scores following GnRHa with partial recovery after GAHT, indicating a potentially suboptimal bone turnover rate.
PRISMA Flow Diagram Depicting the Article Selection Process.
Limitations
This review highlights specific knowledge gaps in long-term outcomes regarding hormonal therapy within transgender adolescents that are necessary, given the increasing prevalence of GD and hormone prescription. 3 First, there is a lack of RCTs for transgender individuals taking hormone treatment. Given the uncertainty of long-term outcomes, it is necessary to conduct more randomized studies to minimize bias and establish causal effects to ensure that healthcare professionals and patients understand all the risks and benefits that can be derived from treatment. It is important to note that appropriate control groups within clinical trials can be challenging due to ethical considerations, blinding restrictions, and participant adherence. However, current research lacks knowledge about the long-term side effects and the potential risks associated with adverse health outcomes among transgender individuals. Second, the included studies observed and analyzed data at a group level, excluding intra-individual changes. Since the WPATH standards stress the need for individualized treatment among transgender individuals, it would be more appropriate to examine these individual differences to account for personal factors. This method would allow for a better understanding of intra-individual reactions to treatment and potential confounding variables.
Third, there is a lack of diversity and generalizability. Studies that reported race and ethnicity have a majority of White samples. Individuals from different racial backgrounds may experience unique challenges that impact their response to treatment. Fourth, there is a lack of studies examining long-term outcomes. The duration of treatment was rarely over five years, while most studies followed transgender individuals for less than two years. Since many transgender individuals initiate lifelong treatment as minors and hormone therapy can take up to five years to see the maximum effect, the absence of long-term outcomes is concerning. Therefore, future longitudinal studies should aim to follow patients for a minimum of five years to better assess long-term outcomes. Fifth, there is a lack of research examining individuals who cease treatment. It is important to capture their trajectory of development after terminating treatment to ensure that minors can achieve their developmental milestones. Sixth, only 57% of the studies that used both puberty suppressors and GAHT reported distinct effects of each treatment, and most examined skeletal outcomes. Future studies should examine the distinct and combined effects of puberty suppressors and GAHT for psychological and cardiovascular outcomes.
Conclusion
The outcomes examined provide insight into potential risks and benefits that transgender individuals may experience long-term. Further research is needed to examine the long-term psychological and physical outcomes, especially among adolescents progressing into later adulthood. Future studies should investigate these outcomes with longer treatment duration and within larger groups of diverse transgender individuals. Studies measuring psychological, cardiovascular, and skeletal outcomes should account for potentially confounding variables and separate outcomes by gender identity. Due to the limitations mentioned, there is limited evidence within previous studies to determine with certainty the long-term effects of hormone treatment in adolescents. The review indicates that hormone therapy may improve psychological symptoms and alter cardiovascular and skeletal health profiles. Moreover, clinically significant levels of cardiovascular and skeletal measures may increase an individual’s risk for pathogenesis and should be continually monitored throughout treatment. However, there is insufficient data to conclude the adverse cardiovascular and skeletal events among transgender individuals taking hormone therapy. This area of research urgently needs high-quality longitudinal studies as the prevalence rate of GD and hormone medication prescriptions increases.
Supplemental Material
Supplemental material for this article available online.
Footnotes
Authors’ Contribution
HGP: Conceptualization, data curation, investigation, and writing.
AJD: Methodology, project administration, editing, supervision, and verification.
BLP: Editing and verification.
Data Availability Statement
The data that support the findings of this study are not publicly available due to participant confidentiality.
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval
This research did not involve human subjects and was therefore exempt from ethical approval. Data was obtained from publicly available sources and did not require IRB approval.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
All data were obtained from publicly available, de-identified sources and did not involve direct interactions with human participants. Therefore, informed consent was not required for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.