Abstract
Purpose
Targeted cancer therapies often use a ‘one-size-fits-all’ dosing strategy, despite significant variability in individual drug exposure and its effect on both efficacy and toxicity. Therapeutic drug monitoring (TDM) is a precision dosing method that measures drug levels to guide dosing. This single-centre review investigates the potential benefits of TDM in cancer patients prescribed everolimus.
Methods
Medical records from Flinders Medical Centre, a public tertiary hospital in South Australia, were retrospectively reviewed for patients treated with everolimus for cancer indications between January 1, 2013, and December 31, 2023. Data on whole blood everolimus concentrations, cancer diagnoses, dosing regimens, survival outcomes and demographics were collected and analysed.
Results
During the study period, 53 patients with a median age of 64 years (range: 38-85) were treated with everolimus for oncological indications. Diagnoses included metastatic breast cancer (MBC, n = 41), metastatic renal cancer (n = 6), neuroendocrine tumours (n = 4) and recurrent ovarian cancer (n = 2). Among these, 14 patients (26%) underwent at least one everolimus blood level assessment. All MBC patients who underwent TDM-guided dose adjustments avoided treatment discontinuation due to adverse effects.
Conclusion
Prospective studies are warranted to establish therapeutic ranges and confirm the clinical benefits of TDM in cancer care.
Background
Despite advancements in precision oncology driven by targeted therapy, cancer drug dosing methods remain largely outdated, often following a ‘one-size-fits-all’ approach.1, 2 This dosing strategy does not account for individual patient variability in drug exposure, which can compromise treatment tolerability, reduce quality of life and lead to premature treatment discontinuation. 2 Initiatives such as Project Optimus highlight the importance of dose optimisation during oncology drug development, 3 but they do not fully address the challenges of optimising the dosing of already marketed therapies, many of which show poorer tolerability in real-world clinical settings than in tightly controlled clinical trial populations. 4 Precision dosing strategies such as therapeutic drug monitoring (TDM), pharmacogenomics and model-informed precision dosing have been proposed to fill this gap. 5
Everolimus, a targeted therapy with antiproliferative and immunosuppressive effects,6, 7 exemplifies the challenges and opportunities in precision dosing. It is approved for the prevention of organ transplant rejection, tuberous sclerosis complex and multiple cancer indications, including metastatic breast cancer (MBC), metastatic renal cancers (MRC) and neuroendocrine tumours (NET).8, 9 The typical dose in cancer ranges from 5 to 10 mg orally, with dose adjustments made when adverse effects become intolerable.10–13 A gradual dose escalation schedule beginning at 2.5 mg and increasing to 10 mg daily, has also been recommended to mitigate the risk of grade ≥3 stomatitis. 14
In contrast to oncology practice, TDM is mandated for transplant patients receiving everolimus to prevent graft rejection. 15 However, it is not routinely performed for cancer indications, 16 partly due to limited prospective studies defining optimal target concentrations, lack of clinician awareness and the frequent use of everolimus in combination with other therapies, which complicates monitoring.17–19 These challenges are compounded by the drug’s narrow therapeutic range and highly variable pharmacokinetics in cancer patients, influenced by drug interactions, genetic polymorphisms and organ dysfunction.20–22 Notably, a consensus report recommends maintaining everolimus trough concentrations between 12 and 20 µg/L for cancer indications. 15
Therefore, this single-centre review examines local prescribing practices and patient outcomes among oncology patients receiving everolimus, focusing on dosing patterns, TDM utilisation and toxicity profiles.
Methods
Study Design and Setting
This retrospective cohort study was conducted at the Flinders Medical Centre, a public tertiary hospital in South Australia. Ethics approval was obtained from the Southern Adelaide Clinical Human Research Ethics Committee, and the study was deemed low risk. The requirement for patient consent was waived as it met the criteria outlined in section 2.3.10 of the National Statement on Ethical Conduct in Human Research (2023). 23
Patient Identification and Data Collection
Electronic medical records and the oncology clinic database were reviewed to identify all patients who received everolimus for cancer treatment between January 1, 2013, and December 31, 2023. Patients were included in the study if they were documented to have been prescribed everolimus for cancer indications. Identified patients were then categorised into cohorts based on their cancer diagnosis for further analysis.
Data extraction was carried out by one author (JT) and independently verified by another (RH). Collected variables included demographic information, cancer type, treatment duration, survival data, everolimus dosing regimens, adverse events and TDM status. When available, everolimus whole blood concentrations were recorded alongside the sample timing relative to dosing (e.g., steady-state, trough level). For this study, everolimus blood samples collected between 23 and 25 hours post dose were considered trough levels.24, 25 The samples were also considered at steady state if patients had received the same dose for at least two consecutive weeks prior to sampling. 25
Within the MBC subgroup, patients who required modifications from the manufacturer-recommended dose of 10 mg for any reason (e.g., treatment interruptions, dose reductions, or initiation at a lower dose) were further subdivided into two groups. Those who had everolimus levels measured at any phase of therapy were classified as having ‘TDM-guided dose adjustments’, whereas those without level assessments were grouped into ‘standard-of-care dose adjustments’.
Outcomes and Statistical Analysis
The primary outcome was the frequency and pattern of TDM utilisation in patients receiving everolimus for cancer. For the MBC cohort, overall survival (OS) was calculated using the Kaplan–Meier method, with group differences assessed by the log-rank test. Cox proportional hazards regression was used to determine hazard ratios (HR) and 95% confidence intervals (CI), with a P value <.05 considered statistically significant. Spearman’s rank correlation coefficient (rs) was employed to evaluate the association between treatment duration and OS, while Cramer’s V (V) was calculated to explore the relationship between the method of dose adjustment and treatment discontinuation due to toxicity.
Descriptive statistics summarised dosing, toxicity profiles and survival outcomes for patients with other malignancies. Treatment end dates (for patients still receiving everolimus) and survival times for descriptive analysis (for patients still alive) were censored using the study end date if patients were still on everolimus or alive, respectively. In those who underwent TDM, clinical interventions—including dose interruptions, escalations, or discontinuations—were noted if they occurred near the time of blood sample collection. This assessment was based on the recommended reference range for everolimus in cancer treatment, regardless of whether the blood levels represented steady state or true trough concentrations. All statistical analyses were performed using R version 4.4.1.
Results
Patient Characteristics
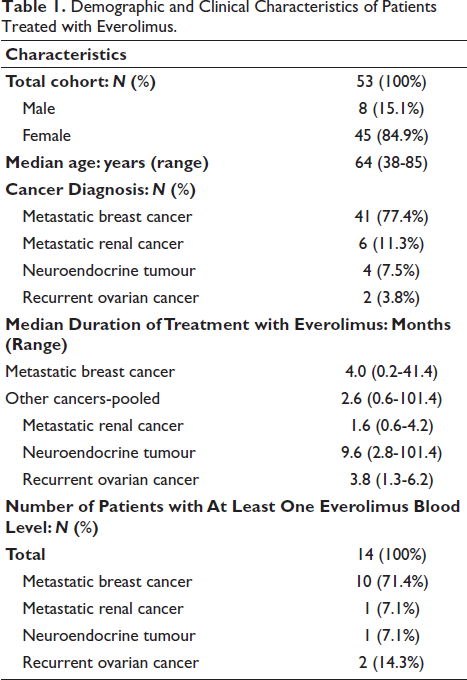
During the study period, 53 patients with a median age of 64 years (range: 38-85) were treated with everolimus for an oncological diagnosis. The distribution of cancer diagnoses included 41 patients with MBC, six with MRC, four with NET and two with recurrent ovarian cancers (ROC). Patient characteristics are summarised in Table 1.
Demographic and Clinical Characteristics of Patients Treated with Everolimus.
Everolimus Dosing and Therapeutic Drug Monitoring
Across all cancer indications, prescribed doses of everolimus ranged from 2.5 to 10 mg. Most patients (84.9%) started on a 10 mg once daily (OD) dose, while the remaining began at 5 mg OD. One patient’s starting dose was unknown. During the study period, 14 patients (26%) had at least one everolimus blood level measured (10 MBC, 1 ROC, 1 MRC and 2 NET), yielding a total of 34 concentration values. Two measurements (from a patient on everolimus for transplant rejection) were excluded for analysis (Table 2). A majority of patients who had undergone TDM had an everolimus dose of <10 mg at any point during treatment (11 of 14; 78.6%) compared to those that did not (5 of 39; 12.8%).
Everolimus Plasma Concentrations and Timing in Patients Undergoing Therapeutic Drug Monitoring.
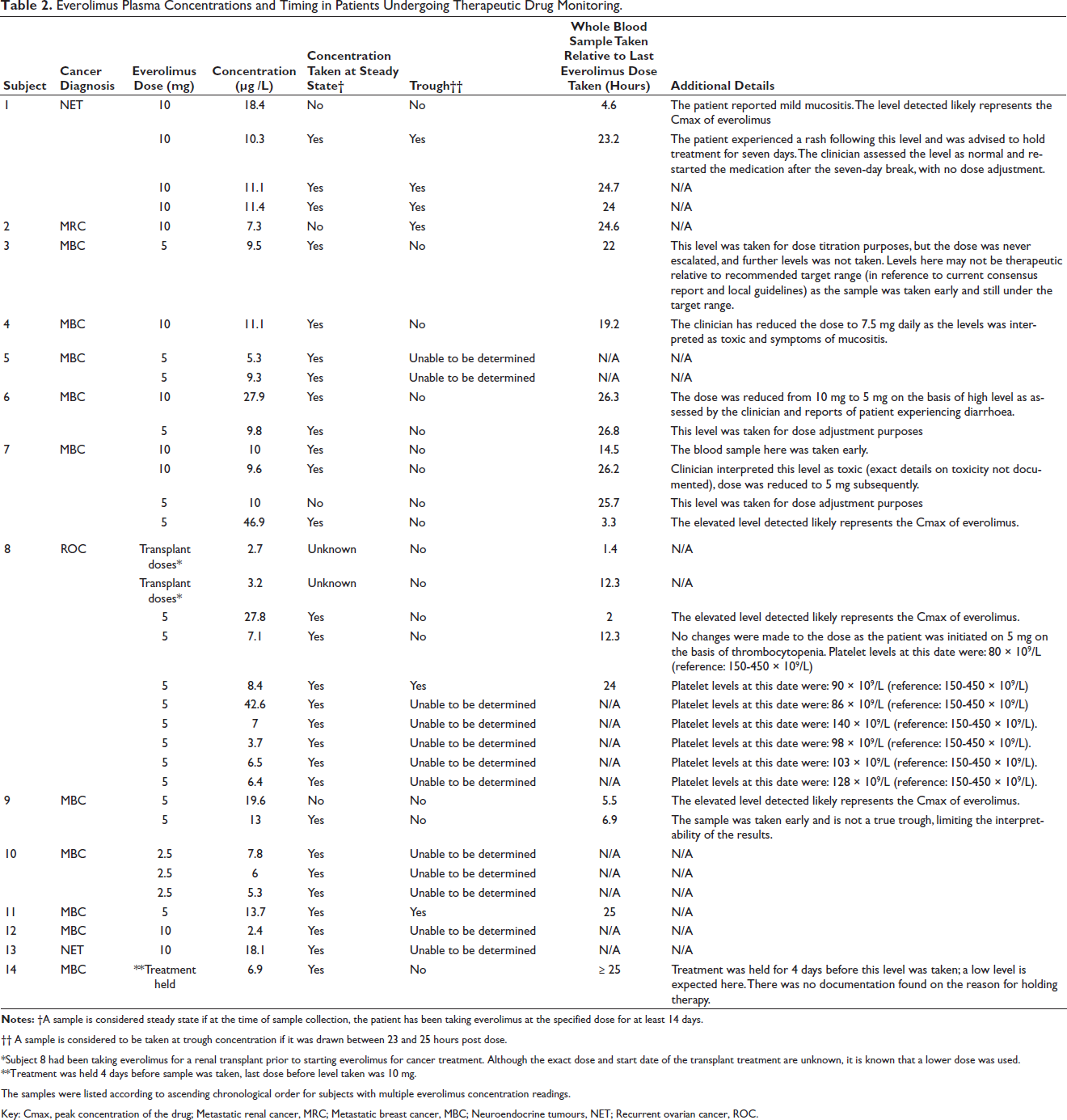
†† A sample is considered to be taken at trough concentration if it was drawn between 23 and 25 hours post dose.
*Subject 8 had been taking everolimus for a renal transplant prior to starting everolimus for cancer treatment. Although the exact dose and start date of the transplant treatment are unknown, it is known that a lower dose was used.
**Treatment was held 4 days before sample was taken, last dose before level taken was 10 mg.
The samples were listed according to ascending chronological order for subjects with multiple everolimus concentration readings.
Key: Cmax, peak concentration of the drug; Metastatic renal cancer, MRC; Metastatic breast cancer, MBC; Neuroendocrine tumours, NET; Recurrent ovarian cancer, ROC.
Everolimus whole blood concentrations varied widely in the cohort. In MBC patients, concentrations ranged from 2.4 to 46.9 µg/L; in ROC, from 3.7 to 42.6 µg/L; in NET, from 10.3 to 18.4 µg/L; and a single reading of 7.3 µg/L for MRC. Regardless of the target range utilised to assess the blood levels of everolimus in our cohort, a majority (>70%) of the levels that were collected were either under or above the recommended target range as proposed by the consensus report (12-20 µg/L) for cancer indications, 15 and our local laboratory for MBC (10-15 µg/L). Over 80% of the collected measurements were classified as non-trough or indeterminate due to incomplete information on sampling times (Figure 1).
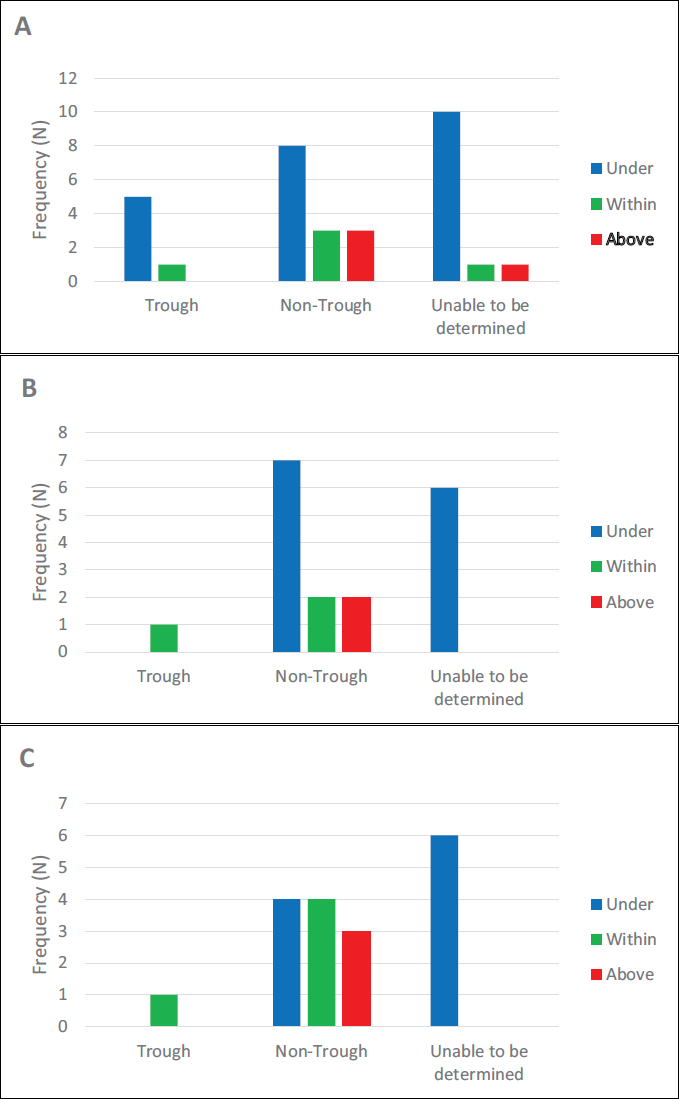
A: Using reference range of 12-20 µg/L, overall therapeutic drug monitoring cohort (N = 32).
Adverse Events
Adverse events were reported in 35 patients (66%). The most frequent were mucositis (n = 11) and pneumonitis (n = 6), with other events including rash, fatigue, diarrhoea, nausea, vomiting and anaemia. Of these 35 patients, 17 (49%) required dose reductions or treatment interruptions (14 with MBC; 3 with NET). Sixteen patients discontinued everolimus for reasons unrelated to disease progression. In 13 of these 16 patients, discontinuation was due to everolimus-related side effects; 11 (85%) had not undergone TDM.
Dose Adjustments and Discontinuation Due to Everolimus Side Effects in Metastatic Breast Cancer
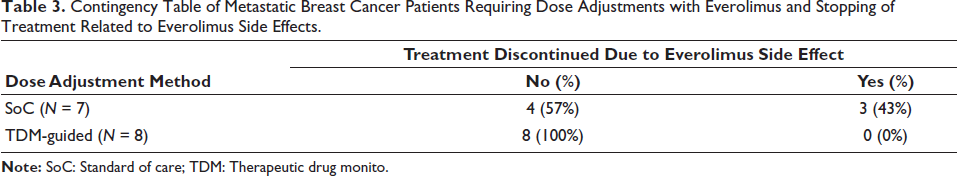
In the MBC cohort, 15 patients required dose adjustments during any point of treatment with everolimus. All patients (N = 8) who had dose adjustments guided by TDM did not discontinue everolimus due to treatment-related side effects (Table 3), and a strong association was found between the method of dose adjustments and stopping treatment due to everolimus-related side effects (V = 0.53). 26
Contingency Table of Metastatic Breast Cancer Patients Requiring Dose Adjustments with Everolimus and Stopping of Treatment Related to Everolimus Side Effects.
Clinicians’ Assessment and Interventions Guided by Everolimus Blood Levels
Four documented everolimus levels, from four patients (three with MBC: subjects 6, 7 and 10, and one with NET: subject 1), directly informed clinical management (Table 2). The recorded levels ranged from 9.6 µg/L to 27.9 µg/L (Table 2). The patients with MBC had their doses reduced by at least 25% after being assessed as having high blood levels. The NET patient had an everolimus level of 10.3 µg/L, which clinicians considered on target; however, treatment was temporarily halted for toxicity and then resumed at the same dose. Follow-up blood levels were obtained for the NET patient and two MBC patients after these interventions.
Overall Survival in the Metastatic Breast Cancer Cohort
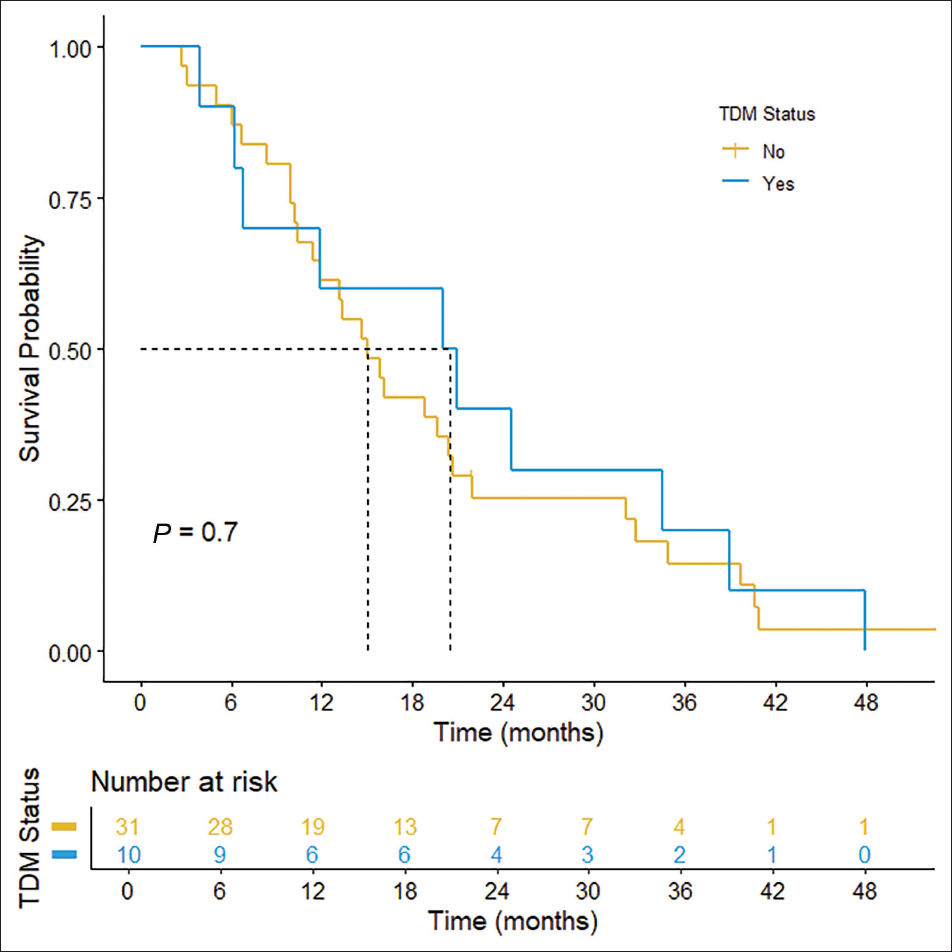
In the MBC cohort, one patient was still alive but was not receiving everolimus at the end of the study period. The median OS was 20.5 months (95% CI: 6.8 – not estimable) for patients who underwent TDM (n = 10) compared to 15.1 months (95% CI: 11.4-21.9) for those who did not (n = 31), although this difference was not statistically significant (P = .70; HR = 0.87, 95% CI: 0.42-1.79) (Figure 2). Four out of 41 MBC patients had missing treatment end dates, preventing the estimation of their treatment durations. The median treatment duration was 3.7 months (IQR: 2.1-4.7) in the TDM group (n = 9) and 4.2 months (IQR: 2.3-5.6) in the non-TDM group (n = 28). No significant correlation was observed between treatment duration and OS in patients who underwent TDM (rs = 0.60, P = .097). However, a moderate positive correlation was found between treatment duration and OS in patients who did not receive TDM (rs = 0.48, P < .05).
Overall Survival in Other Cancers
Among patients with MRC, NET or ROC, the median OS was 12.0 months (IQR: 9.9-35.6) for those who underwent TDM (n = 4) and 4.0 months (IQR: 2.6-9.5) for those who did not (n = 8). The median treatment duration was 9.9 months (IQR: 5.7-35.6) for TDM patients and 1.6 months (IQR: 1.1-2.5) for non-TDM patients. Two NET patients in the TDM group continued to receive everolimus at study end. No significant correlations were identified between treatment duration and OS in the TDM (rs = 1, P = .083) or non-TDM groups (rs = 0.55, P = .17).
Discussion
This retrospective analysis indicates that TDM for everolimus is underutilised in oncology and, when used, is often sub optimally applied, as a significant proportion of the samples were non-trough levels, limiting their utility for meaningful dose adjustments. Despite the potential benefits of TDM for optimising everolimus dosing, only 26% of patients in our cohort had at least one blood level measured. These findings highlight a gap in current clinical practices and the need for standardised TDM protocols in oncology settings.
In the MBC cohort, patients who had everolimus levels measured experienced numerically longer median OS, though this difference was not statistically significant. Variability in TDM practices (e.g., non-trough sampling) and small sample sizes likely limited the power to detect meaningful survival benefits. Patients with other malignancies (MRC, NET and ROC) also showed a trend towards prolonged OS and treatment duration when TDM was performed. These findings, though preliminary, support the hypothesis that TDM may enhance clinical outcomes by enabling more personalised dosing.
Consistent with prior studies, mucositis and pneumonitis were the most frequently reported toxicities.27, 28 However, our study recorded a lower overall incidence of adverse events and dose interruptions, likely due to incomplete medical documentation. Prior studies have reported a nearly 100% adverse event rate with everolimus in oncology, observed at doses similar to those described in our study.27, 29, 30 The large variability in everolimus whole blood concentrations, ranging from 2.4 to 46.9 µg/L, emphasises the need for individualised dosing. Furthermore, everolimus toxicity is related to high blood levels15, 18 and our study identified a strong association 26 (V = 0.53) between the dose adjustment method and treatment discontinuation due to side effects in our MBC cohort. Notably, none of the MBC patients requiring dose adjustments guided by TDM stopped treatment due to everolimus-related side effects. These findings suggest that TDM could help manage side effects safely through tailored dose adjustments.
Several factors may account for the low uptake and inconsistent application of TDM in this cohort. First, the absence of a universally accepted therapeutic range for everolimus in cancer, coupled with limited prospective evidence, reduces clinician confidence in acting on measured levels. 15 Second, local laboratory recommendations for an MBC-specific range (10-15 µg/L) only emerged during the latter half of the study period and may not have been widely disseminated; this also likely contributed to the higher proportion of MBC patients observed in the overall TDM cohort compared to other cancers. Furthermore, the only available consensus report on everolimus at the time (published in 2016) concluded that more studies were required to define concentration targets and clinical utility in oncology indications. 24 The presence of this statement in the consensus report may have discouraged clinicians from actively applying TDM for oncology indications. We also suspect that when TDM was applied, some clinicians may have inadvertently referenced the therapeutic range for transplant patients, which typically has lower targets than cancer treatment. This inference is supported by two levels (9.6 and 11.1 µg/L) being interpreted as toxic, despite being well below the toxic range for cancer indications.15, 24 Lastly, the frequent use of combination therapies, genetic variability and differing organ functions in cancer patients complicate TDM implementation.20–22
This study has several limitations. Its retrospective, single-centre design and small sample size, particularly in the non-MBC cohorts, limit the generalisability of our findings. Additionally, the absence of standardised TDM protocols and incomplete medical documentation may have introduced variability and underreporting of adverse events. Furthermore, because everolimus blood samples collected were either non-trough concentrations or unconfirmed as a trough due to incomplete information, interpretation of the blood levels and their correlation with survival and toxicity were limited in our study. We also lacked information on factors such as prior cancer treatments, performance status, toxicity grading and disease progression dates, which could have provided further context on the findings of our study. Lastly, potential confounding factors such as patient adherence, comorbidities and concomitant medications were not fully accounted for, which may have influenced the observed outcomes.
Our findings suggest that TDM for everolimus is underutilised in oncology despite potential benefits in optimising dosing to manage toxicity. Developing Bayesian models to estimate everolimus trough concentrations is essential to enhance the feasibility of everolimus TDM in real-world settings, especially given the challenges of obtaining trough samples as highlighted in our study. Randomised trials are needed to define target trough concentrations, confirm the benefits of TDM and establish standardised TDM protocols to improve treatment outcomes for cancer patients treated with everolimus. More broadly, our findings highlight an unmet need for precision dosing strategies in oncology, extending beyond the drugs targeted by current regulatory initiatives such as Project Optimus.
Footnotes
Data Availability
The data supporting this study will be made available upon reasonable request and subject to approval by the data custodians. Access may be granted in accordance with relevant ethical and institutional guidelines.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Approval for this study was obtained from the Southern Adelaide Clinical Human Research Ethics Committee (reference number:105.24, issued on 04 June 2024).
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Supported by Flinders Foundation, The Ryan Hodges Fund research grant. N.D.M is supported by research grants from The Hospital Research Foundation (2023-S-DTFA-005) and Tour De Cure (RSP-117-FY2023).
Patient Consent
The requirement for patient consent was waived as it met the criteria outlined in section 2.3.10 of the National Statement on Ethical Conduct in Human Research (2023).
