Abstract
Background:
Bile leakage following hepatectomy is a challenging issue that poses difficulties for hepatobiliary surgeons. Endoscopic treatment is the primary method for managing this complication, although its effectiveness is still uncertain.
Objectives:
The aim of this study was to assess the effectiveness and risk factors of endoscopic treatment for bile leakage after hepatectomy.
Design:
A retrospective cohort study.
Methods:
A retrospective study was performed on 15 patients who experienced bile leakage following hepatectomy and were successfully treated with endoscopic retrograde cholangiography (ERCP). The primary focus was to examine how factors like the site of bile leakage, the position of biliary drainage, and late-set leakage impact early clinical success (ECS).
Results:
Out of the 15 cases, 10 successfully achieved ECS through endoscopic treatment. The highest success rate (80.0%, 4 out of 5) was seen when biliary drainage was performed by bridging, while drainage placed near the leakage site had a success rate of 75.0% (6 out of 8). The best outcomes occurred when bile leakage was located in the tertiary or higher branch bile ducts, with an 83.3% success rate (5 out of 6). Additionally, early-onset leakage had a higher ECS success rate compared to late-onset bile leakage (75.0% (6/8) vs 57.1% (4/7)). Among the 15 cases, 12 involved extensive liver resections of more than two segments, 14 included segments 7 or 8, 11 had received preoperative adjuvant therapy, and 9 had undergone transarterial chemoembolization (TACE). In cases with late-onset bile leakage, 6 out of 7 (85.7%) had undergone TACE. Bile leakage caused by TACE was more likely to be accompanied by bile duct-bronchial fistula, and the healing process took significantly longer.
Conclusion:
Endoscopic management of bile leakage following liver resection is both safe and effective, making it a preferred first-line treatment. Improved results can be obtained by placing a stent to bridge and drain near the leakage site. Extensive liver resection, resection involving segments 7 or 8 of the liver, and preoperative adjuvant therapies are significant risk factors for bile leakage, particularly TACE, which may lead to bile duct-bronchial fistulas and extended recovery periods.
Plain language summary
Hepatectomy is one of the main methods for treating benign and malignant liver diseases. However, bile leakage occurs in some patients after partial hepatectomy. Endoscopic retrograde cholangiopancreatography (ERCP) is the main treatment for post-hepatectomy bile leakage (PHBL). Due to the lack of large-sample clinical studies, the success rate of ERCP is not clear. The factors affecting the success of endoscopic treatment are also not clear. This study retrospectively analyzed the clinical data of 15 patients with PHBL successfully treated by ERCP, and explored the role of endoscopic treatment in the treatment of PHBL and the factors affecting the success rate. The results showed that endoscopic treatment of PHBL was safe and effective and could be used as the first-line treatment for PHBL.
Introduction
Hepatectomy is one of the principal approaches for treating both benign and malignant liver disorders. The surgical techniques encompass anatomical liver resection, non-anatomical liver resection, liver segment and liver lobe resection, and so forth. With the advancement of surgeons’ skills and the utilization of energy devices and fluorescence imaging equipment, the implementation of hepatectomy has become more extensive and precise, reducing the perioperative mortality rate, blood loss, and hospital stay. Despite such progress, certain complications still inevitably occur after hepatectomy. It is reported that 4%–17% of patients undergoing hepatectomy have posthepatectomy biliary leakage (PHBL), 1 which remains one of the most common complications following hepatectomy. The International Study Group of Liver Surgery (ISGLS) defines posthepatectomy biliary leakage as a ratio of bilirubin in the drainage fluid to serum bilirubin of ⩾3 at day 3 after resection or later; in addition, those requiring percutaneous drainage or reoperation intervention due to biliary peritonitis are also classified as PHBL. 2
According to the influence of bile leakage on clinical management, the ISGLS categorizes PHBL into grades A, B, and C. Patients with grade A PHBL do not need to modify the existing clinical treatment approaches. Patients with grade B PHBL require additional diagnostic or interventional surgeries (such as puncture drainage or endoscopic treatment), but do not need laparotomy. Moreover, patients with grade C PHBL need re-laparotomy. 2 Postoperative bile leakage usually results in delayed abdominal drainage, a prolonged hospital stay, and high costs. In severe cases, it can also cause abdominal sepsis and may even lead to death. Besides affecting the perioperative prognosis, bile leakage may also have an impact on the long-term prognosis of patients undergoing surgical treatment for malignant diseases.3–5
Endoscopic biliary drainage is one of the main treatment approaches for grade B bile leakage. For bile leakage resulting from extrahepatic bile duct injury, such as that after cholecystectomy, the overall outcome of placing biliary stents or nasobiliary drainage under Endoscopic retrograde cholangiopancreatography (ERCP) is relatively favorable, with an effective rate ranging from 87% to 100%. 6 However, regarding bile leakage after hepatectomy, due to the scarcity of large-sample clinical studies, the efficacy of endoscopic treatment remains unclear. The location and severity of bile leakage, the type of bile duct injury, and the presence of associated stenosis are all factors influencing the success of treatment. Concerning the risk factors for bile leakage, the conclusions of existing research reports are not uniform. The extent of hepatectomy, BMI, gender, diabetes, and whether there is neoadjuvant chemotherapy may all be factors affecting bile leakage. With the current alterations in the treatment modality of liver tumors, whether preoperative local interventional therapy and systemic chemotherapy, targeted and immunotherapy increase the incidence of bile leakage and affect healing requires further exploration. Therefore, this study preliminarily examines the role of endoscopic treatment in the management of bile leakage after hepatectomy by analyzing the results of endoscopic treatment of 15 cases of bile leakage after hepatectomy in our department and related predictive factors, in combination with literature learning.
Patients and methods
Patients
The research subjects were 17 patients with bile leakage after hepatectomy who underwent endoscopic treatment.
This is a retrospective study on bile leakage after hepatectomy at the Sixth Affiliated Hospital, Sun Yat-sen University and the Second Affiliated Hospital of Guangzhou Medical University from July 2020 to August 2024. Among the 76 patients, those meeting the following conditions were included in this study: (1) male or female aged 18–75 years, (2) hepatectomy was performed, including hemihepatectomy, lobectomy, segmentectomy, or non-anatomical hepatectomy, (3) the daily bile drainage volume of the abdominal drainage tube exceeds 200 milliliter (ml), (4) providing signed informed consent, and (5) meeting criteria for posthepatectomy biliary leakage: (a) a drain bilirubin to serum bilirubin ratio ⩾ 3 at day 3 after resection or later, (b) the need for radiologic or operative intervention resulting from biliary collections or bile peritonitis. Exclusion criteria: (1) bile leakage that occurs after non-hepatectomy, such as after cholecystectomy or common bile duct exploration, (2) undergoing non-endoscopic treatment, (3) lost to follow-up. This study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational cohort studies (Supplemental Material).
Endoscopic treatment
This study adopted ERCP as the preferred approach for treating bile leakage after hepatectomy. ERCP was carried out using a TJF-260 V endoscope (Olympus). In most instances, midazolam and pethidine hydrochloride were utilized for sedation and analgesia, and the position was the left lateral decubitus. The cases included in this study were all completed by physicians who had performed more than 2000 ERCP operations. The operator initially conducted angiography to determine the location and type of bile leakage. All patients underwent a minor incision of the duodenal papilla, and the guide wire was selectively advanced to the target bile duct to determine the drainage method and the diameter and length of the drainage tube. The changes in the volume of the abdominal drainage tube were observed after the operation, and anti-infection treatment was administered.
Bile leakage location
In this study, based on the location of bile leakage, it was classified into ① hilar bile duct (primary bile duct); ② hepatic lobar bile ducts (secondary bile ducts); ③ Hepatic segment and sectional bile duct (third and above branches); Bile leakage types were divided into: lateral wall leakage and transectional leakage, with or without bile duct stricture. In this study, the drainage locations under endoscopy were classified into five situations (Figure 1 and Table 1): ① Lateral wall leakage, bridgeable; ② Lateral wall leakage, non-bridgeable, with the drainage placed in the bile duct at the same level; ③ Peripheral transectional leakage, featuring a long distance from the section to the bifurcation point of the upper-level bile duct, and the drainage was placed near the leakage orifice or in the cavity; ④ Peripheral transectional leakage, with a short distance from the section to the bifurcation point of the upper-level bile duct, and the drainage was placed in the bile duct near the leakage orifice; ⑤ Regardless of lateral wall or transectional leakage, the drainage could only be placed in the proximal bile duct of the upper level.
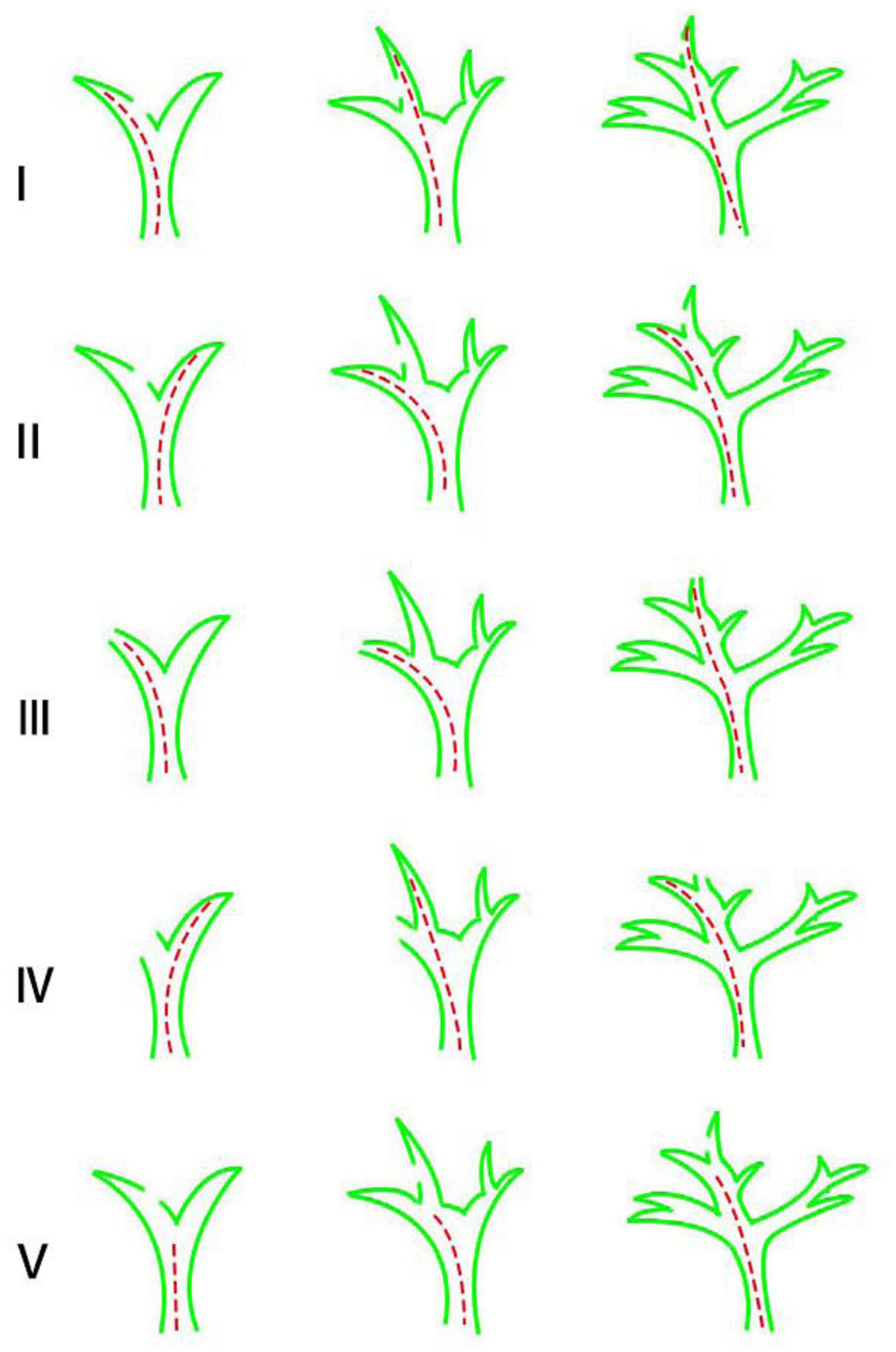
Classification of endoscopic drainage locations after hepatectomy for bile leakage.
The classification system for biliary drainage.

The definition of early clinical success (ECS) 7 was as follows: all the external biliary drainage tubes, including the endoscopic nasal biliary drainage (ENBD) tube, were successfully removed following the endoscopic intervention, and no recurrence of bile leakage occurred within 1 month.
Statistical analysis
Values for continuous variables were expressed as means ± SD. For the univariate analysis, categorical data were analyzed by the χ2 test. Differences between variables were analyzed by Student’s
Results
Patient characteristics
From July 2020 to August 2024, there were 602 patients who underwent liver resection. Among them, 76 cases had postoperative bile leakage, 18 cases received ERCP treatment, 1 case was lost to follow-up, and 17 cases were included in this study (Figure 2). Among them, 15 cases were successfully placed biliary drainage, and 2 cases failed, 1 had duodenal bulb stenosis, through which the duodenoscope could not pass, and another failed in papillary cannulation. Of these two patients with failed ERCP, one chose to continue with conservative treatment, and one opted for percutaneous transhepatic cholangial drainage (PTCD) biliary drainage.

Flowchart showing participant selection and exclusion.
This article analyzed 15 patients with bile leakage after hepatectomy who successfully underwent endoscopic biliary drainage (Table 2). The average age of the patients at the time of surgery was 57.8 years (38.0–82.0), including 10 males and 5 females, and the average BMI value was 23.5 kg/m2 (18.4–26.2). There were 12 cases of primary liver cancer and three cases of colorectal cancer liver metastases (CRLM). Bile leakage comprised eight cases of Early-onset leakage (<2 weeks after surgery) and seven cases of Late-onset leakage (⩾2 weeks after surgery). 8 Among the patients with early-onset leakage, four had abdominal bile accumulation and underwent puncture and drainage. All patients with Late-onset leakage underwent abdominal puncture and drainage. According to the ISGLS definition, this was equivalent to grade B bile leakage, and all received endoscopic treatment. The methods of hepatectomy included left or right hemihepatectomy, extended right hemihepatectomy, right anterior lobe resection, central hepatectomy, combined liver segment resection, and non-anatomical hepatectomy. Regarding the extent of liver resection, 10 cases had a liver resection range of ⩾3 liver segments, and 5 cases had a liver resection range of <3 liver segments. There were no significant differences on baseline characteristics between endoscopic-treatment success and failure groups (Table 3).
Baseline characteristics of the patients who received endoscopic treatment.

CRLM, colorectal cancer liver metastases; HCC, hepatocellular carcinoma.
Baseline characteristics of patients undergoing endoscopic treatment.

Student’s
Chi-squared test.
CRLM, colorectal liver metastases; HCC, hepatocellular carcinoma; SD, standard deviation; TACE, transarterial chemoembolization.
The effect of endoscopic treatment
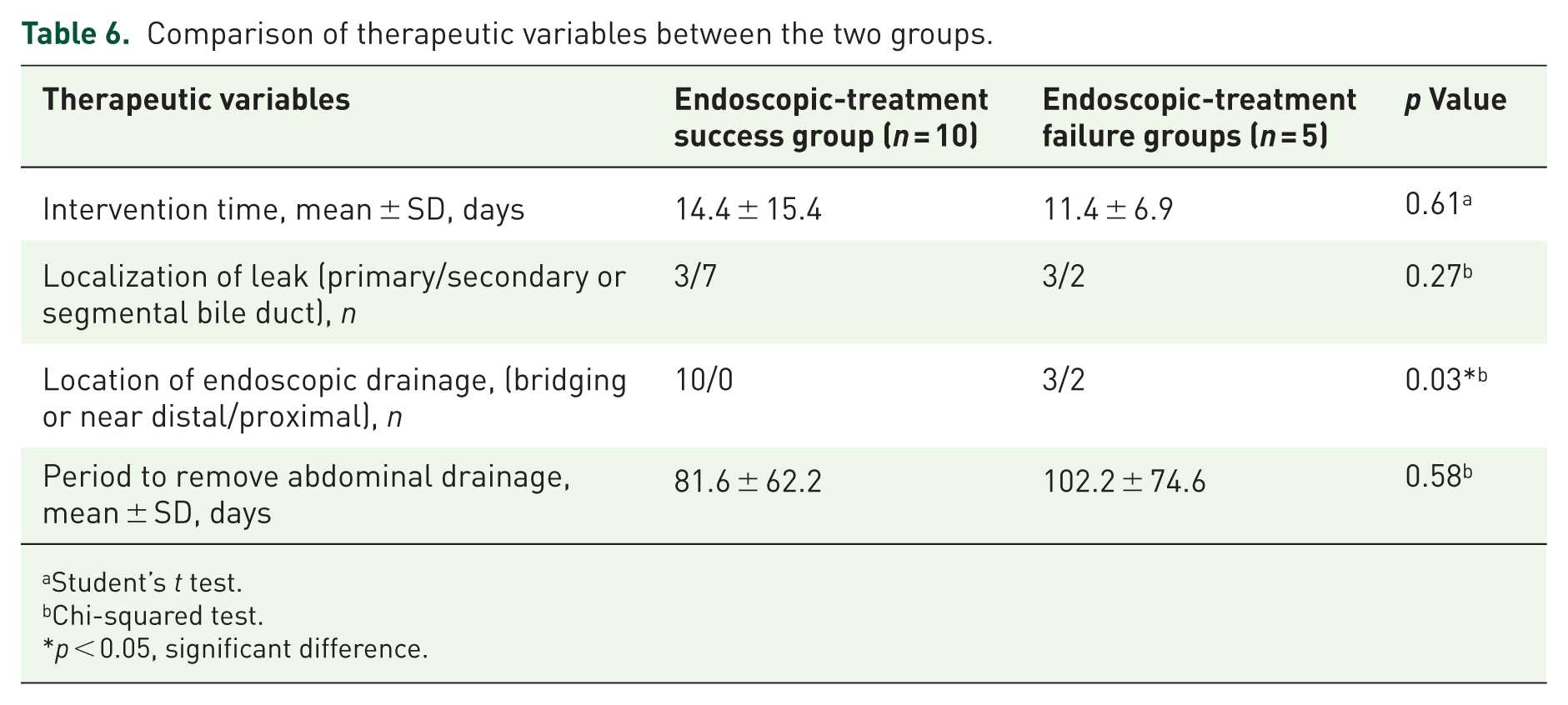
About the therapeutic effect, 10/15 (66.7%) cases achieved early clinical success through endoscopic treatment (Table 4). The most favorable outcome was obtained in those with biliary drainage bridging, with a success rate of 4/5 (80.0%). The success rate was 6/8 (75.0%) for drainage near the leakage site, while the effect was the poorest for drainage at the proximal end of the leakage site, with a success rate of 0/2 (00.0%). The best result was witnessed when the bile leakage was located in the tertiary and above branch bile ducts, with a success rate of 5/6 (83.3%), followed by that in the hepatic lobe bile ducts (secondary bile ducts), with a success rate of 2/3 (66.7%), and the success rate was 3/6 (50.0%) for bile leakage in the primary bile ducts (Table 5). The endoscopic-treatment success group had a shorter time to remove the abdominal drainage tube. The success rate was higher when the bile leakage was located in secondary or segmental bile duct, or when the bile leakage was bridged or the drainage tube was located near the leakage opening (Table 6). Univariate analysis showed that four factors might affect the success rate of treatment (based on a
Endoscopic interventions and outcome.

ECS, early clinical success.
The key therapeutic variables.

Comparison of therapeutic variables between the two groups.
Student’s
Chi-squared test.
Univariate analysis of factors suspected to affect endoscopic success.
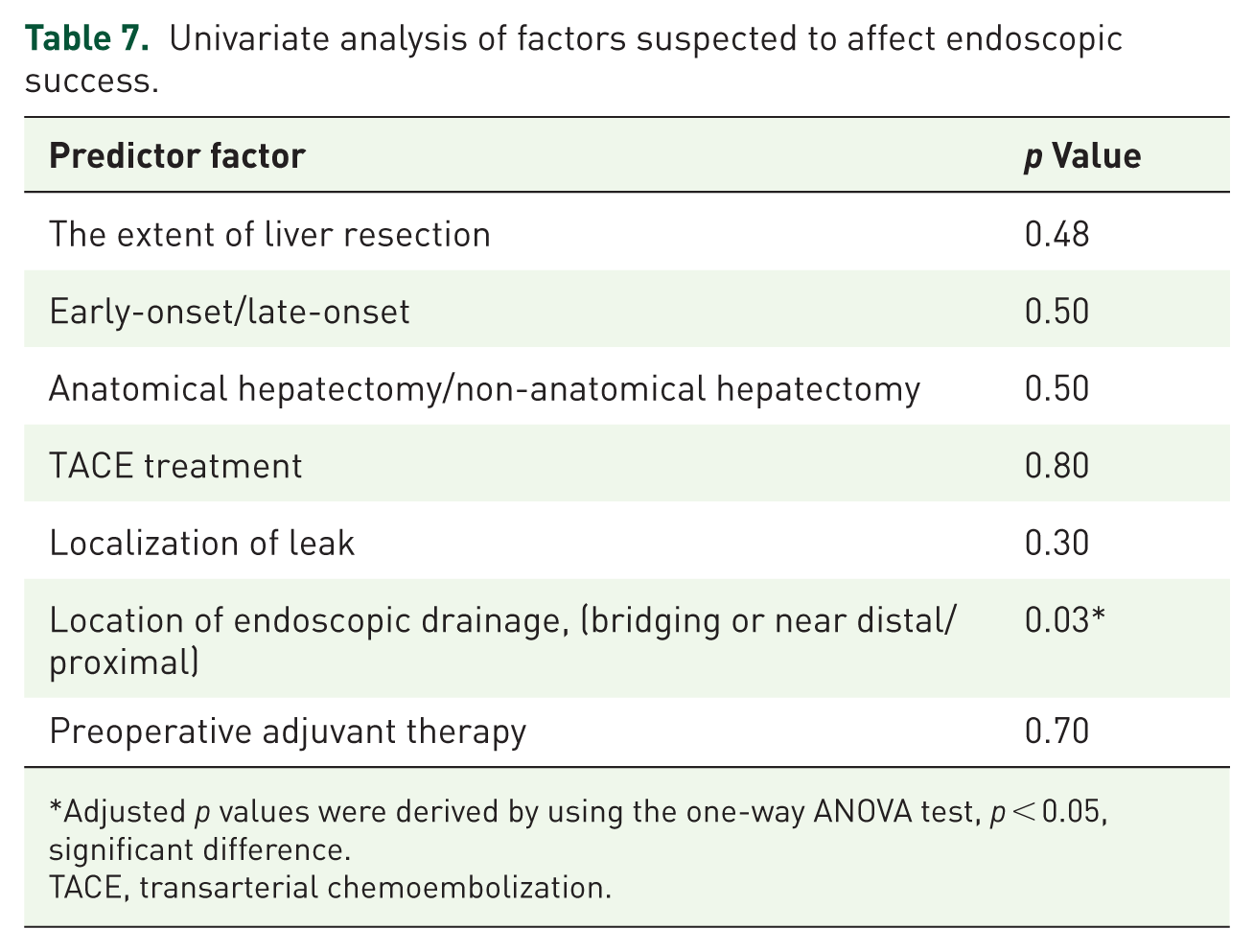
Adjusted
TACE, transarterial chemoembolization.
Early-onset/late-onset
In addition, we compared the effects of different factors on early-onset and late-onset bile leakage. Among the 15 patients with bile leakage, 8 cases were of early-onset leakage and 7 cases were of late-onset leakage. Based on our data, the healing time of endoscopic treatment for early-onset leakage was shorter, averaging 61.0 ± 23.9 days, while that for late-onset leakage was longer, averaging 119.9 ± 76.5 days. The ECS rate of early-onset leakage was superior to that of late-onset leakage (6/8 (75.0%) vs 4/7 (57.1%)). By comparing and analyzing the characteristics of patients in the early-onset leakage group and the late-onset leakage group, we found that TACE, Involving liver resection of segment S7 or S8, low BMI, and anatomical liver resection might be high-risk factors for late-onset bile leakage (
Factors associated with early-onset and late-onset bile leakage.
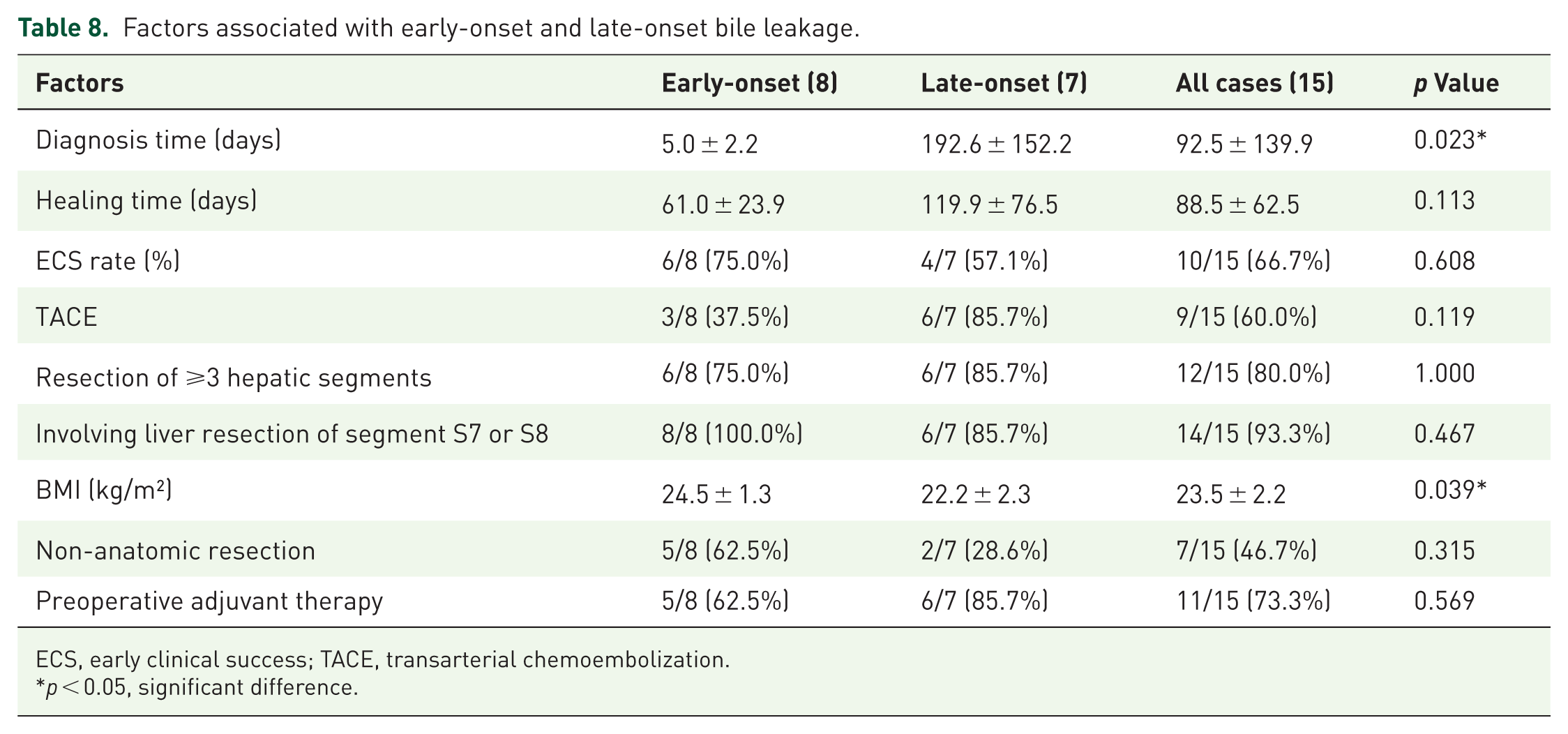
ECS, early clinical success; TACE, transarterial chemoembolization.
Analysis of TACE
With the extensive implementation of conversion therapy for liver tumors and the previously reported influence of TACE on the blood supply of bile ducts, we examined the effect of TACE on bile leakage. Among the 15 patients, 9 cases had received TACE, particularly among the patients with delayed bile leakage, 6/7 (85.7%) had undergone TACE. Among the six cases, five cases had received preoperative conversion TACE, and three cases developed bile leakage after TACE for postoperative tumor recurrence. Among them, two cases had undergone TACE both before surgery and after recurrence, and the healing time of bile leakage in these two cases was more longer. The average healing time of bile leakage patients who had undergone TACE was 118.3 ± 64.4 days, while the average healing time of non-TACE-related bile leakage patients was 43.7 ± 13.7 days (Table 9).
Effect of TACE on healing of biliary leakage.

TACE, transarterial chemoembolization.
A case of bile leakage combined with bronchial leakage after hepatectomy
Biliary bronchial fistula frequently occurs after hepatectomy accompanied by bile leakage. Bile accumulates beneath the diaphragm and fails to be drained promptly, leading to an abnormal connection between the biliary tract and the bronchial tree. This condition is more common in patients with delayed bile leakage. In our cases, three patients with delayed bile leakage developed biliary bronchial fistula, mainly presenting with coughing up bile-like sputum. The symptoms were rapidly controlled after endoscopic drainage. Take one of them as an illustration. A 55-year-old male patient was diagnosed with hepatocellular carcinoma. The tumor was situated in the right liver and compressed the middle hepatic vein. Prior to the operation, TACE, targeted, and immunotherapy were carried out. After the tumor shrank, right hemihepatectomy was conducted. There was no bile-like fluid in the postoperative abdominal drainage tube, and the patient was discharged upon the removal of the drainage tube. A review 13 months after the operation revealed that the tumor recurred in the caudate lobe of the liver. Interventional and targeted immunotherapy were implemented. Two months after the operation, the patient had a fever accompanied by cough with bile-like sputum and came to the hospital for treatment half a month later. CT and MR revealed subphrenic effusion and biliary bronchial fistula (Figure 3). Bile-like fluid could be observed during ultrasound-guided abdominal puncture and drainage for several consecutive days, with a daily drainage volume of approximately 200 ml. Endoscopic treatment was implemented. Contrast imaging indicated a leak in the left hepatic duct combined with biliary stricture, and two 7Fr*15 cm biliary stents were placed (Figure 4). Two days after the stent placement, the abdominal drainage volume was significantly reduced to about 20 ml per day. Seven months later, no bile-like fluid was drained from the abdominal drainage tube, and the drainage tube was removed.

CT and MR of patients with bile leakage after hepatectomy. (a) CT reveals the iodized oil deposition after TACE of the recurrent tumor in the caudate lobe of the liver, (b) CT indicates the subphrenic effusion on the liver section surface, (c) MR shows the subphrenic effusion on the liver section surface, and (d) MR demonstrates the location of bile duct-bronchial fistula and bile duct stenosis at the hepatic hilum.
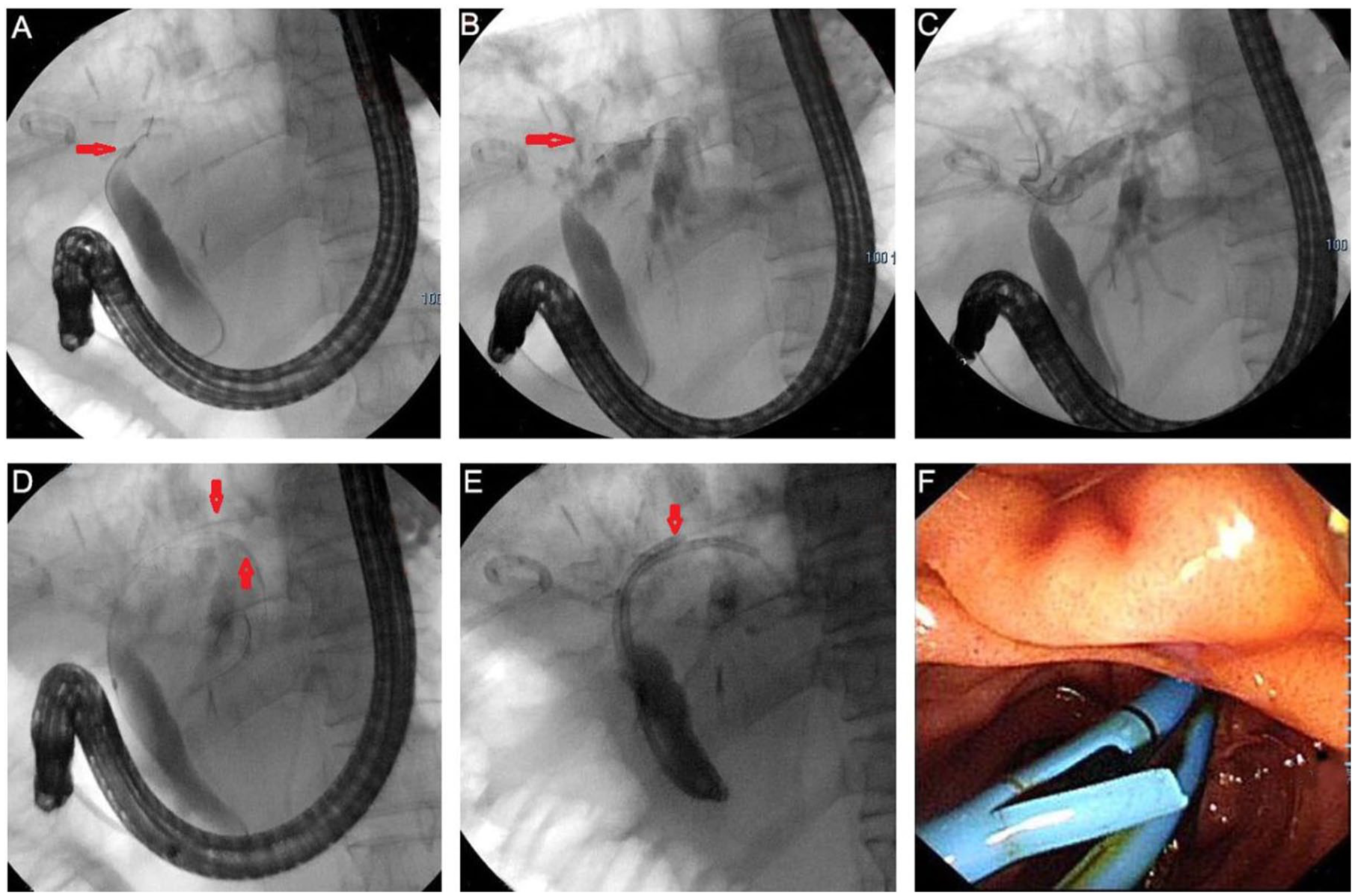
The endoscopic treatment procedure of bile leakage. (a) After intubation, angiography reveals stenosis of the hilar bile duct, and the indwelling abdominal drainage tube is visible, (b) angiography indicates extravasation of the contrast agent, leaking from the left hepatic duct to the subphrenic area, (c) the incision knife passes through the stenosis, and angiography shows the bile ducts of segments S2 and S3 of the liver, (d) double guide wires are indwelled, (e) double stents are placed, and (f) the endoscopic image of double stents.
Discussion
Despite ongoing technological progress, postoperative bile leakage continues to be a significant issue in liver surgery. In fact, apart from minor complications like liver failure and surgical wound infections, postoperative hepatic bile leakage (PHBL) is among the most frequent complications after liver resection. When abdominal drainage tubes placed during or after surgery are functioning properly, bile leakage can usually be managed effectively. However, if the leakage persists, biliary decompression methods become necessary. In the past, percutaneous transhepatic biliary drainage was commonly the first choice. Although the effectiveness of ERCP biliary drainage for treating postoperative bile leakage has been documented, its success rate is only 44.9%. 7
In this study, the overall success rate of ECS was 66.7% (10/15), which is slightly lower than the 75% success rate reported by Alexander et al. 9 This difference may be due to varying criteria for defining successful treatment of bile leakage. Additionally, among the patients who received endoscopic treatment in our study, 46.7% (7/15) experienced delayed bile leakage, and the ECS success rate was lower in this group. Conversely, patients with early bile leakage had an ECS success rate of 75.0% (6/8), consistent with previous literature. The average healing time for bile leakage in our study was 88.5 ± 62.5 days, with early bile leakage healing in 61.0 ± 23.9 days and delayed bile leakage in 119.9 ± 76.5 days. We defined healing time as the period until removal of the abdominal drainage tube. Previous studies report this duration as 4–28 days.7,10 Our results suggest that even early bile leakage requires a longer healing time than previously reported. This may be because most cases in our study underwent transarterial chemoembolization (TACE) and preoperative chemotherapy. Another reason is that the removal of the abdominal drainage tube was delayed; typically, the tube was removed only when bile-like fluid ceased draining or drainage volume was under 10 ml, and drainage tube angiography confirmed that the leakage cavity was connected to the bile duct. Premature removal of the drainage tube could lead to peritoneal effusion and necessitate re-puncture.
Besides the timing of bile leakage onset, two main factors influence the effectiveness of endoscopic treatment for bile leakage: the placement position of biliary drainage and the location of the leakage site. Other factors such as the severity of bile leakage, underlying health conditions, and the extent of surgical intervention also impact the success rate of endoscopic treatment after hepatectomy. Based on the biliary drainage placement, it is categorized as bridging the leakage, near the leakage site, or proximal to the leakage site. Our findings indicate that among all cases undergoing endoscopic sphincterotomy (EST), the ECS success rate was 80% for bridging placement, 75% for drainage near the leakage site, and 0% for drainage proximal to the leakage site. These results align with previous studies. The bridging approach yielded the best outcomes, with ECS success rates ranging from 50% to 100%, while drainage placed proximal to the leakage site was least effective. Most drainage tubes used were plastic stents, with some cases involving covered metal stents and nasobiliary catheters.7,11,12 In 14 cases, we chose biliary drainage using a 7 or 8.5Fr plastic stent, while in one case, due to significant angulation of the intrahepatic bile duct, an ENBD was inserted. Based on the site of bile leakage, we categorized it into leakage from the first-level bile duct, second-level bile duct, and third-level or higher branch bile ducts. Some studies classify bile leakage as occurring in either peripheral or central bile ducts. A recent study by Alexander et al. 9 reported that endoscopic treatment resolved 85% of peripheral bile leakages and 72% of central bile leakages. Anja et al. 11 found success rates of 92.3% for central bile duct leakages and 63.6% for peripheral leakages. Our data show a success rate of 55.6% for central bile duct leakages and 83.3% for peripheral leakages. Theoretically, endoscopic treatment is more successful for central bile duct leakages because bridging is easier unless bile duct strictures prevent guide wire passage. The lower success rate for central bile duct leakages in our cases is attributed to a higher number of delayed bile leakages. We proposed five classifications based on the location of bile leakage and endoscopic biliary drainage, which helps in selecting the most appropriate drainage placement according to the leakage site.
Risk factors for bile leakage after hepatectomy include high BMI, extensive hepatectomy, resections involving liver segments 4–8, preoperative neoadjuvant chemotherapy, repeat hepatectomy, Child-Pugh liver function grade B or higher, diabetes, and others.3,5,13–16 In our cohort of 15 patients, 12 underwent extensive hepatectomy involving more than two liver segments, 14 had resections including segments 7 or 8, and 11 received preoperative adjuvant therapy. Adjuvant treatments for primary liver cancer include TACE, targeted therapy, and immunotherapy. Most patients with colorectal liver metastases received neoadjuvant chemotherapy and targeted therapy before surgery. Our preliminary findings suggest that bile leakage after hepatectomy is associated with extensive resections involving more than two segments, removal of segments 7 or 8, and preoperative adjuvant therapy. With advances in treatment and widespread use of conversion therapy, the combination of TACE with targeted and immunotherapy is increasingly common for primary liver cancer. However, we observed that TACE raises the risk of bile leakage post-hepatectomy. Among the 15 patients, 9 underwent TACE, and notably, 6 of 7 patients with delayed bile leakage had received TACE. Some cases were identified due to abdominal bile accumulation accompanied by bile duct-bronchial fistula. Among our seven patients experiencing delayed bile leakage, four developed bile duct-bronchial fistulas. Those who underwent TACE following endoscopic treatment had significantly longer healing times compared to patients who did not receive TACE. Houyun Xu et al. found that bile duct injury is closely linked to damage of the bile duct microvasculature and subsequent chronic biliary tract infection. The presence of lipid alcohol deposits in the common bile duct wall and a sharp rise in ALP levels to 200 U/L within one week after TACE can serve as predictors of bile duct injury.17–20
Regarding the management of bile leakage after hepatectomy, endoscopic biliary drainage is the preferred treatment for grade B bile leakage; however, it is not always successful. Failures may result from technical issues such as unsuccessful intubation during ERCP, bile leakage accompanied by biliary stricture, or inability to pass the guide wire through the stricture, as well as ineffective treatment outcomes. In such cases, PTCD drainage or docking techniques become necessary. 21 For severe grade C bile leakage, reoperation may sometimes be unavoidable. 16
This study has several limitations. Firstly, this is a single-center non-randomized controlled study with a relatively small sample size, making it difficult to obtain statistically significant results. Second, there is a lack of comparisons with patients who received conservative treatment for bile leakage after hepatectomy. Third, because of the limited sample size, the counts of both successful and unsuccessful endoscopic treatment cases, as well as early and late bile leakage cases, are relatively small. Consequently, the statistical strength of high-risk factors identified through univariate analysis is relatively weak, and future studies with larger sample sizes are needed to perform multivariate analysis.
Conclusion
Endoscopic treatment is effective for bile leakage following hepatectomy and should be considered the first-line therapy for grade B cases. Placing a stent to bridge and drain near the leakage site is likely to benefit patients. Extensive hepatectomy involving more than two liver segments, resections including segments 7 or 8, and preoperative adjuvant therapy are high-risk factors for bile leakage after hepatectomy. Special attention should be given to the effects of TACE on the biliary system, as TACE may cause delayed bile leakage combined with biliary-bronchial fistula and prolonged healing times.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745251388159 – Supplemental material for Endoscopic management of biliary leakage after hepatectomy and analysis of its influencing factors
Supplemental material, sj-docx-1-cmg-10.1177_26317745251388159 for Endoscopic management of biliary leakage after hepatectomy and analysis of its influencing factors by Zongyan Li, Weidong Pan, Guozhi Xu, Zuxiao Chen, Lei Zhang, Zheyu Zheng, Xiaoming Huang, Liangqi Cao and Dawei Zhang in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
