Abstract
Background:
Insulinomas are the most common functional pancreatic neuroendocrine tumors (pNETs). Minimally invasive approaches like endoscopic ultrasound-guided radiofrequency ablation (EUS-RFA) and ethanol ablation (EUS-EA) are emerging treatment options for small insulinomas (<2 cm).
Objectives:
To assess the safety, efficacy, and long-term outcomes of EUS-RFA and EUS-EA in treating small pancreatic insulinomas.
Design:
A retrospective, single-center study of nine patients diagnosed with insulinomas and treated with EUS ablation methods.
Methods:
Nine lesions (mean size 11 mm; range: 6–19 mm) were treated with EUS-RFA (n = 7) or EUS-EA (n = 2). EUS-RFA procedures had a mean ablation time of 31 s (range: 17–69 s), while EUS-EA used a mean ethanol volume of 1.4 ml and 0.5 ml for respective patients. All patients were followed up radiologically with CT and/or EUS for a median of 25 months (range: 6–46 months).
Results:
All patients had immediate hypoglycemia relief after a single treatment. The median clinical follow-up was 40 months (range: 22–60 months), with all patients remaining asymptomatic. Complete radiologic regression was observed in six patients. Two patients had minor adverse events; no severe complications occurred.
Conclusion:
EUS-guided ablation (RFA or EA) is a safe and effective treatment for small pancreatic insulinomas, providing symptom relief and radiologic regression. Further studies are needed to evaluate long-term efficacy and recurrence rates.
Introduction
Insulinomas are the most prevalent functional pancreatic neuroendocrine tumors (pNETs), characterized by unregulated insulin secretion that can result in debilitating hypoglycemia. Although these tumors are rare, with an estimated incidence of 1–4 per million in the general population, their clinical impact is significant due to the potential for life-threatening hypoglycemic events. 1
Surgical resection remains the gold standard of curative treatment for insulinomas, with a reported high success rate. However, this procedure may be associated with complications, and alternative treatments are sought for patients who are poor surgical candidates due to age or comorbidities. 2
Recent advances in endoscopic techniques have introduced endoscopic ultrasound (EUS)-guided ablation methods, such as ethanol ablation (EUS-EA) and radiofrequency ablation (EUS-RFA). These approaches offer a minimally invasive option, leveraging the precision of EUS to target and ablate the tumor while preserving surrounding pancreatic tissue.
EUS-EA induces necrosis by directly injecting ethanol into the lesion, while EUS-RFA uses thermal energy to achieve coagulative necrosis. Both techniques have shown potential for symptom control and tumor reduction.3,4
This study aims to evaluate the safety and long-term efficacy of EUS-guided ablation for pancreatic insulinomas < 2 cm in size, focusing on clinical and radiological outcomes.
Methods
Study design and population
This retrospective study included patients diagnosed with pNETs at a single center (Endoscopy Unit, Hospital of the Ministry of Interior and Administration, Szczecin, Poland) between 2011 and 2023. Patients were identified based on histological confirmation of G1 pNETs through EUS-guided fine-needle biopsy. Among 275 patients diagnosed with pNETs during this period, 34 underwent EUS-guided ablation (30 with RFA and 4 with EA). For this analysis, we included nine adult patients with insulinomas meeting the following criteria (Figure 1): (1) confirmed diagnosis of insulinoma with clinical symptoms and visible lesion on imaging (CECT); (2) tumor size ⩽ 2 cm with no evidence of lymph node involvement or distant metastases; (3) unsuitability for or refusal of surgery; (4) informed consent obtained prior to the procedure. Exclusion criteria included tumor size > 2 cm, contraindications to endoscopy or RFA.
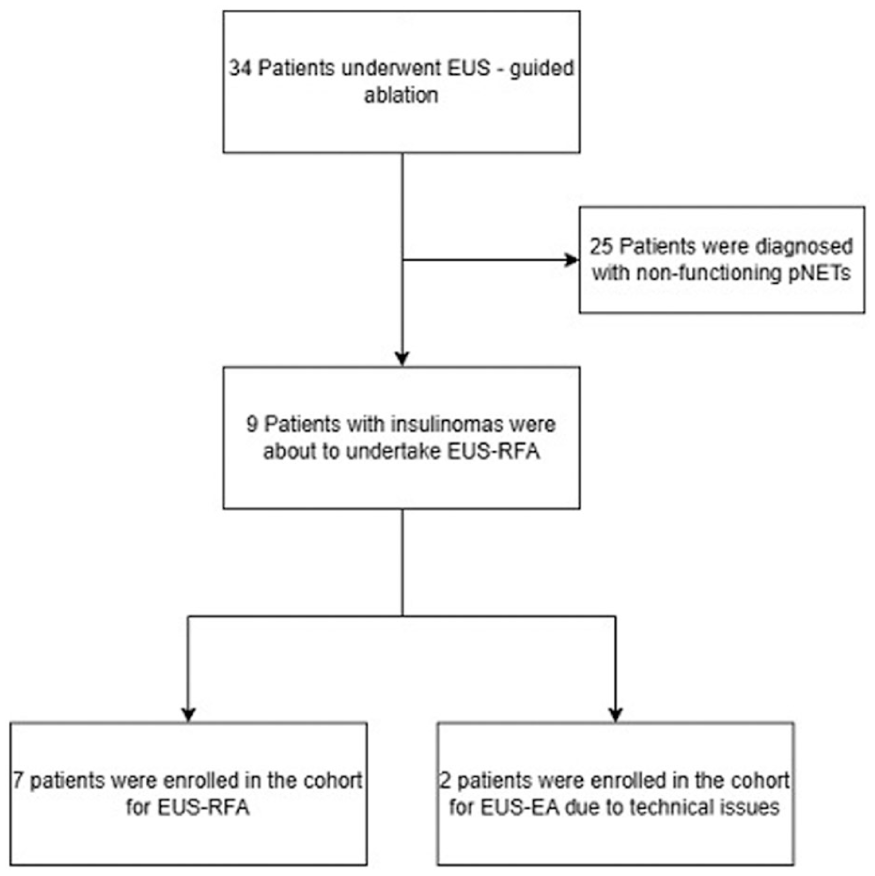
Flow chart of patients with pNETs.
Procedural details
All procedures were performed by two experienced endoscopists under deep sedation. For EUS-RFA, a monopolar electrode needle with an active tip (5–10 mm) was used in conjunction with an internal cooling system (EUSRA system, Taewoong, Combo VIVA™ generator STARmed), Gyeonggi-do, South Korea. Under EUS guidance, the electrode needle was advanced into the lesion, maintaining a safe distance from adjacent structures. Particular care was taken to identify the pancreatic duct to avoid thermal injury. Particular care was taken to identify the pancreatic duct to avoid thermal injury. The needle tip was positioned at the far end of the tumor. RFA was performed at 30–50 W until the lesion appeared as a hyperechoic zone, indicative of coagulative necrosis (Figure 2). In initial cases, higher power (50 W) was used, later reduced to 30 W with longer application times for safety.

EUS-RFA ablation of 7 mm pNET. (a) Pretreatment: in the body of the pancreas, a well-confined, round, hypoechoic tumor, (b) rich vascularization of the tumor in the EUS-DFI imaging, (c) Intraprocedural: EUSRA 5 mm needle advanced in the lesion with the tip positioned at the far end of the tumor (white narrow), (d) Intraprocedural: RFA ablation seen as hyperechoic zone, and (e) after ablation: heterogeneous region of necrosis with mixed echogenicity.
EUS-EA was performed using a 22-gauge fine-needle aspiration (FNA) needle (EZShot, Olympus, Tokyo, Japan). Absolute ethanol was injected into the lesion under EUS guidance until the hyperechoic blush extended throughout the tumor. (Figure 3) EA was selected only in technically difficult locations where RFA was unsafe.
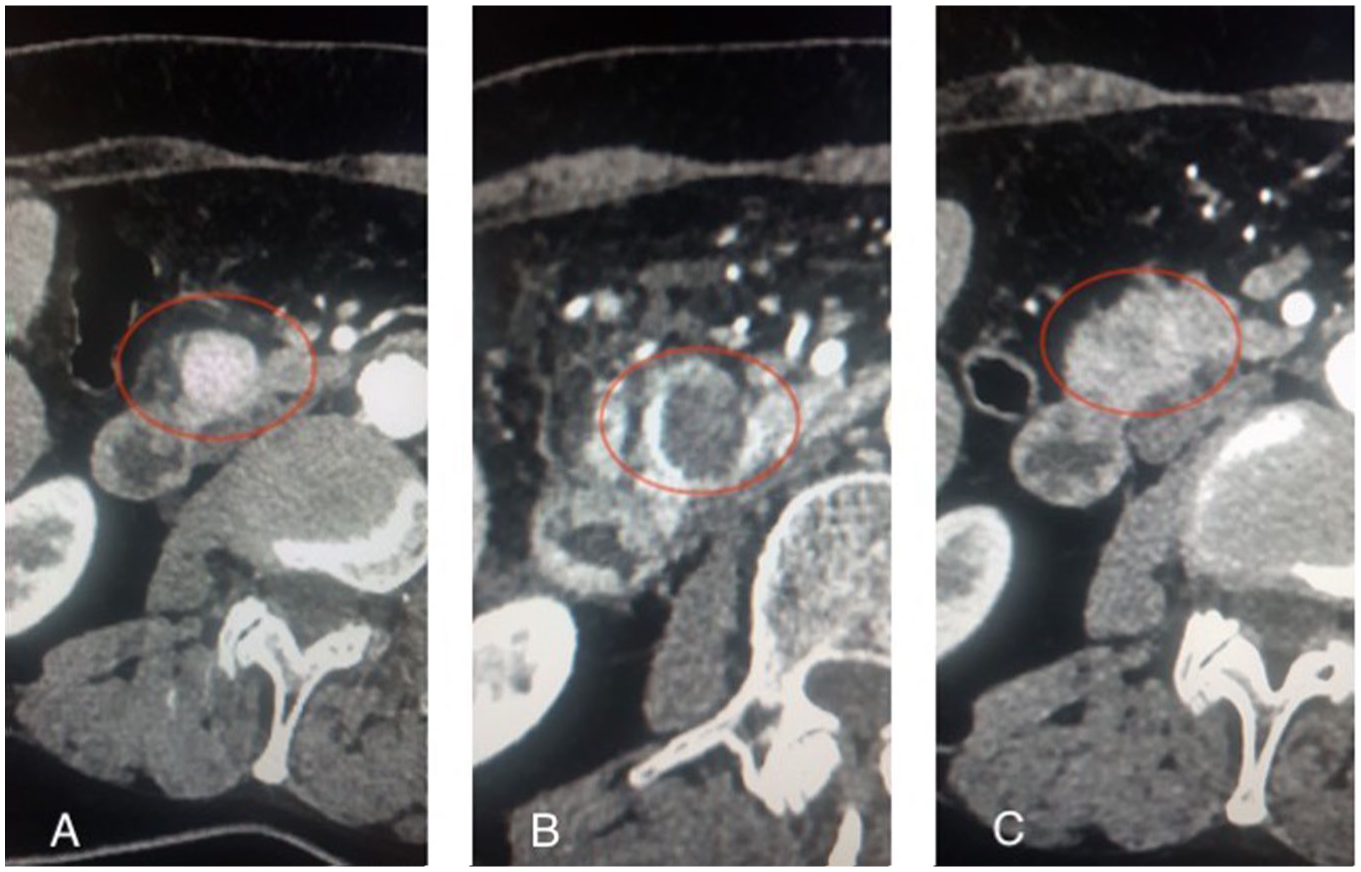
CT scans of a patient with pancreatic insulinoma. (a) Pretreatment: in the head of the pancreas, a well-confined, arterially enhancing, hyperdense tumor, (b) Two days after EUS-RFA: hypodense, non-enhancing area of necrosis surrounded by a hyperdense stroma. Increased density of surrounding adipose tissue and fluid in the course of pancreatitis after procedure, and (c) after 4 months: a small hypodense lodge at the tumor site, with a faintly enhancing, irregular area of residual tumor visible at its periphery.
Both techniques were directly followed by a post-procedure EUS evaluation to assess treatment completeness, with additional ablation sessions performed for residual lesions.
After the procedure, all patients received anti-inflammatory prophylaxis (rectal Diclofenac 100 mg), were fasted for 12 h and received intravenous fluids (30–40 ml/kg/day) with analgesic (No-spa and Pyralgin); no antibiotic prophylaxis was administered; patients were monitored for adverse events (AEs) during a 48–72 h hospital stay.
Outcome measures
The primary endpoint was relief of hypoglycemia-associated symptoms within 3 months of the procedure. Secondary endpoints included complete radiological response, defined as the absence of residual tissue on imaging (contrast-enhanced EUS (CE-EUS) and/or contrast-enhanced computed tomography (CECT)) (Figure 3), and the occurrence of moderate and severe AEs. Follow-up included CECT and/or CE-EUS with Sonovue contrast (Bracco, Milan, Italy) performed at 3 months and subsequent intervals based on clinical need. In case of any contrast enhancement within the lesion, another ablation procedure was performed. If the complete regression was described, meaning absence of enhancing tissue next follow-up was planned after 12 months. Patients were also controlled by endocrinologists.
AEs were reported during 3 months after the procedure and were classified in accordance with AGREE classification. 5 Abdominal pain and mild increase of amylases were not recorded.
Statistical analysis
Descriptive analysis was summarized and demonstrated with medians (ranges) for continuous variables, and frequencies (percentages) for categorical variables. The reporting of this study conforms to the STROBE statement (Supplemental Material). 6
Results
Patient characteristics
Nine patients (seven females, mean age 49 years) with monofocal pancreatic insulinomas (mean size 11 mm; range 6–19 mm). Five lesions were located in the pancreatic head, and four in the neck. All insulinomas were histologically confirmed as NETs G1 (Ki-67 Index < 3%), and all patients experienced symptomatic hypoglycemia. Seven of nine patients underwent EUS-RFA, and two patients underwent EUS-EA due to the difficult location of the lesion. Four of seven lesions treated with EUS-RFA were ablated with a power setting of 50 W, and three of seven with 30 W. The details of enrolled patients are reported in Table 1.
Characteristics of nine pancreatic insulinoma patients treated with EUS-RFA/EA.
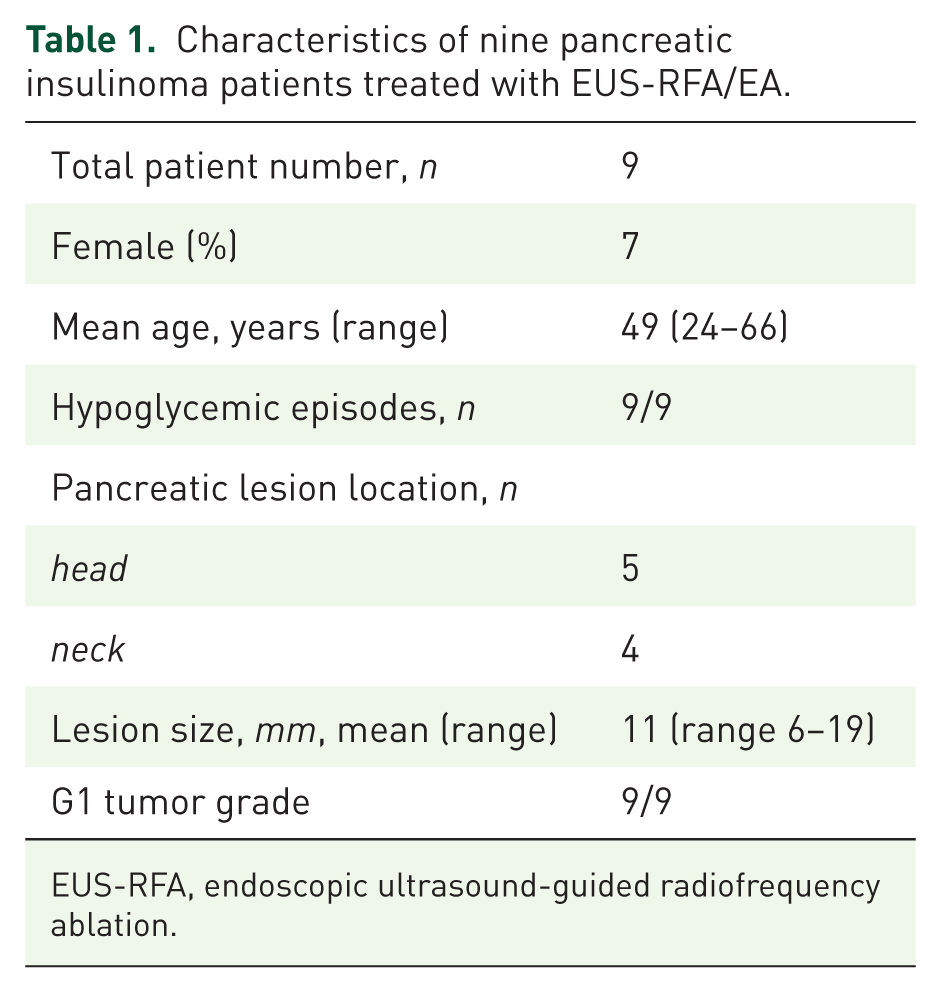
EUS-RFA, endoscopic ultrasound-guided radiofrequency ablation.
Clinical and radiological outcomes
Outcome of EUS-RFA and EUS-EA per patient is reported in Table 2. All patients achieved immediate relief from hypoglycemia following a single treatment session. Median follow-up was 40 months (range, 22–60 months); Radiological median follow-up was 25 months (range, 6–46 months), with six patients achieving complete lesion regression. Three patients required additional ablation sessions due to partial regression. The mean number of sessions was 1.5 (range 1–3). During follow-up, no patient experienced a relapse of symptomatic hypoglycemia.
Outcome of EUS-RFA and EUS-EA per patient.

CECT, contrast-enhanced computed tomography; CR, complete response; EA, ethanol ablation; EUS, endoscopic ultrasound; IR, incomplete response; NA, not applicable; RFA, radiofrequency ablation.
Safety
Two patients experienced AEs. One patient developed a localized hematoma, which was managed conservatively (Grade II), while another had upper gastrointestinal bleeding, successfully treated endoscopically (Grade III). No delayed or severe complications were observed.
Discussion
Management of functional pancreatic neuroendocrine tumors, particularly insulinomas, poses unique challenges. Although surgical resection has curative potential, its association with significant morbidity—including rates of pancreatic fistula (up to 58%), delayed gastric emptying (up to 18%), and postoperative bleeding (6%)—emphasizes the need for safer, less invasive alternatives. 7 EUS-guided ablation techniques (EUS-RFA and EUS-EA) were thus developed and assessed recently as a new, non-surgical, effective, and safe therapeutic option for patients with pancreatic insulinomas. In the present study, we retrospectively reported nine cases of patients with insulinomas treated with EUS-RFA or EUS-EA. To our knowledge, this is one of the largest long-term series of patients with insulinomas focusing on both EUS-guided RFA and ethanol ablation in the literature so far.
There are multiple studies describing the application of EUS-guided ablation methods in pancreatic insulinomas, most of them with a very limited number of patients.1,3,4,8,9-13 Our study contributes to a growing body of evidence supporting EUS-guided ablation techniques, specifically RFA and EA, as viable options for patients who are not surgical candidates. Among our cohort, hypoglycemia was promptly resolved in all cases after a single session, aligning with reported clinical success rates of 95.5%–100% in previous studies.2,9,10 Radiological outcomes were similarly encouraging, with complete regression observed in 67% of cases. Notably, two patients declined further sessions after symptom relief, which limited the rate of complete regression. Moreover, the stringent definition of complete radiological response—absence of any residual tissue enhancement—may also affect this rate. Meta-analysis involving 292 patients with pancreatic NETs (functioning and non-functioning) confirms high complete radiological response of 87.1%, partial response of 11.4% and the pooled clinical response rate for functional pNENs of 94.9%, with no heterogeneity. 14
Safety is paramount when considering ablation for insulinomas. Our findings corroborate previous reports of a favorable safety profile for EUS-guided ablation, with no severe complications observed. 2 The two moderate AEs noted in this series were managed conservatively without long-term sequelae. These findings are in accordance with previous literature, supporting EUS-guided ablation as an alternative to surgery.
The most common complications during the procedures of ablation reported in the literature are bleeding, local abdominal pain, pseudocyst formation, and acute pancreatitis. Furthermore, Crinò et al. 10 demonstrated that EUS-RFA is associated with significantly lower adverse event rates (18%) compared to surgery (61.8%), further supporting its role in managing insulinomas. Jurgensen et al. 12 compared EUS-EA versus surgical resection of insulinomas and reported higher complication rate in the surgery group (54% vs 11%, p = 0.026). The above-mentioned meta-analysis reports pooled incidence of AEs of 20.0%; most AEs were mild to moderate in grade with the pooled incidence of severe AEs of 0.9%, which confirms the safety of the procedure. 14
Finally, long-term follow-up data are essential to assess recurrence and the potential for malignant transformation of residual or recurrent lesions. While functional pNETs are typically indolent, achieving complete ablation may reduce recurrence risk and the need for repeated interventions.
There is no head-to-head, prospective comparison of EUS-guided ablation methods. EUS-RFA seems to be better validated and studied than EUS-EA, so it was our method of choice while referring the patient for an ablation procedure. Nevertheless, technical challenges remain, particularly in lesions located deeply in the head or uncinated process of the pancreas or near critical structures such as the mesenteric vessels, pancreatic duct, or bile ducts. In such cases, EA may provide a practical alternative to RFA due to the nitinol FNA needle precision of ethanol injection. For this reason, EA was used only in two cases where lesion location (head/uncinate process near vessels or ducts) made RFA technically unsafe. The absence of standardized protocols for procedural settings, including power settings, ablation time, and ethanol volumes, highlights a critical gap in the current literature. The power setting was set up at the discretion of the endoscopist. Specifically, in the first cases, a higher power (50 W) was used to treat the lesion. Thereafter, the applied wattage was reduced, and concurrently, the time of RF application per session increased to potentially optimize effectiveness, better control extension of the treated area, and minimize the damage of pancreatic tissue and the AEs. This was in line with previous studies, in which a higher power setting was noted to be associated with smaller ablation zones and with a major risk of procedure-related AEs. 15 However, the superiority of 30 W over 50 W has not been proven yet. Future prospective studies are needed to establish consensus guidelines and optimize procedural outcomes.
Our study has several obvious limitations, which deserve discussion. First, the retrospective design limits the ability to control for confounding variables and may introduce bias in patient selection and outcome assessment. Second, the small sample size and absence of formal sample size calculation restrict the generalizability of the findings and preclude detailed subgroup analysis. Larger, multicenter studies are necessary to confirm the findings and establish external validity. Third, all procedures were performed by experienced operators in a specialized center, which may not reflect outcomes in less experienced hands or in centers with differing levels of technical resources. The outcomes may, therefore, represent an idealized scenario and may not be universally reproducible. Fourth, the lack of standardized follow-up protocols represents a notable limitation. Radiological follow-up varied, with some patients undergoing both CECT and CE-EUS, while others were assessed with only one modality. Although no discordances between modalities were reported, the variability in follow-up methods could affect the assessment of residual disease and treatment efficacy.
Conclusion
EUS-guided RFA and EA offer promising outcomes for the management of pancreatic insulinomas, combining efficacy with a favorable safety profile. Standardization of procedural protocols and extended follow-up studies are necessary to fully integrate these techniques into clinical practice.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745251401315 – Supplemental material for EUS-guided radiofrequency and ethanol ablation of pancreatic insulinomas: a single-center experience
Supplemental material, sj-docx-1-cmg-10.1177_26317745251401315 for EUS-guided radiofrequency and ethanol ablation of pancreatic insulinomas: a single-center experience by Katarzyna Kozlowska, Katarzyna Monika Pawlak, Kaja Kozlowska, Sebastian Jedruszek, Wiktoria Bosy-Gasior, Nina Hsino-Sito and Anna Wiechowska-Kozłowska in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
