Abstract
Thiacalix[4]arene with four sulfur-bridging atoms and four hydroxy coordinating groups in the lower rim is a promising candidate for powerful electrocatalytic hydrogen production. However, there are only a few examples reported in the literature where conjugated thiacalix[4]arene is used as a pre-catalyst for the hydrogen evolution reaction. The present work evaluates the electrocatalytic performance of a nickel(II) coordination complex based on generic thiacalix[4]arene. The coordination complex is characterized by cyclic voltammetry, where two reduction peaks are observed at −0.57 and −1.33 V for the two redox couples Ni2+/Ni+ and Ni+/Ni, respectively. The proton reduction occurs at the second peak potential with a slight shift at about −1.0 V, with increasing peak currents directly related to the number of acetic acid equivalents. The maximum peak current was observed to be at about 67 µA for 20 equiv. of acetic acid.
Keywords
Introduction
The discovery of eco-friendly and sustainable energy sources is an important target for researchers because of the depleting levels of fossil fuels and environmental concerns.
1
Electrochemically generated hydrogen is considered as a promising alternative energy source. However, it is necessary to use a catalyst for its production, either by proton reduction or water splitting. In attempting to improve the performance of catalysts for the hydrogen evolution reaction (HER) using low-cost transition metals, many researchers have developed nanostructured compounds.2–8 In addition, transition metal-based catalysts with macrocyclic organic frameworks such as porphyrin have become the subject of intensive studies by many scientists in recent years.
9
Among the many macrocyclic ligands, calix[n]arenes have emerged as a unique core because of their tunable bowl-shaped supramolecular cavity and easy variation/derivatization.10–12 They constitute chemically and thermally stable circular platforms that can accept up to four coordinating units containing heteroatoms such as phosphorus, sulfur, nitrogen, and oxygen. Besides, these macrocycles can be easily obtained by condensation between para-tert-butyl phenol and formaldehyde. Depending on the base, temperature, and reaction time, they can be prepared with different sizes ranging from 4 to 20 phenol groups connected through methylene bridges.13–15 Calix[4]arenes

The chemical structures of calix[4]arene (
Recently, the electrocatalytic activities of calix[n]arenes have started to attract more attention in HERs, glucose oxidation reactions (GORs), hydrogen oxidation reactions (HORs), oxygen evolution reactions (OERs), oxygen reduction reactions (ORRs), and carbon dioxide reduction.17–26 Interestingly, TC4As require further studies, and to the best of our knowledge, only a very few papers report their use in photocatalytic and electrocatalytic energy production.19–23 Wang et al. 19 reported an ultrafine Pt nanocluster confined in a calixarene-based {Ni24} coordination cage, which catalyzes the electrochemical production of hydrogen at low overpotential with a high current density. In addition, glassy carbon electrodes modified by calixarene-functionalized graphene oxide composites for HER and OER have been reported by Zhang et al. 24 Genorio et al. 25 have demonstrated good selectivity toward hydrogen oxidation and oxygen reduction by patterning of platinum with C4A molecules. In all the above-mentioned examples, researchers have used noble metals as electrocatalysts, even if they are confined within the calixarene at very low concentrations.
Very recently, Liao and co-workers 20 reported the electrocatalytic oxidation of glucose using a thiacalixarene-based {Ni18} coordination wheel with onset and peak potentials of 0.3 and 0.46 V, respectively, in an alkaline medium. Besides, the encapsulation of a metallic cluster such as AuPd inside the cavity led to a higher current density than the commercially available Pt/C electrode toward the HER. Recently, the same authors reported four nickel-thiacalixarene complexes, an {Ni8} cluster, an {Ni20} coordination wheel, an {Ni24}coordination bowl, and a twisted double-bowl-shaped {Ni32}, in the presence of 2,5-dihydro-1,3,4-thiadiazole-2,5-dithiol. Efficient electrocatalytic activity toward glucose oxidation was apparent for the {Ni20} coordination wheel, which was attributed to its accessible active nickel sites and a through-hull channel at an appropriate size. 21 Earlier, we demonstrated the electrocatalytic activity of a polyoxothiomolybdate derivative toward hydrogen evolution while the oxygen-containing equivalent, a polyoxometalate derivative, could not promote the electrocatalytic reaction. 27
Sulfur-embedded ligands and the corresponding complexes show better electrocatalytic performances than the oxygen analogues. We herein present the electrocatalytic performance of a nickel(II) TC4A complex toward hydrogen evolution on a glassy carbon electrode.
Results and discussion
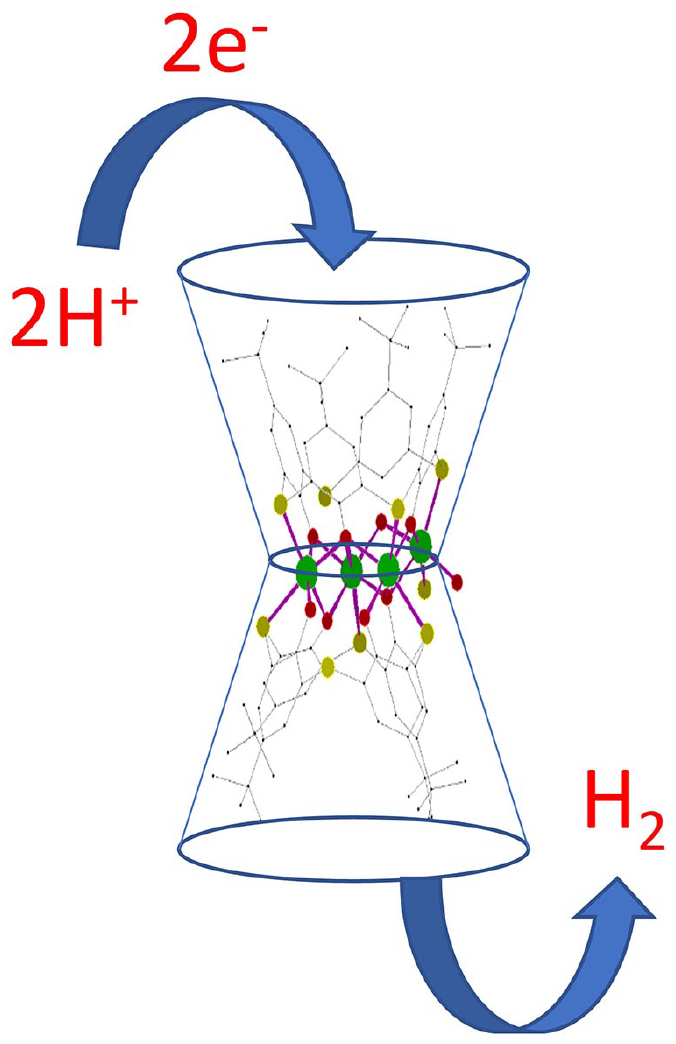
Compared to C4A, the S-containing counterpart (TC4A) possesses delocalized electrons, making it a powerful candidate for electrochemical applications, including HER. The TC4A molecule has hydrophobic cavities formed by benzene rings and tert-butyl groups, capable of hosting organic molecules. In addition, the TC4A molecule contains free electron pairs on sulfur and the oxygen atoms of hydroxy groups, which can form chelates with metal ions through coordinate–covalent bonds. Metal ions, for example, nickel, in complexes with the thiacalix[4]arene are necessary for electrocatalytic reactions. As shown in Figure 2, the prepared nickel complex, obtained from mixing TC4A and [Ni(DMSO)6](ClO4)2 in DMF in the presence of triethylamine (see section “Experimental section”), contains two parts interconnected by an Nipart1–Spart2 bond. 28 The first moiety is formed from two TC4A units connected through four nickel ions using three sulfur and four oxygen donor atoms. The second part is composed of two TC4A units, where the first is coordinated to two nickel ions through its four oxygens and two sulfur atoms. The nickel ions are then coordinated to three oxygens and two sulfurs of the second TC4A units. The presence of the six nickel ions close to the lower rims of the TC4As and the small sizes of both H+ and H2 gas indicated that the nickel ions catalyze the proton reduction reaction in a confined space close to the hydrophobic cavity.

Structure of
The complex was characterized by cyclic voltammetry in DMF solution containing 0.2 M tetrabutylammonium tetrafluoroborate ([NBu4][BF4]) and complex

Cyclic voltammetry of the Ni(II) complex (2.5 mM) in DMF solution containing [NBu4][BF4] (0.2 M) at the carbon electrode versus Ag/AgCl at a scan rate of 100 mV.

Cyclic voltammetry of Ni(II) complex
where A is the surface area exposed to the electrolyte in cm2, Cf is the capacitance in Farads, including both double-layer capacitance, and v is the scan rate. The anodic peak currents for the electrode in the investigated electrolyte are shown in Figure 4 as a function of the square root of the scan rate. Linear dependence was obtained, which indicates a non-absorption process at the electrode.
Complex

Cyclic voltammetry obtained from the catalyst-free solution of DMF containing CH3COOH (20 mM) and [Bu4N][BF4] (0.2 M), scan rate 100 mVs−1 at a glassy carbon electrode under N2.

(a) Cyclic voltammograms obtained from using solutions of DMF containing Ni(II) complex
In the past, researchers have also investigated the electrocatalytic potential of Ni(II)-based coordination complexes for proton reduction into hydrogen. For example, Ni(II) coordination complexes

The chemical structures of different porphyrin derivatives used for HER in the previous studies.
Conclusion
A nickel complex based on the TC4A ligand has been synthesized. Its electrochemical activity for proton reduction into hydrogen has been evaluated. Cyclic voltammetry studies at negative potentials show two reduction peaks for Ni2+/Ni+ and Ni+/Ni at Epred = −0.57 and −1.33 V. The presence of a catalyst results in a potential shift of about 850 mV to a more positive potential for the second peak potential. In addition, the peak current increases based on the number of acetic acid equivalents until a maximum of 67 µA for 20 equiv. of acid. The studied nickel(II) complex gave better electrocatalytic results compared to some well-known nickel(II)-based porphyrin derivatives. The presence of cavitands in the complex structure with a hydrophobic interior suggests a catalytic process in a confined space where the hydrogen is formed on nickel sites close to the lower rim of the TC4A ligand.
Experimental section
Materials
All starting materials, including dry solvents, were purchased from Afaq Sada Trading Est and used as received. Chemicals including 4-tert-butylphenol, sulfur, tetraethylene glycol dimethyl ether, sodium hydroxide, hydrochloric acid, 37% triethylamine, and solvents were obtained from Merck or Acros Organics. The TC4A ligand was synthesized according to a protocol described in the literature. 32 All analytical data were consistent with the literature data and confirmed the structure of the synthesized complex.
Synthesis
Ni6(TC4A)(TC4A-H)2(TC4A-H2)·4CH3CN·6H2O (
Electrochemical measurements
Electrochemical measurements were performed at 25 °C using a standard three-compartment cell with a working glassy carbon electrode (surface = 0.07 cm2), a reference Ag/AgCl electrode and platinum wire as the counter electrode. Before use, the working electrode was polished using emery papers of different grades, then washed with the used solvent and dried. A potentiostat (Autolab PGSTAT 128N) and NOVA 1.10 software were used for recording and processing the electrochemical data.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by the Scientific Research Deanship at University of Ha’il, Saudi Arabia, through project no. RG-20 058.

