Abstract
Objective
To assess the safety and effectiveness of a self-expandable metallic stent (SEMS) combined with Iodine-125 seeds strand to treat hilar malignant biliary obstruction (MBO).
Methods
This retrospective study included patients who had received SEMS with 125I seeds strand (seeds group) or SEMS alone (controls) to treat hilar MBO. Demographic, biochemical, stent patency, overall survival and complications data were extracted and analysed.
Results
A total of 76 patients were included (seeds group,
Conclusion
SEMS combined with 125I seeds strand is safe, feasible, and tolerable in treating patients with hilar MBO, and may be effective in prolonging stent patency time and overall survival.
Introduction
Hilar malignant biliary obstruction (MBO) mainly invades the common hepatic duct, and bifurcation of the left and right hepatic ducts. 1 Because it is asymptomatic at an early stage, most cases of cholangiocarcinoma are diagnosed at an advanced stage, at which the opportunity for radical surgery has been lost. 2 For patients with unresectable MBO and life expectancy exceeding 3 months, self-expandable metallic stent (SEMS) deployment is the standard treatment option, and may effectively relieve jaundice and improve quality of life.3,4 However, stent restenosis can occur, mainly due to tumour overgrowth or ingrowth, biliary epithelial cell proliferation and biliary sludge formation and accumulation. 5 Although several methods aim to improve biliary stent patency and therefore prolong survival in patients with hilar MBO, e.g. covered SEMS, stent combined with chemotherapy, photodynamic therapy and radiofrequency ablation,5–8 the results of such regimes are not optimal. The efficacy of endobiliary radiofrequency ablation remains controversial,5,9 and is associated with a risk of biliary perforation. 10 The availability, photosensitivity in patients, and procedural cost limit the clinical application of photodynamic therapy. 6 Research has shown that external beam radiotherapy or brachytherapy may be effective in the treatment of hilar cholangiocarcinoma, however, external beam radiotherapy may cause severe toxicities, including dehydration and mucositis. 11 Interstitial Iodine-125 (125I) brachytherapy has been widely applied in tumour treatment to locally control lung cancer, prostate carcinoma, and malignant portal vein thrombus among others, all with promising results.12–14 125I seeds strands have also been applied to treat MBO,15,16 however, the use of 125I radioactive particles in hilar MBO has rarely been described.17–19
The aim of the present study was to perform retrospective analyses of the effectiveness and safety of SEMS combined with 125I seeds strand in the treatment of patients with Bismuth type I–IV hilar MBO.
Patients and methods
Study population
This retrospective study included data from sequential patients with hilar MBO who had undergone biliary SEMS with or without 125I seeds strand at Beijing Chaoyang Hospital, Capital Medical University between January 2017 and July 2018. Inclusion criteria were: (1) aged between 18 and 90 years; (2) hilar MBO diagnosis by clinical and radiological findings, or confirmed by histological examination; (3) total bilirubin increased three times higher than normal levels due to MBO; (4) ineligible for surgery, or declined resection due to poor Karnofsky performance status (score < 70); (5) life expectancy > 3 months. The following exclusion criteria were applied: (1) suspected benign biliary obstruction; (2) previous radiotherapy for treatment of hilar MBO; (3) previous metallic biliary stent insertion, surgery, chemotherapy or cancer-targeted treatment; (4) massive ascites; (5) uncontrolled coagulation disorder; (6) uncontrolled infection; (7) patients who refused biliary stent with or without 125I seeds to treat hilar MBO. Treatment with SEMS alone without 125I seeds was performed under the following conditions: (1) patient refusal to receive 125I seeds; and (2) the possibility of the patient having close contact with pregnant women, infants and children. Demographic and clinical data, including tumour type, Bismuth-Corlette classification 1 and Karnofsky performance status score, were extracted from patient records and anonymized prior to analyses.
The study was approved by the ethics committee of Beijing Chaoyang Hospital, Capital Medical University, Beijing, China, and was conducted according to standards of the Declaration of Helsinki. Due to the retrospective characteristics of this study, the requirement for informed consent was waived by the ethics committee.
Interventional procedures
125I seeds strand preparation
The 125I seeds (CIAE-6711) were provided by Zhibo Gaoke Biotechnology (Beijing, China). The core radioactive source was a silver wire containing 125I, which was covered by a laser sealed medical titanium alloy tube. 125I seed activity was 0.6 millicurie (mCi) per particle. Each 125I seed had a half-life of 60.1 days, and was 4.5 ± 0.5 mm in length and 0.8 mm in diameter. The following formula was used to determine the number of 125I seeds (N) required: N = length of biliary stricture (mm)/4.5 + 4. 16 The 125I seeds strand, used for loading the seeds, was made by inserting the appropriate number of 125I seeds into a 4F catheter (Cook Medical, Bloomington, IN, USA) that was then heat sealed at either end. The estimated radiation dose of the 125I seed strand at the reference points (5 mm from the source axis) was calculated according to the American Association of Physicists in Medicine (AAPM) Task Group No. 43 update report (TG-43U1). 20 The incipient dose rate and the cumulative dose (1 half-life, measured at the dose reference points) were 8.03–8.59 cGy/h and 83.15–88.94 Gy, respectively.
SEMS and 125I seeds strand implantation
Two hydrophilic wires were arranged across the bile duct stricture. The SEMS (Cook Medical), usually 8 or 10 mm in diameter and 40–100 mm in length, was deployed via one wire. A 6F guiding catheter (Cook Medical), used for 125I seed deployment, was inserted via the other wire. After the stent was released, a prepared 125I seeds strand was inserted into the 6F guiding catheter and located at the bile duct stenosis. While withdrawing the 6F guiding catheter, the 125I seeds strand was released between the SEMS and bile duct wall. Finally, a 7.0–8.5F drainage catheter (Cook Medical) was placed indwelling above the SEMS. The 125I seeds strands were handled to prevent unintended radiation according to the International Commission on Radiological Protection recommendations.
Follow-up and definitions
Technical success was defined as follows: SEMS covered the biliary stricture segment with residual stricture < 30%; contrast agent passed smoothly through the SEMS; and the 125I seeds strand also covered the whole biliary stricture segment. Clinical success was defined as a decrease in bilirubin level by at least 75% after 1 month compared with the pre-procedural value. Procedural time (duration) was defined as the time from the start of the procedure to the stent release. Overall survival refers to the time interval from SEMS deployment to patient death or the last follow-up date. Stent patency time refers to the time interval from SEMS deployment to stent restenosis. If stent restenosis had not occurred when the patient died, the patency time was equal to the survival time but censored. Stent restenosis was defined as recurrence of jaundice and cholangitis symptoms, and an increase in total bilirubin, and was confirmed by imaging examination or cholangiography. Complications were divided into early or late complication according to whether they occurred within 30 days, or after 30 days, of stent deployment. All patients were followed up by telephone interview, outpatient appointment or inpatient examination every 3 months following the procedure. The follow-ups were concluded on 31 December 2018.
Statistical analyses
All clinical data were analysed using SPSS software, version 22.0 (IBM, Armonk, NY, USA). Continuous data conforming to normal distribution are presented as mean ± SD and were compared using Student’s
Results
Patient characteristics
Out of 76 patients in total, 40 had received SEMS combined with 125I seeds strand (seeds group) while 36 had received SEMS only (control group). The seeds group comprised a total of 21 male and 19 female patients (age, 70.2 ± 13.8 years; range, 30–90 years) and the control group comprised 21 male and 15 female patients (age, 68.1 ± 12.2 years; range, 42–89 years). Baseline clinical characteristics were balanced between the two groups (Table 1). A total of 71 out of 76 patients (93.42%) were diagnosed with hilar MBO by clinical and radiological findings, and five patients (6.58%) were diagnosed by histological examination. In the seeds group, three patients received 2–4 cycles of transarterial chemoembolization (TACE); two patients received 1–2 liver microwave ablations; and one patient was deployed a SEMS due to duodenal obstruction. In the control group, two patients received 1–2 cycles TACE and two patients had stents inserted due to intestinal obstruction. In the seeds group, a total of 608 seeds were deployed (mean, 15.2 ± 4.1 [range, 8–25] seeds per patient); 47 SEMSs were inserted in 40 patients, and seven cases received double biliary SEMSs. In the control group, 40 SEMSs were inserted in 36 patients, while four cases received double biliary SEMSs. Representative cases are shown in Figures 1 and 2.
Baseline demographic and clinical characteristics in patients with hilar malignant biliary obstruction treated with either self-expandable metallic stent (SEMS) plus 125I seeds strand (seeds group) or with SEMS alone (control group).

Data presented as mean ± SD or
No statistically significant between-group difference at

Representative images from a 71-year-old male patient with cholangiocarcinoma: (a) Abdominal enhanced computed tomography (CT) showing a large tumour and local biliary branch expansion in the right hepatic lobe, (b) Abdominal enhanced CT showing a giant tumour embolus (arrow) in the right hepatic common biliary duct, (c) Percutaneous transhepatic cholangiography showing hilar malignant biliary obstruction (Bismuth type IV; right intrahepatic biliary duct, common hepatic duct and common bile duct were undetected), (d) Cholangiography showing KMP directional catheter (Cook Medical, Bloomington, IN, USA) crossing the biliary stricture segment, and giant tumour embolus (arrow), (e) A self-expandable metallic stent (diameter, 8 mm; length, 80 mm; Cook Medical) was inserted into the biliary stricture and a linearly arranged (arrow) 125I seeds strand with 22 seeds (0.6 mCi per seed) was fixed steadily between the stent and the malignant biliary duct wall and (f) Abdominal enhanced CT at 3 months following the procedure, showing a patent biliary stent and significantly reduced tumour thrombus (arrow).

Representative images from an 82-year-old male patient with hilar cholangiocarcinoma: (a) Magnetic resonance cholangiopancreatography showing widely expanded intrahepatic biliary duct, and hilar biliary obstruction with no communication between the right and left bile ducts, (b) Percutaneous transhepatic cholangiography showing hilar bile duct obstruction, Bismuth type III, (c and d) One self-expandable metallic stent (SEMS; diameter, 8 mm; length, 60 mm; Cook Medical, Bloomington, IN, USA) was inserted into the biliary stricture to connect the right hepatic and common bile ducts; the other SEMS (diameter, 8 mm; length, 40 mm; Cook Medical) was inserted into the biliary stricture to connect the right and left bile ducts; two linearly arranged 125I seeds strands with 12 seeds (0.6 mCi per seed) were fixed steadily between the two stents and the malignant biliary duct wall.
Procedural outcomes
The technical success rate of SEMS and 125I seeds strand release was 100% in all 76 patients. The clinical success rate was 95.0% (38/40) in the seeds group and 97.2% (35/36) in the control group (
Biochemical parameter improvement
Compared with pre-procedure values, there were statistically significant improvements in both groups regarding total bilirubin, direct bilirubin, alkaline phosphatase, glutamyl transferase, aspartate aminotransferase and alanine transaminase levels, following the procedure (all
Improvements in biochemical parameters at 1 month following the procedure in patients with hilar malignant biliary obstruction treated with either self-expandable metallic stent (SEMS) plus 125I seeds strand (seeds group) or with SEMS alone (control group).
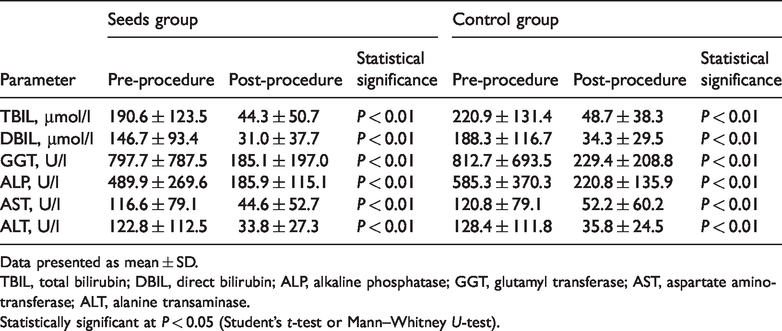
Data presented as mean ± SD.
TBIL, total bilirubin; DBIL, direct bilirubin; ALP, alkaline phosphatase; GGT, glutamyl transferase; AST, aspartate aminotransferase; ALT, alanine transaminase.
Statistically significant at
Complications and re-intervention
The incidence of complications was 50.0% (20/40) in the seeds group versus 38.9% (14/36) in the control group, with no statistically significant differences (χ2 = 0.290,
Early and late complications in patients with hilar malignant biliary obstruction treated with either self-expandable metallic stent (SEMS) plus 125I seeds strand (seeds group) or with SEMS alone (control group).

Data presented as
No statistically significant between-group difference at
Overall survival and stent patency time
Up to 31 December 2018, 10 patients remained alive and 30 patients had died in the seeds group; 19 of the 30 patients had died without jaundice and with patent biliary stents. Causes of death included multiple organ failure secondary to carcinoma progression (

Kaplan–Meier survival curves from patients with hilar malignant biliary obstruction treated with either self-expandable metallic stent (SEMS) plus 125I seeds strand (seeds group) or with SEMS alone (control group), showing: (a) cumulative overall survival, and (b) duration of stent patency (patency time).
Discussion
According to the Bismuth-Corlette system, hilar MBO can be classified into four types (Type I–IV). 1 The percutaneous approach of biliary drainage is more effective and safer than endoscopic methods in hilar MBO, particularly in Bismuth type IV. 21 Because of the silence and insidiousness in clinical progress of hilar MBO, only a few patients are suitable for radical resection, 22 and the recurrence rate after surgery is also very high. 23 Tumour overgrowth or ingrowth, biliary epithelial cell proliferation and biliary sludge formation and accumulation are the main reasons for stent restenosis. 5
The half-life of 125I seed is about 60.1 days, with an effective radiation radius of 17 mm and dose attenuation according to the distance. Due to such characteristics, 125I seed is considered to be the first choice for intraluminal radiotherapy. 125I seed can induce a therapeutic effect by injury to the DNA double helix structure and induction of tumour cell apoptosis.
24
In 1986, 125I seed was appoved by the US Food and Drug Administration for tumour treatment, and to date, it has been widely used in the treatment of various primary tumours and metastases, e.g. prostate and lung cancers.13,25 Chen et al.
26
first reported the use of a 125I seeds strand in the pig bile tract, assessing technical feasibility in treating the human bile duct. The first encouraging results of 125I seeds stent in the clinical setting were reported by Zhu et al.
15
who’s interim analysis showed that the 125I seeds stent was safe and feasible in the treatment of MBO, and appeared to extend overall survival compared with the conventional stent (7.40 months versus 2.50 months).
15
Since then, low dose-rate 125I seeds combined with SEMS have been applied to treat MBO, with promising results,16–18,22,27 however, the application of 125I seeds in hilar biliary obstruction has rarely been reported.17,18 A prospective study on the treatment of hilar MBO (Bismuth I–II) with SEMS combined with 125I seeds, showed that overall survival and stent patency were significantly prolonged in the seeds group compared with control treatment.
17
A retrospective analysis of 132 patients (95 with biliary obstruction located at the hilar region) who received SEMS with 125I seeds strand (seeds group) or SEMS alone (control group) for MBO, showed that, compared with controls, treatment with SEMS plus 125I seeds significantly prolonged median stent patency (194 days versus 86 days,
At present, there are three methods for deployment of 125I seeds: (1) 125I seeds strand inserted in a drainage catheter;
16
(2) an 125I seeds-loaded stent;18,29 and (3) an 125I seeds strand fixed between SEMS and the bile duct wall.17,28 The advantage of the first method is replaceability of the 125I seeds strand, but a disadvantage is the limitation of external biliary drainage. A disadvantage of the second method is the composition of two-layer stents and larger diameter sheath. The third method was adopted in the present study for its simple procedural process and good tolerance, however, this method also has three disadvantages: (1) the 125I seeds strand is difficult to retrieve when 125I seeds stop functioning after 6 months; (2) pain medication should be administrated to relieve intra-procedural pain due to SEMS deployment without a sheath; and (3) the procedural time for delivering SEMS combined with 125I seeds strand may be longer than SEMS alone, as was shown in the present study (46.4 ± 13.1 min versus 39.4 ± 12.1 min,
The 76 patients with hilar MBO were analysed retrospectively in the present study. Among the participants, there were 54 cases of Bismuth type I–II MBO, and 22 cases of Bismuth type III–IV MBO. A total of 608 seeds were deployed in 40 patients (seed group), with a mean of 15.2 ± 4.1 (range, 8–25) seeds per patient. The incipient dose rate and the cumulative dose (1 half-life, measured at the dose reference points and calculated according to the AAPM Task Group No. 43 update report (TG-43U1),
20
were 8.03–8.59 cGy/h and 83.15–88.94 Gy, respectively. Such radiation doses satisfy the minimum threshold for treatment of adenocarcinoma, and have been reported as safe in animal experiments as well as in clinical trials.15,30 In the present study, compared with SEMS alone, SEMS combined with 125I seeds strand significantly prolonged stent patency (387.0 ± 27.9 days versus 121.0 ± 9.1 days,
The results of the present study may be limited by several factors. First, there was no treatment randomization due to this being a retrospective study, and thus, there may be bias in selection of the patient group. Secondly, there was no specific treatment planning system to guide the 125I seeds deployment into hollow organs. Thirdly, most of the patients with hilar MBO were diagnosed by clinical and radiological findings, and the histopathological diagnosis rate of hilar MBO was relatively low.
In conclusion, SEMS combined with 125I seeds strand represents a safe, feasible, and tolerable operation in patients with hilar MBO. Treatment with SEMS plus 125I seeds strand was shown to be effective in prolonging stent patency time and overall survival in patients with hilar MBO.
