Abstract
Background:
Lumen apposing metal stents (LAMSs) have a higher clinical success rate for managing pancreatic fluid collections. But they are associated with adverse events (AEs) like bleeding, migration, buried stent, occlusion, and infection. It has been hypothesized that placing a double pigtail stent (DPS) within LAMS may mitigate these AEs. The present systematic review and meta-analysis were conducted to compare the outcome and AEs associated with LAMS with or without a coaxial DPS (LAMS-DPS).
Methods:
A comprehensive literature search of three databases from January 2010 to August 2022 was conducted for studies comparing the outcome and AEs of LAMS alone and LAMS-DPS. Pooled incidence and risk ratios (RRs) with 95% confidence intervals (CIs) were calculated for all the dichotomous outcomes.
Results:
Overall, eight studies (n = 460) were included in the final analysis. The clinical success rate (RR 1.00, 95% CI: 0.87–1.14) and the risk of overall AEs (RR 1.60, 95% CI: 0.95–2.68) remained comparable between both groups. There was no difference in the risk of bleeding between LAMS alone and LAMS-DPS (RR 1.80, 95% CI: 0.83–3.88). Individual analysis of other AEs, including infection, stent migration, occlusion, and reintervention, showed no difference in the risk between both procedures.
Conclusion:
The present meta-analysis shows that coaxial DPS within LAMS may not reduce AE rates or improve clinical outcomes. Further larger studies, including patients with walled-off necrosis, are required to demonstrate the benefit of coaxial DPS within LAMS.
Keywords
Introduction
Pancreatic fluid collections (PFCs) are a local complication of moderate to severe acute pancreatitis. Most PFCs resolve spontaneously and do not require intervention. Infected collections, persistent organ failure, gastric outlet or biliary obstruction, and symptomatic collections are a few indications for drainage of PFC. 1 Lumen apposing metal stents (LAMSs) are increasingly preferred over plastic stents for drainage of PFCs due to higher clinical success, lower recurrence rate, and fewer additional interventions. 2 LAMS has a bi-flanged shape that allows for tissue apposition and minimizes the risk of stent migration. The wide-diameter lumen provides a non-compressible fistulous tract that facilitates the passage of an endoscope, making necrosectomy possible. The technical and clinical success rates for LAMS in various studies have been reported to be 93–100% and 89–100%, respectively.2,3
However, LAMS is associated with multiple adverse events (AEs), including bleeding, migration, buried stent, occlusion, and biliary obstruction secondary to mechanical compression of the bile duct. 4 Clogging of LAMS with food particles resulting in secondary infection of the collection is commonly encountered. Placing a coaxial double pigtail stent (DPS) within LAMS helps prevent the clogging of food and necrotic tissue and ensures patency of LAMS by preventing the obstruction of the inner flange of LAMS by the collapsed cyst wall. Further, coaxial DPS placement prevents LAMS’s impaction on adjacent vasculature, reducing the risk of delayed bleeding. 5
The addition of coaxial DPS to LAMS was associated with a lower rate of AEs in endoscopic ultrasound-guided drainage of PFC. 6 However, subsequently, another study reported no difference in the AEs, rate of cyst resolution, or reintervention rates in the LAMS-DPS group compared to LAMS alone. 7 A recent guideline also recommended placing a coaxial DPS inside a LAMS as it is associated with a lower rate of AEs, including stent migration and the need for necrosectomy. 8 Hence, the present systematic review and meta-analysis were conducted to study LAMS’s outcome and AEs with or without coaxial DPS.
Methods
Information sources and search strategy
A comprehensive search of all relevant studies was conducted using the databases of MEDLINE, EMBASE, and Scopus from January 2010 to October 2022. The keywords used were: (LAMS OR “Lumen apposing metal stent” OR BFMS OR “Biflanged metal stent”) AND (Coaxial OR Pigtail) AND (WON OR Pseudocyst OR “PFC”). To ensure that no potentially relevant items were overlooked, manual searching of reference lists of the included studies was also undertaken. The study methodology was designed and executed to adhere to the Preferred Reporting Items for Systematic review and Meta-Analyses (PRISMA) guidelines. 9
Study selection
The population, intervention, comparator, outcome (PICO) criteria used for included comparative studies were: (a) Patients—cystogastrostomy for PFC; (b) Intervention—coaxial DPS with LAMS; (c) Comparison—LAMS alone; (d) Outcomes—clinical success and AEs. In accordance with the selection criteria above, the titles and abstracts of all studies were independently reviewed by two authors. A third reviewer resolved any disagreements. The exclusion criteria used were: non-comparative studies, case series, and studies involving persons <18 years of age.
Data extraction
Two independent reviewers performed the data extraction, and a third reviewer resolved any disagreement. Data were collected under the following headings: study author and year, country of study, study design, number of patients, age and sex distribution, details of PFC, procedural details, and clinical outcomes.
Definition of outcomes
The primary outcomes of the analysis were AEs associated with procedures and reintervention. We individually compared all the described AEs, including bleeding, infection, stent migration, and stent occlusion. The secondary outcome of the study was clinical success, defined as improvement in patients’ symptoms and reduction in the PFC to less than 2 cm on follow-up.
Risk of bias in individual studies
After data extraction, the same two reviewers performed a risk of bias (quality) assessment using validated tools. The Cochrane risk of bias tool was used for randomized controlled trials (RCTs), and the Cochrane collaboration’s risk of bias in non-randomized studies of interventions tool for non-randomized studies.
Statistical analysis
Risk ratios (RRs) with 95% confidence intervals (CIs) were calculated for all the dichotomous outcomes. Regardless of heterogeneity, the Mantel-Haenszel test for random effects was used. A Cochran’s Q test and I2 statistics were used to determine the heterogeneity between the studies. A p-value of Q test < 0.1 or the I2 value > 50% was significant. Visual inspection of funnel plots was used for publication bias assessment. The sensitivity analysis was performed using a leave-one-out meta-analysis, in which one study is excluded at each analysis to analyze each study’s influence on the overall effect-size estimate and identify influential studies. RevMan software (version 5.4.1, Cochrane collaboration), and STATA software (version 17; StataCorp., College Station, TX, USA) were used for statistical analysis.
Results
Study characteristics and quality assessment
The search strategy yielded 468 records, out of which eight studies6,7,10 –15 were included in the final analysis. Figure 1 shows the PRISMA diagram for the study selection and inclusion process. Table 1 shows the baseline characteristics of the included studies. Four studies were full-text articles,6,7,10,14 and four were conference abstracts.11 –13,15 Except for the study by Vanek et al., 15 all were retrospective studies. One study included only pseudocysts, 6 one included only WON, 15 and the rest included both. The body of the pancreas was the commonest location, and transgastric was the commonest route of drainage. Supplemental Figure 1 shows the risk of bias assessment for individual studies. Of the included studies, four had moderate,6,11,14,15 and four had a high risk of bias.7,10,12,13

PRISMA flowchart for study identification, selection, and inclusion process.
Baseline characteristics of the included studies.

DPS, double pigtail stent; Etiology (A/B/O), alcohol/biliary/others; LAMS, lumen apposing metal stent; Location of PFC (H/B/T/M), head/body/tail/multiple; PFC, pancreatic fluid collection; Site of drainage (TG/TD/B), transgastric/transduodenal/both; Type of PFC (PP/WON/PS), pseudocyst/walled-off necrosis/post-surgical.
Clinical success
A total of five studies (n = 302) compared the clinical success between both groups.6,7,12 –14 The pooled clinical success rate with LAMS alone and LAMS-DPS was 88.5% (95% CI: 81.3–95.8) and 87.0% (95% CI: 74.7–99.4), respectively (Table 2). Clinical success was comparable between both groups (RR 1.00, 95% CI: 0.87–1.14; I2 = 62%) (Figure 1).
Pooled event rate for various outcomes with LAMS with or without double pigtail stent.

DPS, double pigtail stent; LAMS, lumen apposing metal stent.
AEs
Overall, seven studies (n = 411) reported on the total no. of AEs associated with either of the procedures.6,7,10,12 –15 The pooled incidence of AE with LAMS alone was 33.5% (95% CI: 25.8–41.2), while it was 18.8% (95% CI: 6.9–30.7) in the LAMS-DPS group. However, there was no difference in the risk of AE between the procedures RR 1.60 (95% CI: 0.95–2.68; I2 = 45%) (Figure 2). Of all the AEs, only four were intraprocedural (2 perforations, 1 bleeding, and 1 aspiration), and the rest were post-procedural. Hence, we compared the post-procedural AEs between the groups.

Forest plot comparing clinical success between lumen apposing metal stent (LAMS) alone and LAMS with double pigtail stent (LAMS-DPS).
A total of eight studies (n = 460) compared the incidence of post-procedural bleeding with LAMS with or without DPS.6,7,10 –15 The pooled incidence of post-procedural bleeding with LAMS alone and LAMS-DPS was 6.3% (95% CI: 2.3–10.2) and 3.1% (95% CI: 2.2–6.6), respectively. Using a coaxial stent did not reduce the risk of post-procedural bleeding compared to LAMS alone (RR 1.80, 95% CI: 0.83–3.88; I2 = 0%) (Figure 3).
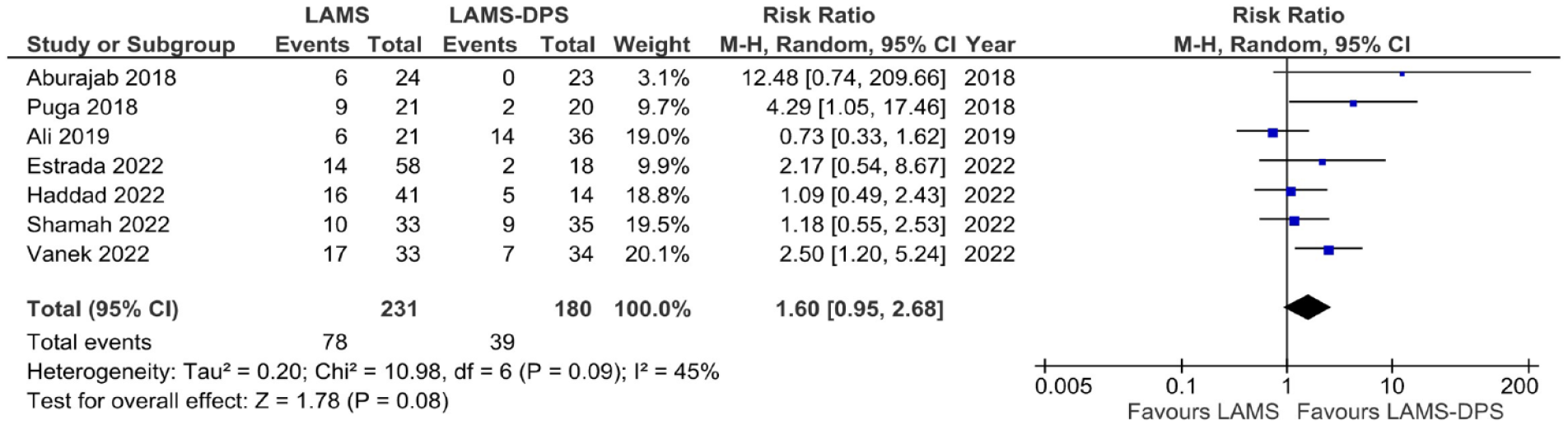
Forest plot comparing risk of adverse events between lumen apposing metal stent (LAMS) alone and LAMS with double pigtail stent (LAMS-DPS).
Analyzing other AEs, the risk of infection (RR 1.78, 95% CI: 0.34–9.47; I2 = 43%), stent migration RR (0.81, 95% CI: 0.33–2.01; I2 = 0%), stent occlusion (RR 1.72, 95% CI: 0.90–3.27; I2 = 0%), and reintervention (RR 1.43, 95% CI: 0.83–2.45; I2 = 30%) were comparable between LAMS alone and LAMS-DPS group (Figures 4 and 5).

Forest plot comparing risk of bleeding between lumen apposing metal stent (LAMS) alone and LAMS with double pigtail stent (LAMS-DPS).
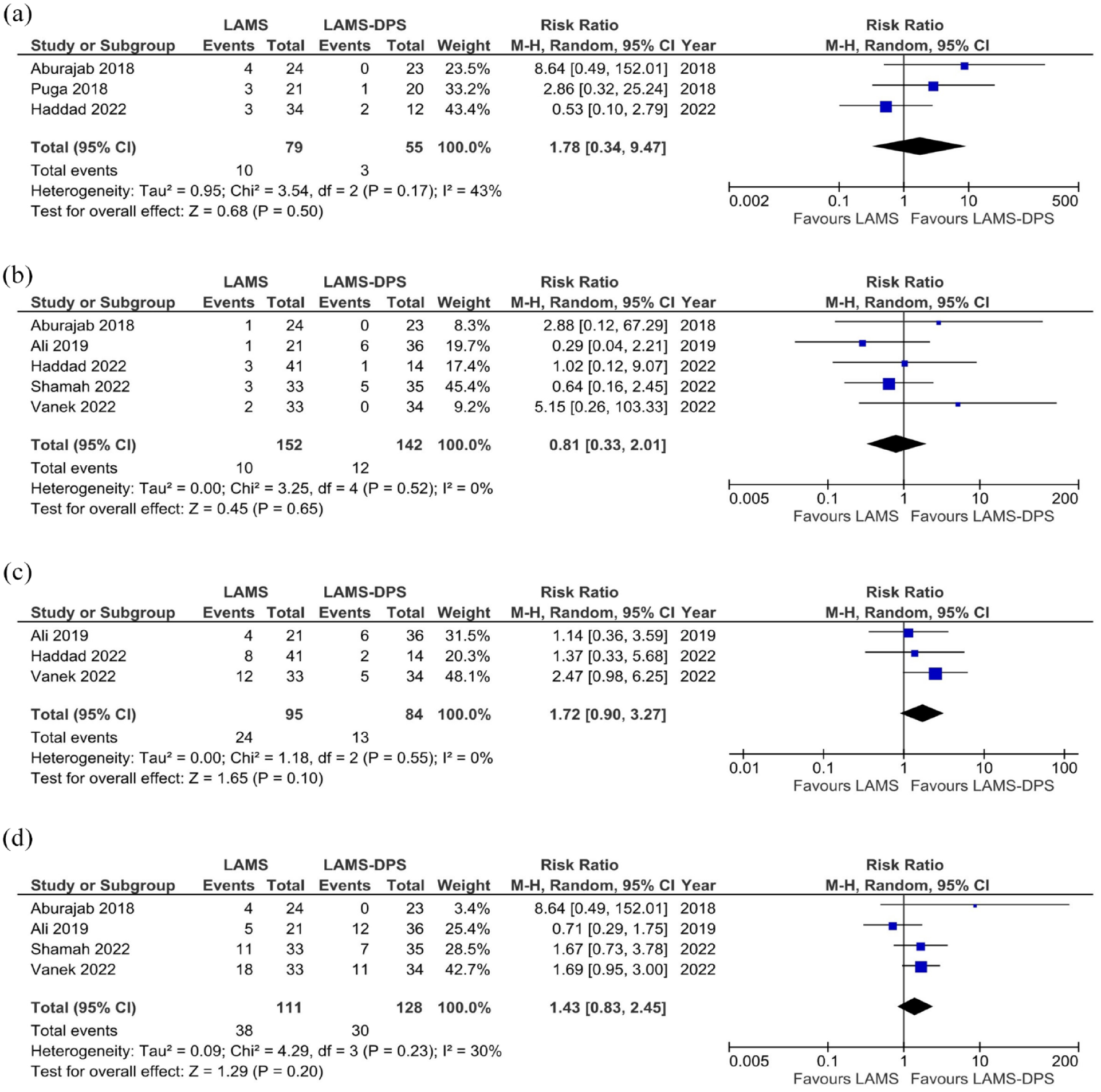
Forest plot comparing risk of (a) infection, (b) stent migration, (c) stent occlusion, and (d) reintervention between lumen apposing metal stent (LAMS) alone and LAMS with double pigtail stent (LAMS-DPS).
Publication bias, sensitivity analysis, and certainty of evidence
There was no evidence of publication bias for the outcomes of clinical success, AE, and bleeding (Supplemental Figure 2). On leave-one-out analysis, there was a significant change in the risk of various outcomes between the groups. Table 3 shows the summary of findings with a grade of evidence.
Summary of findings and grade of evidence.
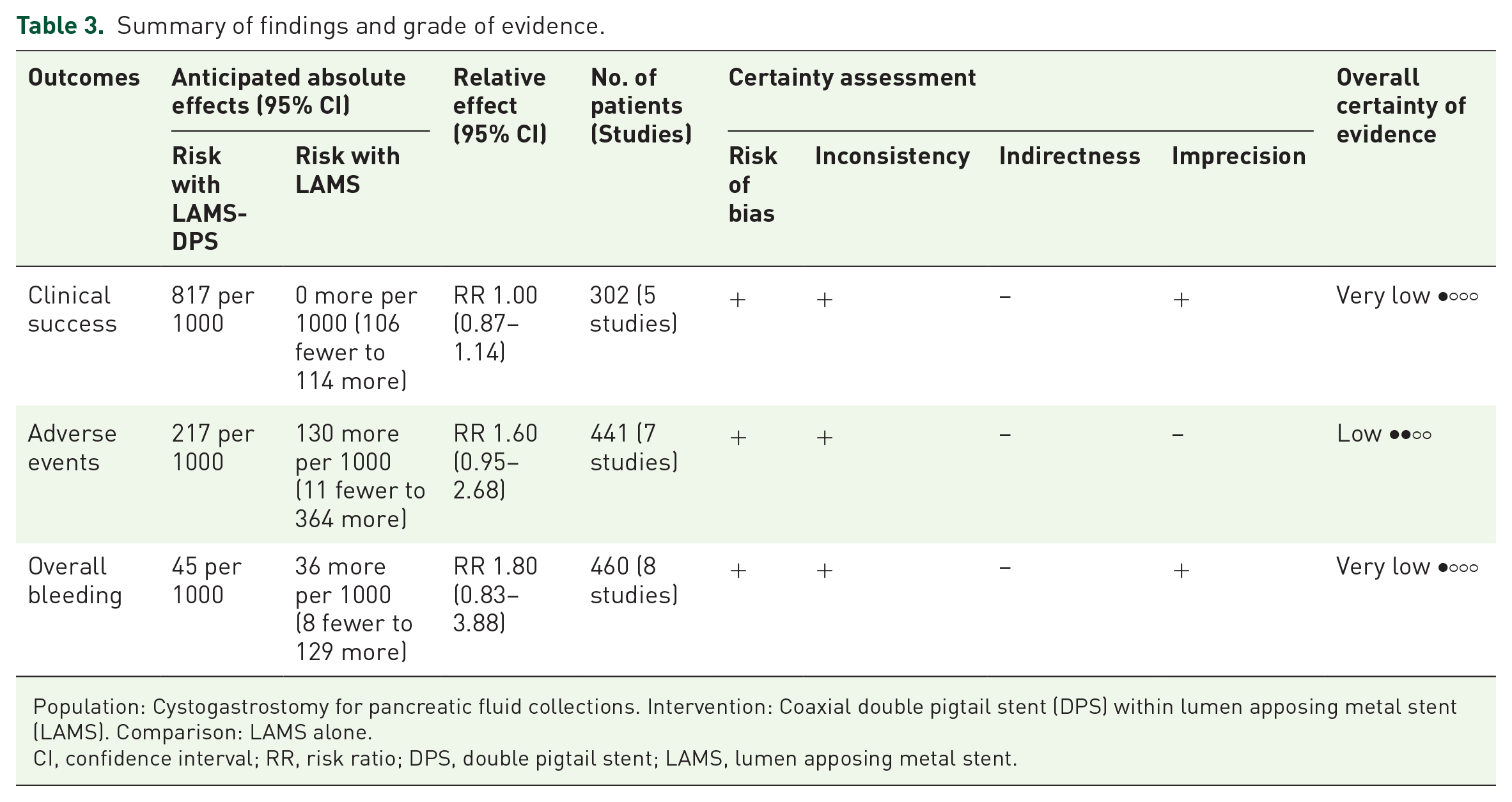
Population: Cystogastrostomy for pancreatic fluid collections. Intervention: Coaxial double pigtail stent (DPS) within lumen apposing metal stent (LAMS). Comparison: LAMS alone.
CI, confidence interval; RR, risk ratio; DPS, double pigtail stent; LAMS, lumen apposing metal stent.
Discussion
Endoscopic ultrasound guided (EUS-guided) drainage has become the current mainstay of therapy for PFCs with high technical and clinical success rates. Although LAMS placement has a high success rate, two recent studies have shown an increased incidence of AEs as well. Bang et al. 16 demonstrated in a RCT, that LAM placement is associated with an AE rate to the tune of 50% versus 0% in the DPS group. Most of these events occurred three weeks after stent placement. Lang et al. 17 demonstrated that LAMS placement was associated with an increased bleeding rate compared to the DPS group (21% versus 1%, respectively, p = 0.03). Brimhall et al. 18 in a retrospective analysis demonstrated an increased bleeding risk from pseudoaneurysm in the LAMS group as compared to DPS.
The reason proposed for the increased AE rates in the LAMS alone group is that PFCs collapse quite rapidly after drainage, causing the internal flange of the stent to erode the regional vasculature and the mucosa, causing bleeding. In addition, the increased rates of stent occlusion in the LAMS alone group may be due to undigested food or debris blocking the stent lumen. Plastic stents project downwards due to gravity as the stent collapses, thereby, in theory moving the internal flange of the stent away from the mucosa and keeping it in the lumen. Also, it prevents stent occlusion by undigested food and necrotic debris. Additionally, plastic stents are comparatively less expensive and easy to remove even after being in place for a long period of time. 19 However, placing plastic stents alone will lead to incomplete drainage in view of small diameters and may lead to infection within the PFCs.
The evidence to support the practice of placing a DPS through the LAMS to prevent AEs is weak. The studies are largely retrospective, with a small number of patients. A prospective study showed that placing a DPS across the LAMS minimized the risk of pseudocyst infection as compared to placing LAMS alone. 6 However, a recent large multicenter study by Shamah et al. 14 demonstrated no difference in technical and clinical success rates and AE profiles with and without the placement of DPS through the LAMS. This result extended when different types of PFC’s were compared as well.
Results of this analysis demonstrate a higher pooled incidence of AE in the LAMS alone group (33.5%, 95% CI: 25.8–41.2) as compared to the LAMS-DPS group (18.8%, 95% CI: 6.9–30.7). However, there was no significant difference in the risk of AE between the two groups (RR 1.60, 95% CI: 0.95–2.68). Similarly, the odds of bleeding were also comparable between the two groups (RR 1.80, 95% CI: 0.83–3.88). On subgroup analysis, even when specific AEs (infection, migration, and occlusion) were examined, there was still no significant difference in the rate of occurrence between the two groups. The study by Aburajab et al. 6 was the first one to demonstrate that the addition of DPS through the LAMS in pancreatic pseudocysts would decrease the risk of infection requiring reintervention. A subsequent similar study by Puga et al. 10 confirmed these findings. However, both these studies were retrospective and were done with a small number of patients. This makes the findings of this meta-analysis more relevant to clinical practice and suggests that the effort, time, and cost associated with placement of a DPS through LAMS may not confer any additional benefit.
In the study by Perez Estrada et al., 12 there was no significant difference in the AE rate between the two groups. However, on subgroup analysis of patients with WON, LAMS-DPS was associated with significantly less endoscopic revisions (1.38 ± 1.50 versus 4.39 ± 3.97; p = 0.042) and shorter time to LAMS removal (45.75 ± 26.46 versus 149.00 ± 34.72 days; p < 0.001). This is in line with the recent large-scale prospective RCT of 67 patients by Vanek et al., 15 which demonstrated lower AE and stent occlusion rates with LAMS-DPS in patients with WON. Thus, LAMS-DPS may have a benefit in patients with WON rather than a pseudocyst. But this needs to be proven in further studies.
To the best of our knowledge, this is the first meta-analysis that comprehensively looked at the clinical advantage of placing a coaxial DPS stent after LAMS placement for the management of PFCs. The findings of the present meta-analysis are relevant for the following reasons. The effort and cost that is involved in the placement of an additional coaxial stent do not translate to better clinical outcomes. Although the overall AE rate was lower in the LAMS-DPS group, the results were not statistically significant. Despite this, there were multiple limitations in our study. First, most of the studies were retrospective. Second, there were no predefined criteria for patient selection for the LAMS alone or LAMS-DPS strategies. Next, we could not perform a subgroup analysis based on the type and size of PFC. In the study by Perez Estrada et al., 12 the authors concluded that taking power = 80%, 95% two-sided significance level. 10% risk of bleeding in LAMS alone and 5% with LAMS-PS, a sample size of >475 in each arm would be optimal. Hence, the present meta-analysis is underpowered to show the benefit of coaxial stents in reducing bleeding risk. Lastly, the risk of LAMS-related AEs is higher after 3 weeks of the procedure. However, a subgroup analysis based on the timing of the AEs in relation to the procedure could not be performed due to the unavailability of data regarding the same.
In conclusion, the present meta-analysis shows that placing coaxial DPS within LAMS is not associated with decreased AE rates or better clinical outcomes. These results highlight the need for better large-scale prospective studies, including patients with WON, to resolve this question.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745231199364 – Supplemental material for Does a coaxial double pigtail stent reduce adverse events after lumen apposing metal stent placement for pancreatic fluid collections? A systematic review and meta-analysis
Supplemental material, sj-docx-1-cmg-10.1177_26317745231199364 for Does a coaxial double pigtail stent reduce adverse events after lumen apposing metal stent placement for pancreatic fluid collections? A systematic review and meta-analysis by Suprabhat Giri, Sidharth Harindranath, Shivaraj Afzalpurkar, Sumaswi Angadi and Sridhar Sundaram in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
