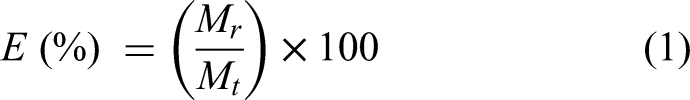Abstract
The significant carbon dioxide emission and high-energy consumption from traditional pyrometallurgical metal alloy production can be eliminated by using hydrogen as a reductant and processing materials in the solid state. In this study, an iron–nickel alloy was synthesised by solid-state hydrogen reduction of 250 g of a 50 wt% Fe2O3-NiO powder mixture. The alloy was produced in a horizontal Fe-Cr-Al tube furnace in multiple steps. The metal oxide mixture was exposed to a reducing atmosphere from room temperature, ramped up to 700 °C over 30 min, and held at 700 °C for 3 h. X-ray diffractometry of the reduced product confirmed the absence of oxides and the presence of body-centred cubic and face-centred cubic phases. A trace amount of oxygen was identified by melt-extraction analysis. The oxygen content results encourage pilot-scale iron–nickel production and the broader application of this process for large-scale metal alloy production from metallurgical by-products.
Introduction
The iron and steel industry is the largest industrial consumer of coal, contributing approximately 7–9% of direct global greenhouse gas emissions. 1 In 2023, the production of one tonne of crude steel emitted about 1.92 tonnes of CO2 and consumed 21.27 GJ of energy. 2 According to the International Energy Agency, a 20% reduction in emissions is required by 2030 and 93% by 2050 to meet net-zero targets. 3 In response, European steel producers have begun implementing low-emission technologies. Key mitigation strategies include replacing carbon-based reductants with hydrogen and biogenic alternatives, as well as transitioning from traditional pyrometallurgical processes to solid-state direct reduction. 4
Despite these efforts, global CO2 emissions from the sector continued to rise in 2024. However, a stable number of operational blast furnaces and increasing hydrogen-based direct reduced iron (DRI) projects signal incremental progress.5–7 Examples of organisations performing such work are Hybrit, SALCOS, and Siderwin. Parallel to industrial developments, metallurgical research also focuses on utilising sustainable alternatives to fossil coal.
As sustainable production of DRI is achieved and realised, metallurgical research is increasingly directed at addressing the next significant issue: improving metallurgical utilisation of by-products from iron and steel making.8,9 In 2016, only 6.8% of steelmaking slags were metallurgically utilised in Europe, while 76.6% were used for construction purposes. 10 The World Steel Association reported that 91% of basic oxygen furnace slags and 70% of electric arc furnace slags were used in road construction in 2017. 11 Enhancing metallurgical utilisation through hydrogen reduction of these by-products offers a path to recover valuable metals for secondary steelmaking. GreenIron in Sweden, for instance, is approaching full-scale implementation of this approach. 12
The current work aims to improve the metallurgical utilisation of the by-products. A fundamental study of the reduction of synthetic oxide mixtures is presented as a first step towards the reduction of industrial by-products. These by-products will not only contain such oxide mixtures, but also impurities and contaminants that will complicate reduction processes. An iron–nickel alloy was produced through hydrogen reduction of 250 g of a powder mixture with equal masses of pure iron and nickel oxides. The fundamental knowledge gained from this study will be applied to the large-scale reduction of by-products, and the experimental setup developed will also be used for this purpose. This study builds on the authors’ previous work involving smaller scale (0.1 g) reductions performed using a similar setup. 13 Figure 1 illustrates the fundamental studies conducted by the authors to date, as well as the planned study where the knowledge obtained from these studies will be applied.
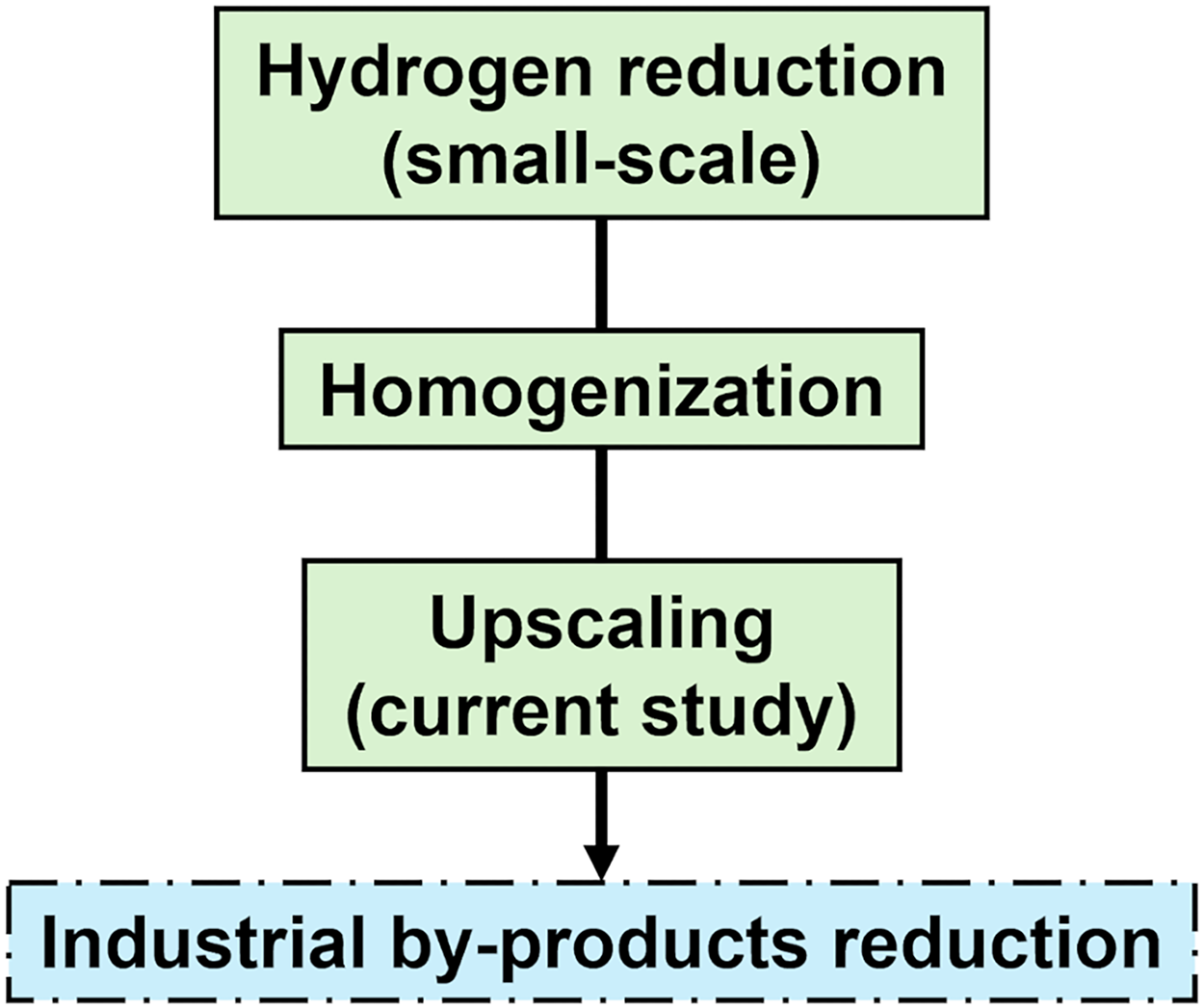
Flow chart showing completed fundamental studies (green, solid outline) and in-progress study (blue, dash-dotted outline).
The endothermic nature of hydrogen reduction and hydrogen being a weaker reductant than carbon at high temperatures challenge process scalability. In this study, using a horizontal fixed-bed reactor introduced an additional challenge of water retention in the furnace and crucible. The hydrogen-to-water partial pressure ratio strongly influences the reaction direction and rate: higher water partial pressure slows the reduction. Additionally, the retained water oxidises the reduced product. To prevent this, the reduction time and heating cycles were optimised, and the best cooling position in the furnace was determined. Future work will address water retention more effectively using a fluidised bed reactor. This reactor has already been used for direct hydrogen-based reduction in published literature, but only to produce metallic iron from fine haematite ore. 14
The iron–nickel alloy powder produced by the proposed process is chemically homogeneous on a macroscopic scale. Microscopic fluctuations in the chemical composition render it unsuitable for direct application in manufacturing. To address this, homogenisation at elevated temperatures over an extended period is required, and this process is currently being studied. However, the microscopic fluctuations fall within the typical limits observed in standard casting processes and can be addressed using conventional metallurgical treatments. This makes the powder suitable for alloy additions in secondary steelmaking. Iron–nickel was selected for this study due to its relatively simple reduction behaviour and lack of hazardous by-products. Chemically homogeneous iron–nickel may also offer a sustainable alternative to cobalt-based binders in tool steel production, as cobalt is classified as a critical raw material.15,16 Another area of interest is 3D ink-extrusion or filament deposition of oxide powders, followed by hydrogen reduction and sintering.17,18 Nevertheless, the primary objective of this work is to investigate the solid-state hydrogen reduction process and assess its scalability. The results presented here can serve as a foundation for adapting the method to other, more complex alloy systems.
Materials
The raw material for the reduction trials was prepared by mixing 50 wt% ferric oxide (Fe2O3, Sigma Aldrich Chemie GmbH, Steinheim am Albuch, Germany, country of origin: China, < 5 µm, > 96% purity) and 50 wt% nickel oxide (NiO, Sigma Aldrich Chemie GmbH, Steinheim am Albuch, Germany; country of origin: China; particles 44 µm; > 99% purity). These powders were mixed using a Retsch planetary ball mill (PM100) with a 500 ml alumina grinding jar and eight 30 mm alumina balls. Mixing was done at 100 rpm for 3 min, minimising mechanical alloying. The mixing direction was changed every minute to avoid clumping. Figure 2 shows the morphology of the 50 wt% Fe2O3-NiO mixture.

Backscattered electron image of the raw material: 50 wt% Fe2O3-NiO.
Experimental methods
The experimental setup involved a temperature-controlled horizontal resistance-heating furnace with a Fe-Cr-Al tube and a push-rod mechanism (Figure 3). The rod was connected to a hemicylindrical Fe-Cr-Al crucible. The tube was heated within a furnace region divided into three zones. The central zone temperature was controlled by a master controller, while slave controllers regulated the adjacent zones. Based on previous experience, adjacent zones were set 20 °C higher than the central zone to ensure uniform temperature across the furnace. The ends of the tube were water-cooled throughout the operation. One end included the push-rod mechanism and a connection to switch between the gas inlet and vacuum. The other end was connected to a split gas outlet, allowing off-gas to flow simultaneously through a flame hood and a connection for a mass spectrometer or a photoacoustic multi-gas monitor. However, the mass spectrometer was not used in this study, as harmful compounds were not expected in the off-gas due to the use of synthetic oxide powders free from such contaminants. Moreover, the laboratory-scale photoacoustic multi-gas monitor used in the previous study to measure water concentration in the off-gas could not be applied here, as its upper detection limit is approximately 14,000 ppm. 13
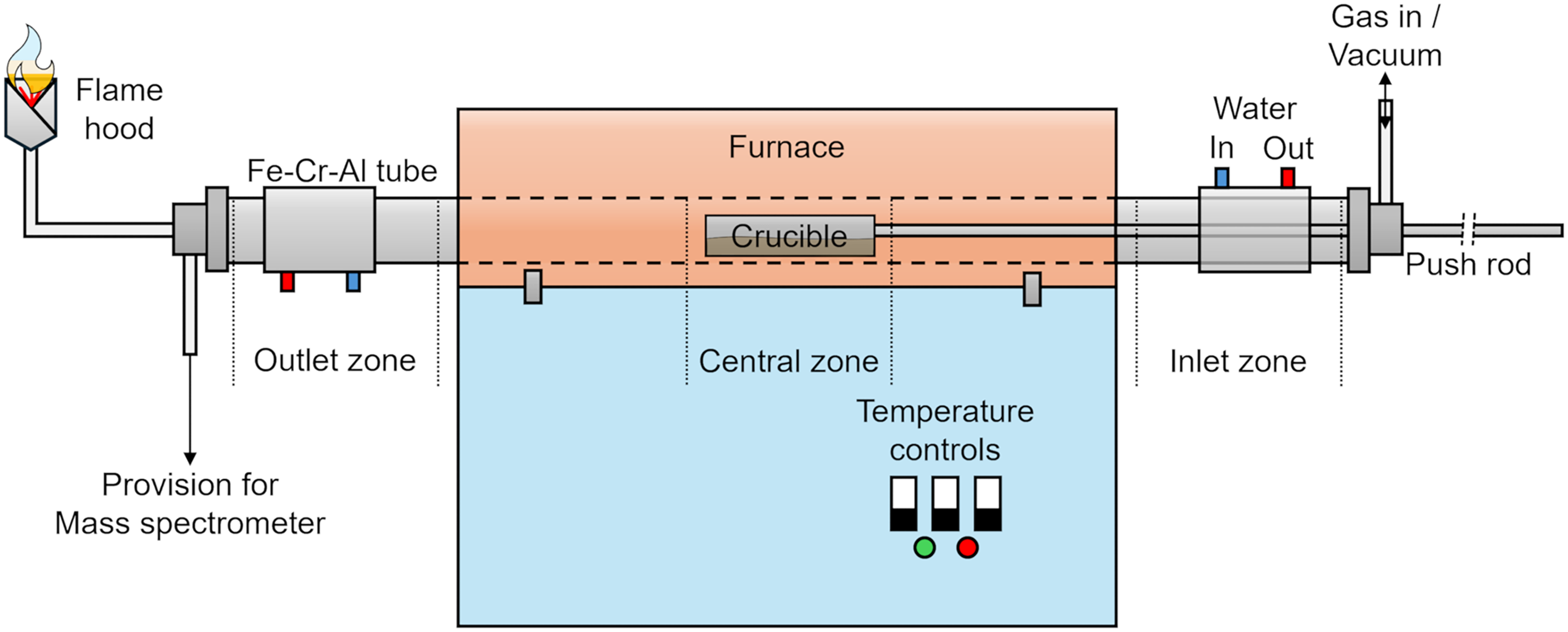
Experimental setup containing a water-cooled horizontal Fe-Cr-Al tube furnace with a push rod connected to Fe-Cr-Al crucible to handle the sample, a gas inlet, and a gas outlet with a connection to a flame hood and a provision for a mass spectrometer.
The furnace was heated to 120 °C and held for about 4 h to dry the tube. The crucible attached to the rod was filled with 250 g of oxide mixture and positioned at the tube's gas inlet end. Both tube ends were tightly sealed to create a gas-tight environment. The tube was evacuated and backfilled with 100% pure argon at 5 Nl min−1 three times. The oxide mixture was then pushed into the hot zone, and the tube was heated.
Five trials were performed (Figure 4). In trials 1 and 2, the tube was heated to 700 °C under argon. At 700 °C, argon was stopped, and 100% pure hydrogen was introduced. Excess hydrogen was ignited at the flame hood. A hydrogen flow rate of 5 Nl min−1 was maintained to prevent backflow and to sustain the flame. The oxide mixtures were reduced for 2 hours. After reduction, samples were cooled in the outlet zone for about 2 hours. The reduction temperature was based on the authors’ previous study. 13 However, its 45-min reduction time could not be directly applied, as the previous study involved only 0.1 g of sample and a hydrogen flow rate of 0.6 Nl min−1.
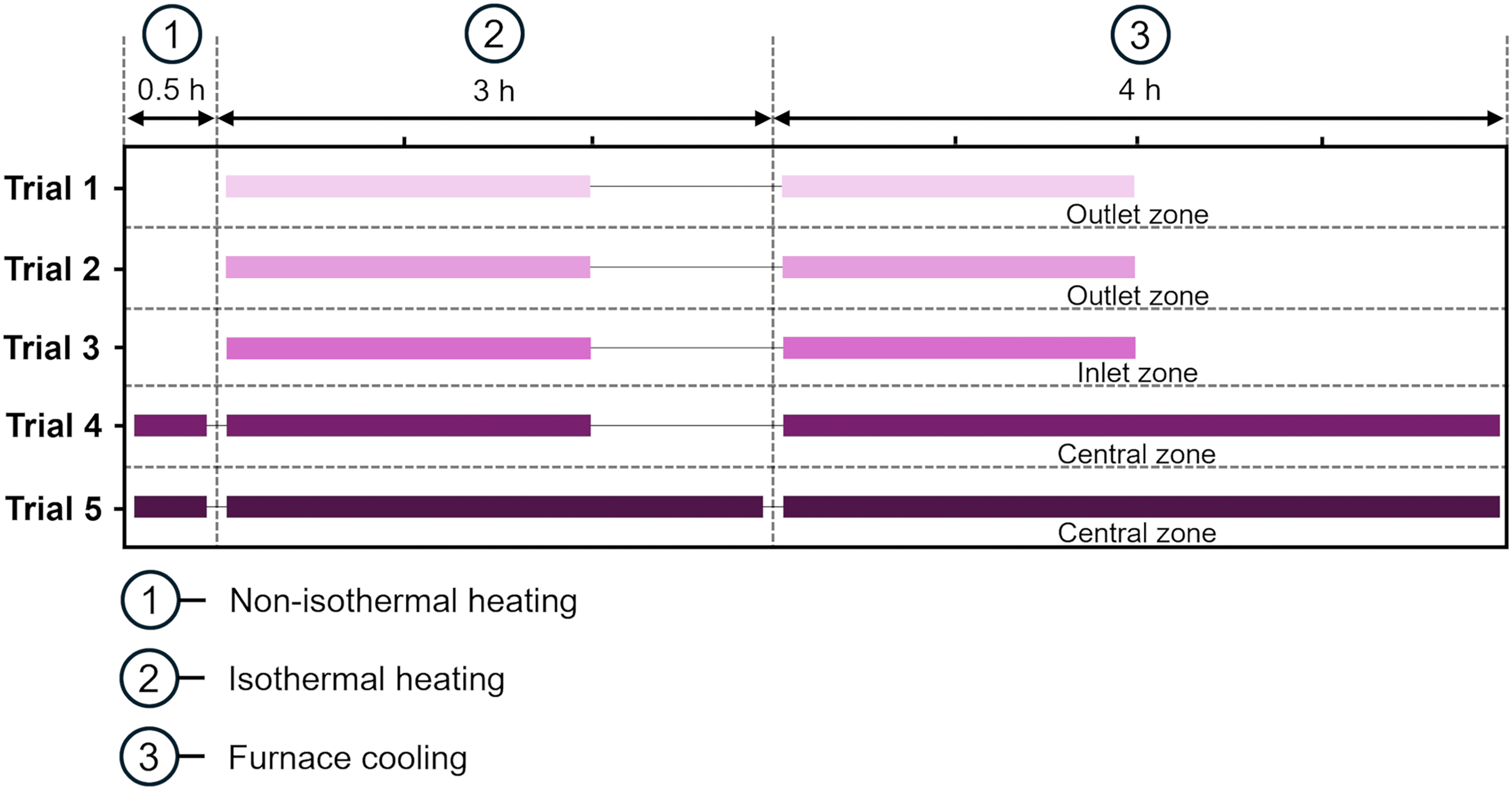
Design of experiments illustrating the variation across the different upscaling trials (1 to 5).
Initially, the reduction was expected to proceed faster in this study, as it was assumed that a hydrogen flow at 5 Nl min−1 would sequentially remove all oxygen from the raw material in 20 min. However, it was realised that 20 min could be too brief to remove 64 g of oxygen from the 250 g mixture, as it took 45 min to remove 0.03 g from 0.1 g in the previous study. Moreover, the expected advantage of a larger surface area of this study's mixture was not obtained because the ratio between the surface areas of the mixtures used in these studies was almost equal to the ratio between the quantities of oxygen in them. Based on this reasoning, a reduction time more than twice that of the previous study was used in trial 1.
Unlike trials 1 and 2, in trial 3, the reduced sample was cooled in the inlet zone. Trial 4 involved non-isothermal heating from room temperature to 700 °C over 30 min, followed by isothermal heating for 2 h, and furnace cooling for about 4 h in the central zone. All steps were performed under a hydrogen atmosphere. Trial 5 followed the same procedure as trial 4, except that the isothermal reduction was performed for 3 h.
Characterisation
The reduced product was divided into eight portions for sampling (Figure 5). Sample 2 from portion 2 represented the worst case, with likely minimal exposure to fresh hydrogen and a higher tendency to retain water formed during reduction. Sample 7 from portion 7 represented the best case, likely having the most exposure to fresh hydrogen and improved water evaporation. These assumptions were later confirmed by the physical appearance of the reduced product.
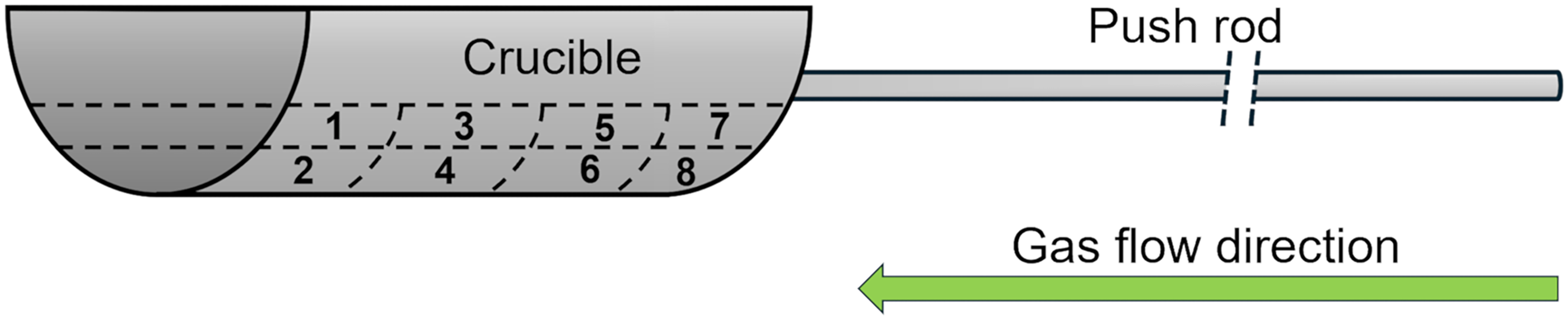
Fe-Cr-Al crucible (length = 24 cm, width = 9 cm, and height = 5 cm) divided into eight portions for sampling, with portions 2 and 7 representing worst-case and best-case scenarios, respectively.
The phases in samples 2 and 7 were investigated using X-ray diffractometry (XRD) immediately after each trial. The XRD was conducted with a Siemens D5000 diffractometer using a copper source operated at 35 mA and 40 keV. The diffractometer was operated in Bragg-Brentano geometry, with a graphite monochromator added to prevent iron fluorescence. Under these conditions, conventional laboratory XRD provides a practical detection limit of approximately 1 wt% for a minor crystalline phase. 19
Morphologies of the oxide mixture and reduced products were examined using secondary and backscattered electron imaging in a Hitachi S-3700N scanning electron microscope (SEM). SEM analysis was performed at an accelerating voltage of 15 kV with a working distance of approximately 12 mm. Energy-dispersive X-ray spectroscopy (EDX) with ESPIRIT software (v.19.1) was used to qualitatively assess the distribution of iron and nickel in the reduced product. A standardless EDX technique was employed, in which the system measures the peak-to-background ratio in the X-ray spectrum.
In upscaling trials 4 and 5, oxygen contents in samples 2 and 7 were quantified using melt-extraction analysis with a Leco ONH-836, following ASTM E1019.20,21
XRD was performed immediately after each trial, and SEM and melt-extraction analyses were conducted the following day. Samples were sealed in resealable pouches with air removed and stored in a vacuum desiccator. When transported, the samples were placed in an additional pouch containing desiccant.
Results and discussion
Upscaling trials 1–3
In upscaling trial 1, a gurgling sound from the flame hood was heard during reduction and cooling, indicating steam generation. Upon removal of the reduced product, water generated by the reduction was observed only in the outlet zone. It was expected from these observations that the stagnated water would have resulted in high-temperature reoxidation of the product. XRD plots (Figure 6(a)) of samples 2 and 7 collected immediately after the trial (Figure 5) confirmed significant FeO presence, indicating either reoxidation or incomplete reduction. 22 Two additional sets of peaks were also observed. One set had a lattice parameter a = 3.58 ± 0.001 Å, with systematic absences consistent with a face-centred cubic structure. This aligns with either Fe3Ni or FeNi3 phases, as supported by the Fe-Ni phase diagram. 23 Microscopic fluctuations in Fe and Ni composition, detected via EDX in SEM, support the coexistence of these two phases. BSE images (Figure 7) further revealed a few thin diffusion bridges between particles, indicating an early stage of sintering. This lack of sintering, combined with reoxidation, likely caused the observed fluctuations in composition.
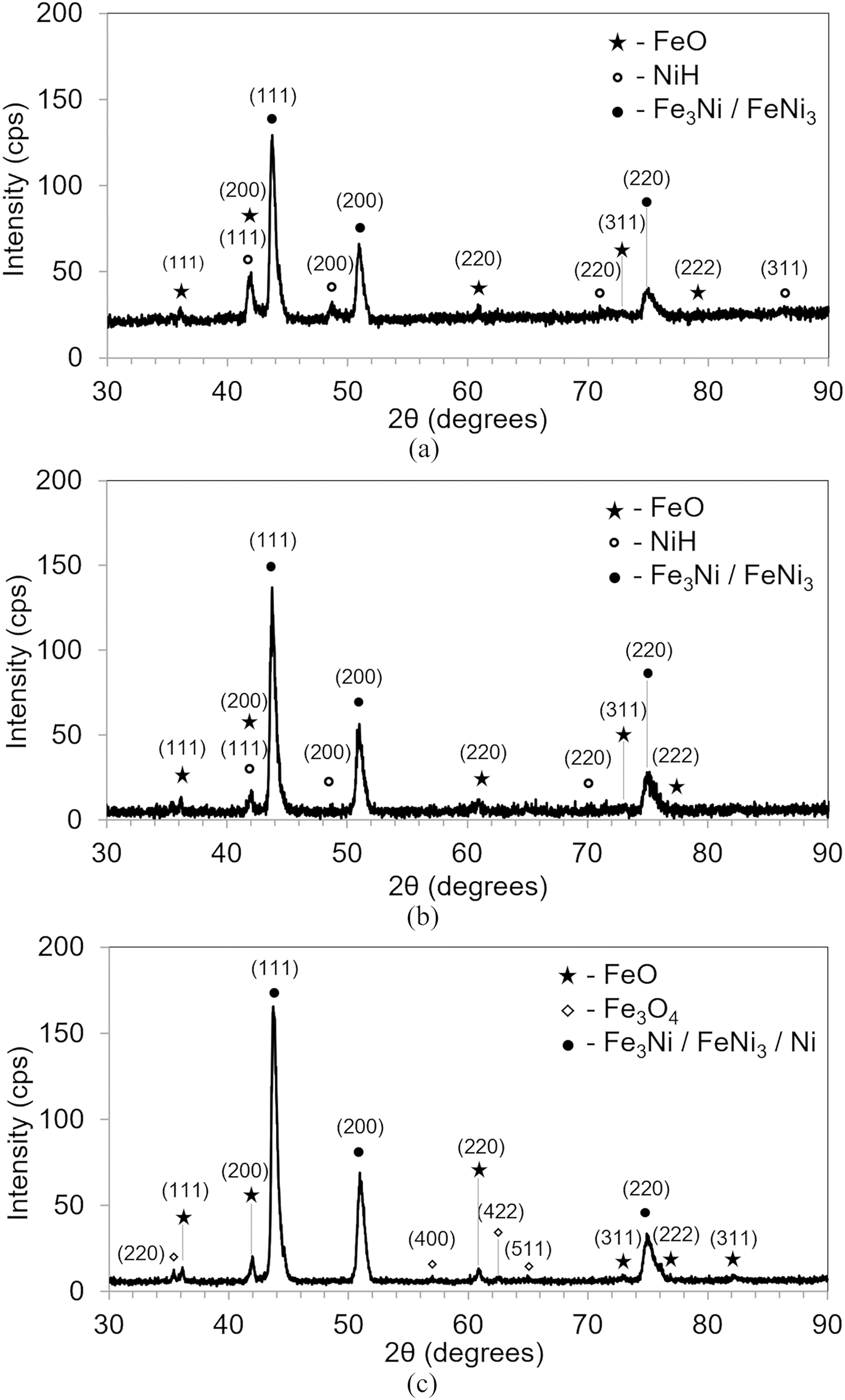
X-ray diffractograms of 47 wt% Fe-Ni alloy produced in upscaling trial 1, with analyses of sample 2 conducted: (a) immediately after the trial, (b) 10 days post-trial, and (c) 30 days post-trial.

Secondary electron image of 47 wt% Fe-Ni alloy (sample 2) produced in upscaling trial 1.
The second set of peaks had a lattice parameter a = 3.74 ± 0.001 Å, matching either a Fe3Ni phase with a primitive tetragonal Z1 structure (Z1_Fe3Ni) or a nickel hydride (Ni-H) phase with a face-centred cubic structure.24,25 Z1_Fe3Ni phase is described as a tetragonal structure with space group P4/mmm, having atomic positions: Ni at (00,0); Fe at (00,1/2), (1/2,1/2,1/4), and (1/2,1/2,3/4), with lattice parameters a = 3.703 Å and c = 10.472 Å. 24 Based on these values, expected Miller indices were identified, and corresponding structure factors, peak positions, and intensities were calculated. While eleven peaks were expected, only three were observed in the XRD plot. Notably, the (110) peak at 2θ ≈ 34°, which should have been twice as intense as the faint (220) peak at 2θ ≈ 72°, was missing. Other strong peaks, such as (004), were also absent. Their absence suggests the peaks do not correspond to the Z1_Fe3Ni phase.
New samples 2 and 7 analysed 10 days after reduction in a second XRD scan, again showed peaks potentially corresponding to the Ni-H phase (Figure 6(b)). The (111) and (200) peaks in plot (b) were observed to have lower intensities than in plot (a). The calculated lattice parameter (a) obtained from both plots precisely matched the reported value for nickel hydride. 25 Nickel hydride was synthesised in that study at 523 K and 2 GPa. At the lower pressure and temperature used in this study, the equilibrium solubility of hydrogen in nickel remains minimal.26,27
A third XRD analysis performed 30 days after the trial showed that peaks previously attributed to Ni-H had disappeared (Figure 6(c)). The test was repeated on fresh samples from the same portions, confirming the absence of those peaks. Comparing all three scans suggests the volume of Ni-H phase significantly decreased after 10 days and became zero 30 days later, consistent with the phase's instability at room temperature and atmospheric pressure.27,28 This supports the conclusion that the transient phase is Ni-H, not the stable Z1_Fe3Ni. The higher background noise in plots (a) and (b) compared to (c) is likely due to sample preparation anomalies.
Trial 2 was a repetition of trial 1, conducted to intentionally generate the Ni-H phase in the product and subject it to advanced characterisation techniques, including electron backscatter diffraction in SEM, focused ion beam with SEM, and atom probe tomography. However, the corresponding Ni-H peaks were not observed. Instead, XRD plots showed FeO and Fe3O4 peaks with similar intensities to those seen in the third scan of trial 1, suggesting comparable levels of reoxidation. In trial 3, the reduced product was cooled in the dry inlet zone to prevent reoxidation from stagnant water. Contrary to expectations, the XRD plot of sample 2 still showed FeO presence (Figure 8). A peak before the FeO (111) peak at 2θ ≈ 36° could not be identified, as related peaks were either missing or obscured by background noise. During this trial, the nut securing the push rod was repeatedly loosened and tightened as the crucible was placed into the inlet zone. This allowed air to enter and caused high-temperature reoxidation. Additionally, stagnant water in the outlet zone could have supplied oxygen, contributing to reoxidation despite cooling in a comparatively dry inlet zone. Condensation in the inlet and outlet zones occurred because these zones were continuously water-cooled throughout the trials. In subsequent trials, the product was cooled in the central zone, the driest part of the tube.
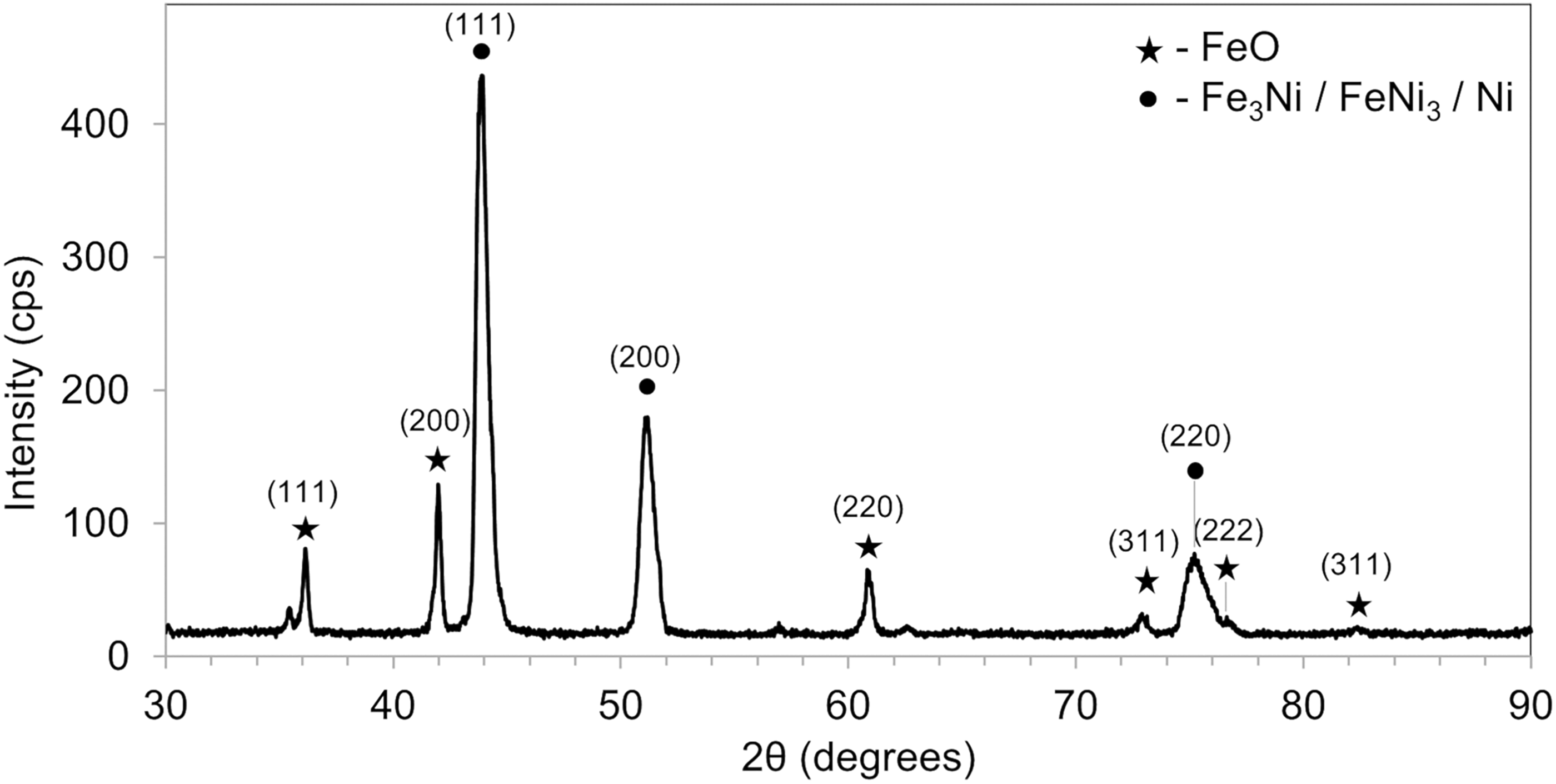
X-ray diffractogram of 47 wt% Fe-Ni alloy (sample 2) produced in upscaling trial 3.
Upscaling trials 4 and 5
In trial 4, the mixture was non-isothermally heated to 700 °C, and the product was cooled in the central zone for 4 h. The cooling followed an isothermal reduction at 700 °C for 2 h. All steps were performed under a hydrogen atmosphere. This trial aimed at facilitating water evaporation by breaking down bulk water generation into smaller steps. However, the gurgling sound persisted, and water stagnated at the outlet zone. XRD analysis revealed ferritic iron, Fe3Ni, and FeNi3 phases (Figure 9(a)). These phases are consistent with the Fe-Ni phase diagram and the slower cooling rate. Like the previous trials, a substantial amount of FeO was also detected (Figure 9(a)). Oxygen content was analysed in the worst-case sample (portion 2) and best-case sample (portion 7), as discussed in the Characterisation subsection. Portion 7 on the surface had 0.58 wt% oxygen, while portion 2 beneath the surface had 1.59 wt%. Assuming retained water oxidised iron at high temperatures, the higher oxygen content beneath the surface suggests water retention near the crucible's inner wall.
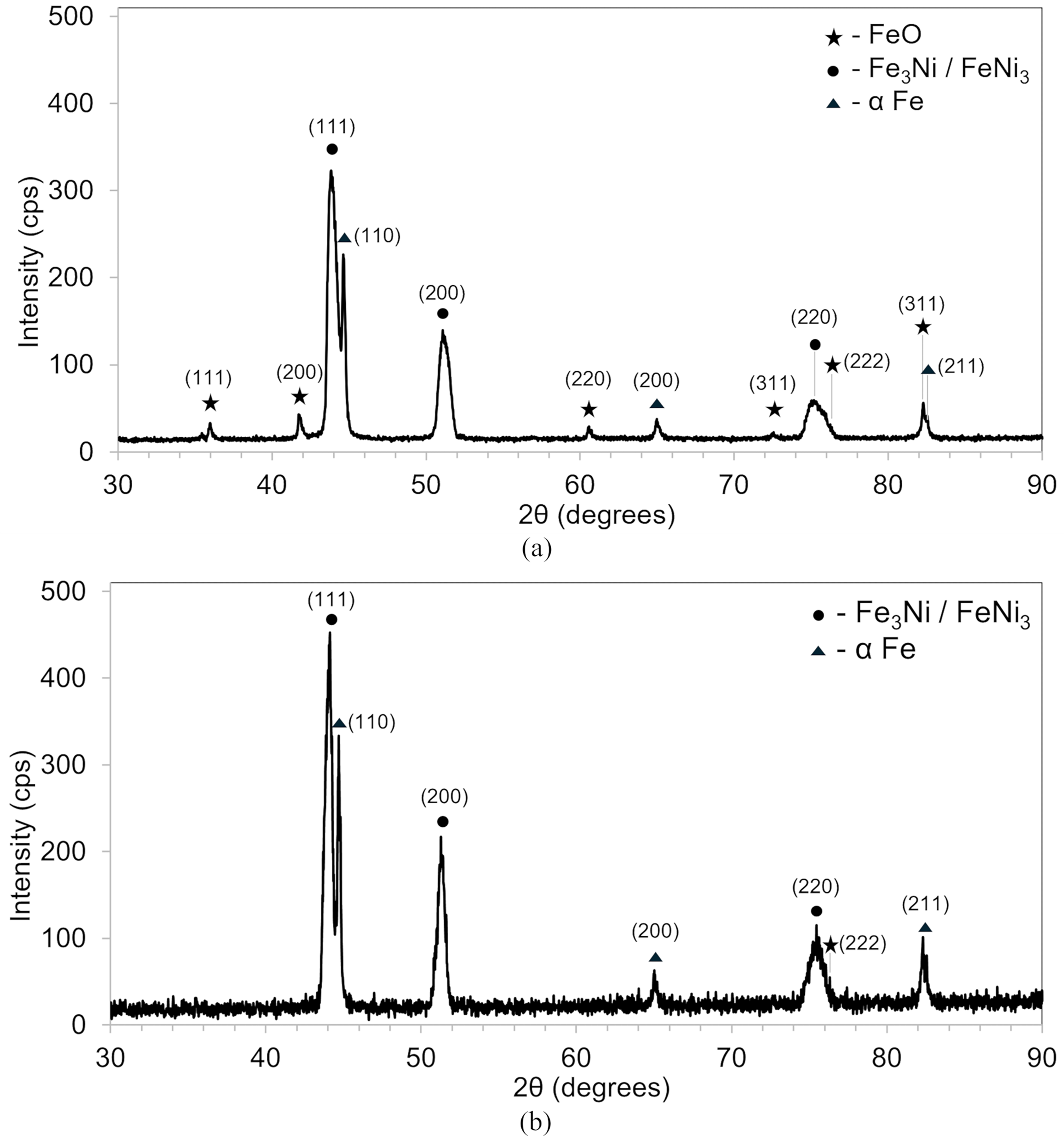
X-ray diffractograms of 47 wt% Fe-Ni alloy (sample 2) produced in upscaling: (a) trial 4, and (b) trial 5.
In earlier trials, the product's surface in the crucible appeared dry and crust-like, while the interior was moist and loose. This suggests that water generated during reduction near the inner surface did not fully evaporate or was not effectively removed by the gas stream. To address this issue, the isothermal reduction time was extended to 3 h in trial 5. Despite these efforts, gurgling persisted, and water stagnated at the outlet zone. While the crucible surface still appeared dry and crust-like (Figure 10), the portion beneath was drier than in trial 4. XRD analysis showed ferritic iron, Fe3Ni, and FeNi3 phases (Figure 9(b)). The oxide peaks were not observed, implying that the reduction was complete. The suspected Ni-H phase was also absent. Oxygen content analysis found only 0.37 wt% oxygen at the surface (portion 7) and 0.45 wt% beneath (portion 2), both lower than in trial 4. This suggests that the extra hour of reduction enhanced hydrogen availability near the inner surface, promoting the forward reaction. Furthermore, the similar oxygen content throughout the product indicates uniform dryness.

Photographs of the powder bed in the crucible from upscaling trial 5, showing: (a) the raw material (50 wt% Fe2O3-NiO) surface, and (b) the reduced product (47 wt% Fe-Ni) surface.
Using the average oxygen content, the total mass of oxygen removed from the mixture,
The chemical homogeneity of products from trials 4 and 5 matched earlier results. EDX maps of samples from trial 4 (Figure 11) and trial 5 (Supplemental Figure S1) showed multiple iron-rich and nickel-rich regions. The BSE image (Figure 11) reveals early-stage interparticle necks, indicating that the product is in the initial stage of sintering. The macroscopic chemical homogeneity of iron–nickel alloy makes it suitable for metallurgical processes involving liquid phase alloying, such as secondary steelmaking. Furthermore, solid-state homogenisation could make it suitable for direct applications, increasing its utilisation.

Backscattered electron image and energy-dispersive spectroscopy mapping of 47 wt% Fe-Ni alloy (sample 2) produced in upscaling trial 4.
Conclusion
Fe-Ni alloy was synthesised by solid-state hydrogen reduction of a 250 g batch of a 50 wt% Fe2O3-NiO powder mixture. The static powder bed was heated from room temperature to 700 °C over 30 min and held at this temperature for 3 h. The process concluded with cooling in the centre of the furnace. All these steps were performed under a hydrogen atmosphere. Success of the upscaled reduction was confirmed by the presence of less than 0.5 wt% of oxygen in the product, as measured by melt-extraction analysis, and the absence of oxide peaks in the X-ray diffractogram. In contrast, a similar procedure with a 2 h isothermal reduction resulted in approximately 1.5 wt% of oxygen, attributed to oxidation at high temperatures caused by water retention near the crucible's inner surface.
In an unsuccessful upscaling trial involving only a 2 h isothermal reduction at 700 °C, a nickel–hydrogen phase appeared in the X-ray diffractogram immediately after reduction. It disappeared after 30 days, implying that it was thermodynamically unstable at room temperature. The phase was absent in a repeat trial with the same parameters.
The main conclusions of the study are the following:
The hydrogen reduction process previously developed by the authors to synthesise less than 1 gram of metal alloys can be scaled up to yield hundreds of grams. The oxygen removal efficiency of the scaled-up process is 98.8%, which shows that the process is efficient in removing oxygen. The reduction process can be applied to large-scale metal and metal alloy production from industrial by-products, thereby increasing the metallurgical utilisation of these by-products. Carbon dioxide emissions associated with metal alloy production can be eliminated through the solid-state hydrogen reduction process identified in this study, provided that the electricity used comes from sustainable sources.
The chemical homogeneity of the reduced product will be enhanced using a previously developed method, making it suitable for direct manufacturing. Techno-economic feasibility can be evaluated against traditional pyrometallurgical methods once the process is further scaled up. Before the process is used for by-product reduction, it will be further validated by the reduction of higher order oxides.
Supplemental Material
sj-docx-1-mpe-10.1177_25726641261439460 - Supplemental material for Scaling-up a hydrogen reduction process to synthesise iron–nickel alloy from a mixture of metal oxide powder
Supplemental material, sj-docx-1-mpe-10.1177_25726641261439460 for Scaling-up a hydrogen reduction process to synthesise iron–nickel alloy from a mixture of metal oxide powder by Arun Kamalasekaran, Pelle Mellin, Pär Göran Jönsson and Christopher Hulme in Mineral Processing and Extractive Metallurgy
Supplemental Material
sj-docx-2-mpe-10.1177_25726641261439460 - Supplemental material for Scaling-up a hydrogen reduction process to synthesise iron–nickel alloy from a mixture of metal oxide powder
Supplemental material, sj-docx-2-mpe-10.1177_25726641261439460 for Scaling-up a hydrogen reduction process to synthesise iron–nickel alloy from a mixture of metal oxide powder by Arun Kamalasekaran, Pelle Mellin, Pär Göran Jönsson and Christopher Hulme in Mineral Processing and Extractive Metallurgy
Footnotes
Acknowledgements
The authors at KTH gratefully acknowledge financial support from the Swedish Energy Agency under project numbers 49836-1 and P2023-00941 within the Industriklivet program.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Energimyndigheten, (grant number 49836-1, P2023-00941).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data regarding this research article can be obtained from the authors upon request.
Supplemental material
Supplemental material for this article is available online.