Abstract
Introduction:
Pancreatic cancer (PC) is one of the most aggressive tumours, and better risk stratification among patients is required to provide tailored treatment. The meaning of radiomics and texture analysis as predictive techniques are not already systematically assessed. The aim of this study is to assess the role of radiomics in PC.
Methods:
A PubMed/MEDLINE and Embase systematic review was conducted to assess the role of radiomics in PC. The search strategy was ‘radiomics [All Fields] AND (“pancreas” [MeSH Terms] OR “pancreas” [All Fields] OR “pancreatic” [All Fields])’ and only original articles referred to PC in humans in the English language were considered.
Results:
A total of 123 studies and 183 studies were obtained using the mentioned search strategy on PubMed and Embase, respectively. After the complete selection process, a total of 56 papers were considered eligible for the analysis of the results. Radiomics methods were applied in PC for assessment technical feasibility and reproducibility aspects analysis, risk stratification, biologic or genomic status prediction and treatment response prediction.
Discussion:
Radiomics seems to be a promising approach to evaluate PC from diagnosis to treatment response prediction. Further and larger studies are required to confirm the role and allowed to include radiomics parameter in a comprehensive decision support system.
Introduction
Pancreatic cancer (PC) is one of the most aggressive tumours, representing the fourth cause of cancer-related deaths with 132,600 estimated new diagnoses, 128,000 deaths per year and accounting for 3.4–6.6% of all cancer cases, respectively.1,2 The incidence and mortality are slightly increasing during the years with a 5-year overall survival (OS) rate lower than 10%. 3 Males are more frequently affected than females and incidence in both sexes increases with age, reaching its peak in people older than 70 years. 4
Primary prevention is of utmost importance as there is currently no effective method of screening for the general population. Nevertheless, molecular or imaging tests can help early diagnosis in high-risk cohorts; for example, endoscopic ultrasound (EUS) and magnetic resonance imaging (MRI) allowed to detect precursor or invasive pancreatic neoplasms in asymptomatic people with an inherited predisposition (5–10% of all patients), thus increasing the resectability of the tumours and the overall prognosis of these patients. 5 Further data are needed to establish the most adequate approach and to select patients who could benefit the most from screening programmes.
Signs and symptoms are indeed not disease-specific and usually appear when it has already become unresectable or distantly spread.
Computed tomography (CT) is the most commonly used imaging diagnostic tool, supported by EUS with fine needle biopsy or aspiration in the identification of small lesions and in providing a definitive diagnosis. 6 MRI and positron emission tomography (PET) may also contribute to the systemic staging of the disease and in characterizing the primary tumour as resectable, borderline resectable or unresectable.
PC is a heterogeneous group of diseases; nearly 90% of the total are adenocarcinomas. Exocrine tumours are known to have a more aggressive behaviour compared with endocrine ones, with a median survival of about 4 versus 27 months, as reported in a large population-based study. 7 Pancreatic adenocarcinoma can progress from different types of precursor lesions, such as pancreatic intraepithelial neoplasia (PanIN) and intraductal papillary mucinous neoplasia (IPMN), under the push of consecutive genetic alterations whose significance is frequently unknown. 8 Some of these mutations have been recognized as having a role in different steps of carcinogenesis or even a prognostic significance, such as SMAD4 loss, which is linked to metastatic spread and lower survival rates. 9 These alterations may have, therefore, the potential to identify precursor lesions and serve as targets for new therapies. Currently, the first treatment option for PC is represented by radical surgery. Unfortunately, few more than 10% of patients are amenable to curative-intent surgery and prognosis remains poor even in this cluster of patients, with 5-year survival rates around 20%. 10
The quantitative analysis of medical images data and the extraction of imaging features, also called ‘radiomics’, represent an emerging approach in personalized medicine and advanced diagnostics, especially for disease characterization or outcome prediction.11–13 The interest towards radiomics is rapidly growing in the multidisciplinary cancer community as it shows an interesting pertinency and efficacy to answer several clinical questions arising in the management of patients affected by other gastrointestinal tumours.14–21
The aim of this study is to systematically assess and summarize evidence published in the scientific literature about the different applications of radiomics in PC.
Materials and methods
We followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) recommendations. 22 A systematic PubMed/MEDLINE and Embase search was performed using the following search strategy: ‘radiomics [All Fields] AND “pancreas” [MeSH Terms] OR “pancreas”[All Fields] OR “pancreatic” [All Fields])’.
Only original articles regarding radiomics applications in PC characterization were selected. Papers published between 1 January 2005 and 2 January 2021 were considered for this analysis. The exclusion criteria were as follows: (1) not original articles (e.g. reviews, editorials, letters, congress communications or posters, book chapters); (2) papers not referred to PC in humans (e.g. benign lesions, atrophy); (3) papers not referred to radiomics and (4) articles that were not in English, French, Spanish, Italian or German.
All the papers were selected and analyzed by a board of five radiation oncologists (ROs; CC, AD, FP, SM, AP), followed by an independent validation of three experts in PC (two ROs, GCM and FC, and one gastroenterologist, IB) and three experts on radiomics (two ROs, ND and LB, for the clinical point of view, and one physicist, DC, for the technical point of view). The whole process, the results and the discussion about potential discrepancies were validated by the other two different independent expert ROs (MAG and VV).
Results
A total of 123 studies were obtained using the mentioned search strategy on PubMed/MEDLINE, and 183 articles were obtained on Embase. Of these, 68 papers were selected based on the title according to the previously described criteria. The selection process, shown in Figure 1, led to the identification of 58 potentially eligible studies according to the abstract (two papers discarded according to the exclusion criterion 1; seven papers discarded for the exclusion criterion 2, and one paper discarded according to the exclusion criterion 3). After full-text analysis, one paper was discarded for the exclusion criterion 1 and one paper according to criterion 2. After the complete selection process, a total of 56 papers were considered eligible for the analysis of the results. All the studies included in the analysis are retrospective; the range of publication year is from 2017 to 2021. The participant centres’ nations were China (n = 26; 46.4%), the United States (n = 13; 23.2%), Italy (n = 6; 10.7%), Canada and Germany (n = 3; 5.4%), Japan (n = 2; 3.6%), France, Russia and South Korea (n = 1; 1.78%). The radiomics analysis was performed on CT (n = 43; 76.8%), PET-CT (n = 6; 10.7%) or MR (n = 7; 12.5%) images. The median number of patients involved in the analysis was 110 (range 10–422); the median number of features analyzed was 410 (range 1–3328). Radiomics, as resulted from the analyzed works, plays a role in many aspects of PC management.

Flowchart of the systematic literature search process.
Diagnostic imaging features were found to correlate with diagnosis and differentiation of pancreatic ductal adenocarcinoma (PDAC) from noninvasive diseases in 10 studies (17.9%).23–32
CT-based features were found able to distinguish PC from normal pancreatic tissue23,28 and to detect cancer in the context of intraductal papillary mucinous neoplasm (IPMN)27,30; CT- and PET-CT-based radiomics could discriminate PC from pancreatitis,24–26,31,32 while MRI was able to discriminate pancreatic neuroendocrine tumours (pNETs) from solid pseudopapillary tumours in one study. 29 Six studies (10.7%) analyzed technical feasibility and reproducibility aspects of radiomics analysis. Yamashita et al. 33 demonstrated that the variation between contrast-enhanced CT scans (e.g. scanner model, pixel spacing and contrast administration rate) affects radiomic feature reproducibility more significantly than the variation in segmentation. Mori et al. 34 confirmed the minimal impact of delineation uncertainty of pancreatic neuroendocrine neoplasms (panNEN) on CT radiomics features extraction. Plautz et al. 35 observed statistically significant changes over the radiotherapy course time in patients’ radiomics feature values, describing interesting delta-radiomics applications also in PC. Chu et al. 36 compared the diagnostic performances between a commercial and an in-house radiomic software. Loi et al. 37 evaluated the impact of interpolation and discretization on the robustness of radiomics features. The reproducibility of radiomics features in patients affected by pNETs was analyzed by Gruzdev et al. 38
A correlation between cancer imaging features and treatment response prediction was found in seven studies (12.5%).39–45 Cozzi et al. 39 analyzed CT-based radiomic features and found a significant correlation with local control (LC) and OS after stereotactic body radiation therapy (SBRT). Nasief et al. 42 investigated the additional predictive power of combining delta-radiomics features with CA 19-9 levels in patients undergoing concomitant chemoradiation therapy, with this combination resulting in an earlier prediction of good and bad responders. Simpson et al. 43 investigated the role of 0.35T MRI-based delta radiomics in the prediction of treatment response. Parr et al. 44 predicted clinical outcomes (OS and recurrences) using a CT-based radiomics model. Cusumano et al. 45 built a 0.35T MRI-based delta radiomics to predict LC. Yue et al. 40 evaluated the prognostic value of PET-CT texture variations in predicting treatment response. Nasief et al. 41 predicted pathologic response to the treatment using CT delta radiomics.
Overall, 15 studies (26.8%) were focused on radiogenomics, correlating image phenotype with specific gene expression, mutations, molecular or pathological findings.46–60 Of these, 8 studies found correlations between CT or MRI features and pNETs grading.50–54,57–59 One study conducted by Chang et al. 60 defined and validated a radiomic model able to predict histological grade in patients affected by PDAC. Ren et al. 46 developed a CT-based radiomic model to perform differential diagnosis between pancreatic adenosquamous carcinoma and PDAC. Attiyeh et al. 49 demonstrated the possibility to predict SMAD4 status and tumour stromal content using CT-based features. Two studies assessed the role of machine learning algorithms, derived from both CT and MRI analysis, in correlating clinical outcomes predictions with molecular and pathological tumour pathways.47,48 Other two studies proposed PET-CT- or CT-based radiomics to predict mutational status or PD-L1 expression. 55
In total, 21 studies (37.5%) described the application of radiomics in PC risk group stratifications, mainly in preoperative setting.40,47,48,61–78
Within this heterogeneous group of papers, four articles62,66,72,74 analyzed the role of CT-based radiomics in lymph nodes (LNs) metastasis risk assessment before surgery. The preoperative evaluation of the risk of a positive resection margin using CT-based radiomics analysis was the aim of three papers.58,69,76 Zhang et al. 61 evaluated the risk of occurrence of pancreatic fistula using a CT-based radiomic model. Overall, 13 articles 40,47,48,63–65,68,70,71,73,75,77,78 analyzed the role of radiomics to predict clinical outcomes, such as OS, progression-free survival (PFS) and early relapse.
Table 1 summarizes the main characteristics of the analyzed studies, including aim, conclusion and the cluster area of radiomic correlation.
Quantitative synthesis of the 56 selected articles.

18F-FDG-PET/CT, 18 F-fluorodeoxyglucose positron emission tomography/computed tomography; ADC, apparent diffusion coefficient; ALT, alternative lengthening of telomeres; CECT, contrast-enhanced computed tomography; CNN, convolutional neural network; CPH, Cox proportional hazard model; CT, computed tomography; DRFs, delta-radiomic features; DWI, diffusion-weighted imaging; ER, early recurrence; LC, local control; LN, lymph node; ML, machine learning; MRI, magnetic resonance imaging; NF-pNET, nonfunctional neuroendocrine tumours; OS, overall survival; panNEN, pancreatic neuroendocrine neoplasms; PDAC, pancreatic ductal adenocarcinoma; pNET, pancreatic neuroendocrine tumours; POPF, postoperative pancreatic fistula; rad-score, radiomics score; RF, radiomic features; RFS, restenosis-free survival; RT, radiation therapy; SBRT, stereotactic body radiation therapy; SMV, superior mesenteric vein; UPC-MBO, unresectable pancreatic cancer with malignant biliary obstruction.
Discussion
Radiomics seems to be an effective approach to evaluate patients affected by PC in several clinical settings and in different clinical contexts, from diagnosis to risk stratification, from biologic or genomic status prediction to treatment response evaluation or assessment. Figure 2 shows an example of clinical implementation of a delta radiomic model during a stereotactic radiotherapy treatment prescribed in five fractions.
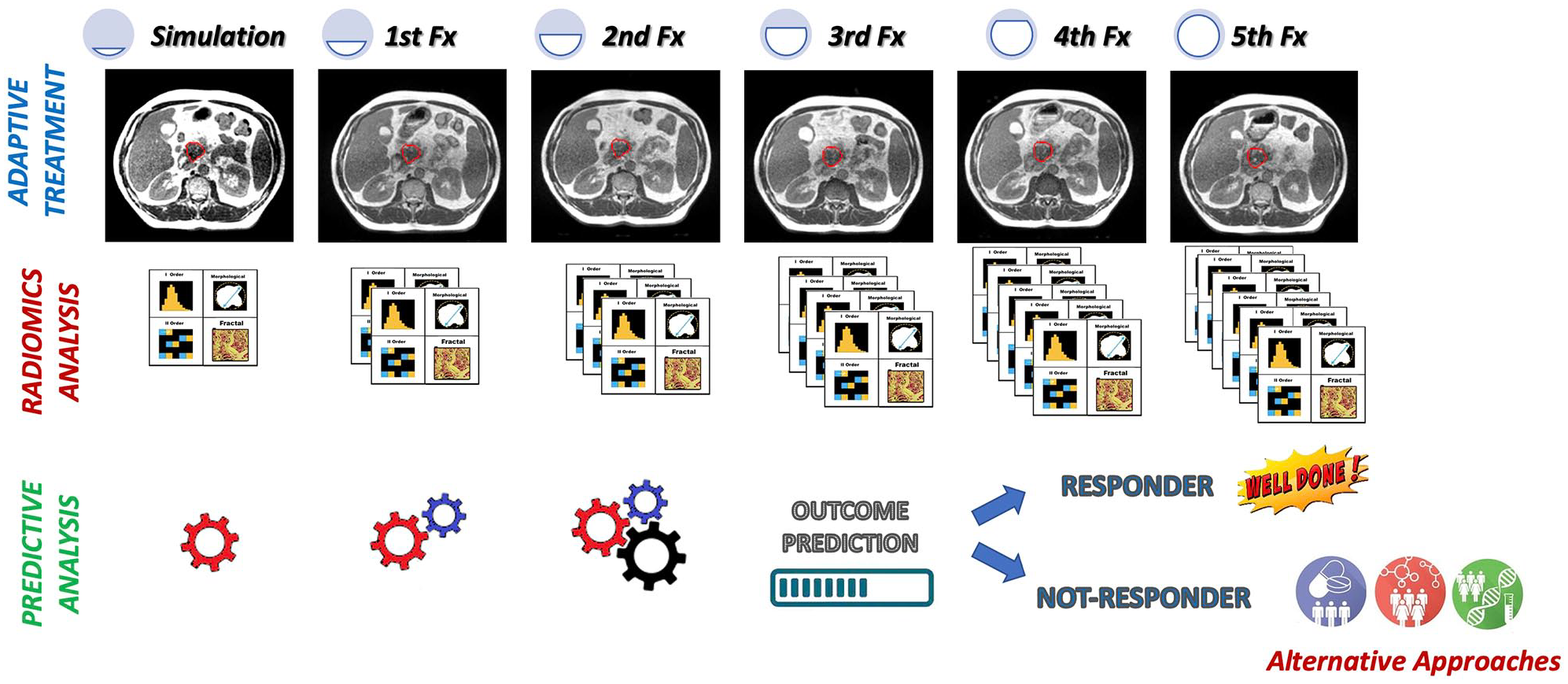
Example of clinical implementation of a delta radiomic model during a stereotactic radiotherapy treatment prescribed in five fractions. Before the start of the treatment, a radiomic model able to predict the LC 1 year from the end to the treatment was trained and tested on a retrospective cohort of patients. The model was based on the radiomic analysis of the MR images acquired during simulation and during fractions 1 and 2. Using the radiomic model, the RO can have a prediction of 1 year LC at the end of the fraction 2, so having the possibility to modify the radiation treatment for the remaining three fractions, increasing the dose or moving towards alternative approaches.
The common lack of technical standardization of the features extraction process and the necessity of external independent validation for each proposed predictive model hamper the feasibility of extensive radiomics studies in PC and impose the involvement of multidisciplinary research teams and the enrolment of larger patient samples.
Another limitation that reduces the generalizability of these observations is represented by the multiple kinds of image modalities and, for each of them, the variability of acquisition protocols. Moreover, the huge heterogeneity of features analyzed in the different studies poses a limitation in the possibility of creating a cluster of significant features for radiomics in this pathology. The same number of features analyzed, study by study, is very variable (from 1 to 3328 features per study) documenting that the resulting models are very much linked to the analysis experience of each centre.
Despite the above limitations, radiomics could potentially have an important role in providing reliable risk stratification (for both outcomes or complications), facilitating surgical choices, predicting clinical response after treatments, allowing differential diagnosis between cancer and other benign pancreatic abnormalities and predicting histological examination, disease differentiation grade or specific gene mutations.
The multidisciplinary management of these patients by dedicated teams of different specialists has proven to be a promising approach in terms of treatment quality and outcomes. 79
Radiation therapy (RT) can play a role in all possible scenarios, being particularly relevant in reducing the risk of relapse of resected cancer (adjuvant setting) and in pursuing LC in locally advanced disease (definitive setting).
Chemotherapy is also part of the management in all stages, and is administered in association with other treatment modalities (neoadjuvant, adjuvant therapy) or as a main part of the treatment for metastatic disease.
An important role to improve diagnostic and therapeutic options for PC patients is represented by the innovative approach of personalization through risk stratification, aiming to better define patients risk category and choose therapeutic patterns accordingly.80,81
Patients’ stratification is mainly performed based on the genotype, but the lack of a convergency in genomics models seems to reveal that the understanding of biological and clinical heterogeneity of PC is still far from completely understood.82,83 Recently, advances in imaging with the possibility of combining endoscopic modalities with image fusion techniques have provided new ways of approaching the diagnosis and stratification of PC.84–86
As part of an omics-guided care pathway, the integration of these modalities with artificial intelligence and deep-learning methods would seem to be a compelling prospect for pursuing personalized care in a highly heterogeneous disease.87–89 In this framework, radiomics is a promising method to be investigated that could provide more information about PC patients and its future integration in multiomics clinical support systems will allow more personalized and efficacious cancer care.
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
