Abstract
Natural polyamines such as putrescine, spermidine, and spermine are crucial in the cell proliferation and maintenance in all the eukaryotes. However, the requirement of polyamines in tumor cells is stepped up to maintain tumorigenicity. Many synthetic polyamine analogues have been designed recently to target the polyamine metabolism in tumors to induce apoptosis. N4-Erucoyl spermidine (designed as N4-Eru), a novel acylspermidine derivative, has been shown to exert selective inhibitory effects on both hematological and solid tumors, but its mechanisms of action are unknown. In this study, RNA sequencing was performed to investigate the anticancer mechanisms of N4-Eru-treated T-cell acute lymphoblastic leukemia (ALL) cell line (Jurkat cells), and gene expression was examined through different tools. We could show that many key oncogenes including NDRG1, CACNA1G, TGFBR2, NOTCH1,2,3, UHRF1, DNMT1,3, HDAC1,3, KDM3A, KDM4B, KDM4C, FOS, and SATB1 were downregulated, whereas several tumor suppressor genes such as CDKN2AIPNL, KISS1, DDIT3, TP53I13, PPARG, FOXP1 were upregulated. Data obtained through RNA-Seq further showed that N4-Eru inhibited the NOTCH/Wnt/JAK-STAT axis. This study also indicated that N4-Eru-induced apoptosis could involve several key signaling pathways in cancer. Altogether, our results suggest that N4-Eru is a promising drug to treat ALL.
Introduction
Acute lymphoblastic leukemia (ALL), characterized by the production of immature lymphocytes, has been one of the major causes of deaths worldwide. 1 In 2015 alone, nearly 876 000 people were affected by ALL and resulted in 111 000 deaths around the globe.2,3 Mostly, children between the ages 2 and 5 are likely to be affected and with the strides in the development of chemotherapy, the 5-year survival rates have increased from 10% in the 1960s to 90% in 2015. 4 However, the adults affected with ALL have a lower survival rate with an aggregate of 35% in elderly patients. 5 Despite the recent developments and novel approaches in designing treatment regimens, there is limited information regarding the targeted molecular pathways and mechanisms involved. 6 The causative factors leading to the development of ALL may be prolonged exposure to environmental carcinogens, genetic mutations, genetic syndromes, and delayed infections.4,7,8
Many previous studies have reported the use of diverse classes of drugs for the treatment of ALL such as thiopurines, glucocorticoids, vincristine, dexamethasone, hyperfractionated cyclophosphamide, and methotrexate.9–12 Specific antibodies in immunotherapy including blinatumomab and inotuzumab ozogamicin13,14 that target CD19 and CD22 are underway. However, due to relapse in the treatment and the adverse effects due to high doses, there is a persistent need to develop novel drugs which can be incorporated in the current chemotherapeutic intervention.
The natural polyamines such as putrescine, spermine, and spermidine are found in all the eukaryotic cells at micromolar concentrations. 15 These biogenic polyamines are crucial for the normal cell proliferation, differentiation, and nuclear stabilization. 16 Nevertheless, in the cancerous state, the requirement for these polyamines rises manifold and the polyamine metabolism pathway can be targeted through the use of polyamines from the external source resulting in cell death.17,18 During the recent few years, the development of polyamine analogues as chemotherapeutic agents against different cancers has fascinated many researchers.18–22 Many polyamine analogues targeting epigenetic modifications of the chromatin were evaluated previously and have shown promising results. The effects of polyaminohydroxamic acid derivatives and polyaminobenzamides as histone deacetylase (HDAC) inhibitors23,24; (bis)guanidine or biguanide polyamine analogues as lysine-specific demethylase (LSD1) inhibitors in in vitro have been extensively studied. 25 T-cell ALL mainly involves dysregulation of the NOTCH1 signaling by constitutive activation of the pathway in nearly 50% to 60% cases. 26 The modulation of the other key genes such as UHRF1, DNMT1, HDAC1, JAK1, BCL3, IDH2 CDKN1A, and RPL527–30 have also been shown to be implicated in the development of T-cell ALL.
Recently, we demonstrated the proapoptotic effects of 4 novel acylspermidine analogues on Jurkat (JK) and MCF7 breast cancer cells; one of these analogue-N4-Erucoyl spermidine (N4-Eru) has shown selective inhibitory effects on both types of tumors. 31 The aim of this study was to investigate and analyze the differential gene expression (DGE) of N4-Eru-treated JK cells through RNA-Seq and to reveal the signaling pathways which could be targeted to induce apoptosis in JK cells.
Material Methods
Cell culture and treatment
T-cell ALL JK cell line was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were maintained in a humidified incubator at a temperature of 37°C and 5% CO2. RPMI1640 media supplemented with 10% fetal bovine serum (catalog no. 16000-044; Lifetech, Carlsbad, CA, USA) and 1% penicillin-streptomycin antibiotics (100 units/mL, catalog no. 15140-122; Lifetech, Carlsbad, CA, USA) was used to maintain cells.
RNA-Seq and differentially expressed genes analysis
To investigate the anticancer mechanisms of N4-Eru and gene expression, JK cells were treated at a concentration corresponding to its half-maximal activity (30 µM) for 24 hours in triplicates. Total RNA was extracted using RNeasy kit Qiagen, Valencia, CA, USA, and the RNA concentration was quantified. RNA quality was assessed using Bioanalyzer (RNA integrity number score >7.0). Then, the sequencing libraries were generated (TruSeq Stranded mRNA Sample Preparation Kit; Illumina, San Diego, CA, USA) from 2.5 μg of total RNA from each of the 3 biological replicates. The 50-bp (base pair) long single-end deep sequencing was performed using Illumina HiSeq 2000 system. The adaptor sequences were removed, and the low-quality base call was filtered by FASTX-Toolkit. The obtained filtered short sequencing reads were mapped to the human genome using TopHat2, and the subsequent gene expression values were quantified using Subreads package Feature Counts function. The differentially expressed gene (DEG) analysis was further performed from the gene expression values after identifying the library size and appropriate data set dispersion. 32 The DGE was tested using R/Bioconductor package edgeR. Differentially expressed genes are determined by log2 fold change (Log2FC) and false discovery rate (FDR; log fold change [LogFC] ⩾0.5 or ⩽−0.5; FDR ⩽ 0.05).
Bioinformatics analysis
Gene set functional analysis was performed using Gene Ontology (GO) search through Enrichr tool.33,34 The gene IDs of interest were converted to EntrezID and loaded to DAVID bioinformatics tools 35 for pathway analysis. The GO analysis and Biocarta/KEGG pathway analysis were then performed by setting all the GO terms and Biocarta pathway genes as background genes. 36 Overrepresented GO terms or pathways were determined by enrichment score (EASE ⩽ 0.1, gene count ⩾2). The heat map of genes responsible for cell death regulation was plotted using the web tool ClustVis as per the mentioned algorithm. 37 In addition, gene interactions of the antiproliferative and apoptotic-inducing genes were represented by employing the tool GeneMania. 38
Statistical analysis
The statistical analysis between the control and the treated samples (the Student “t” test and one-way analysis of variance) was performed using GraphPad Prism 6 software (GraphPad, San Diego, CA, USA). Results are presented as mean ± SEM of triplicates in the same experiment or 3 independent experiments. The significant statistical differences have been designated as *P < .05, **P < .01, and ***P < .001.
Results
N4-Eru-modulated gene expression
Data obtained from JK cells treated with 30 µM of N4-Eru showed that the messenger RNA (mRNA) expression of 1683 genes was altered, with 1052 genes downregulated (LogFC < −1.0; P < .05) and 631 genes upregulated (LogFC > 1.0; P < .05). The number of transcripts altered has been classified based on the LogFC or the “P” value as described in Tables 1 and 2. Altogether, the number of transcripts downregulated exceeded the number of transcripts upregulated.
Total number of transcripts altered in N4-Eru-treated Jurkat cells.
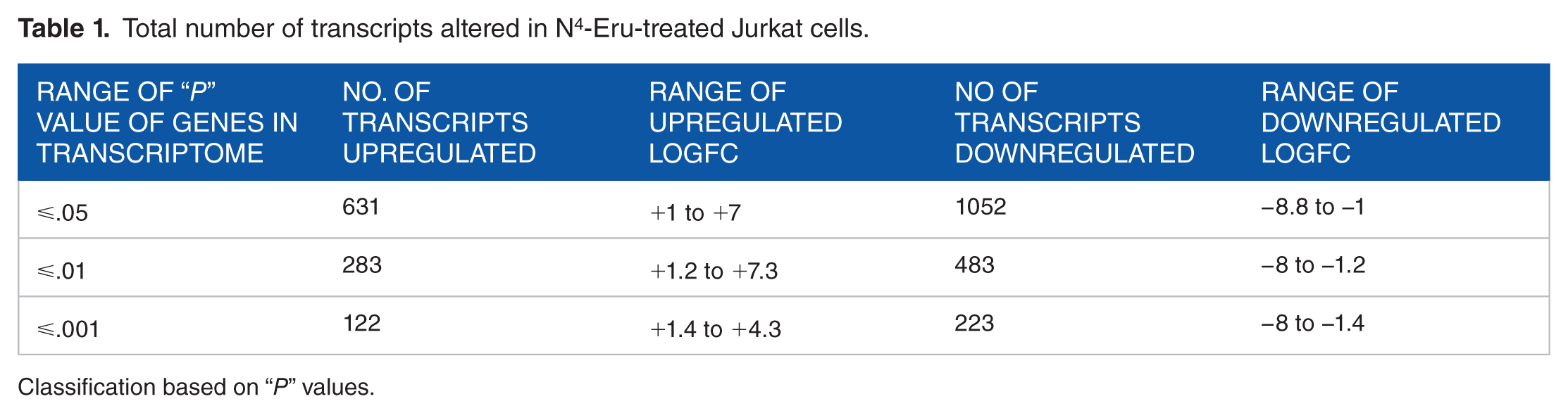
Classification based on “P” values.
Total number of transcripts altered in N4-Eru-treated Jurkat cells.

Classification based on Log fold change (LogFC) values.
Gene enrichment analysis of transcripts
The gene enrichment analysis (P < .01) of GO terms showed that there was an overall decrease in the cell proliferation and negative regulation of the proapoptotic genes accompanied by an increase in the expression of tumor suppressor genes. The GO analysis summary of different biological processes, molecular functions, and cell components in downregulated and upregulated transcripts in N4-Eru-treated JK cells are described in Figures 1 and 2, respectively.
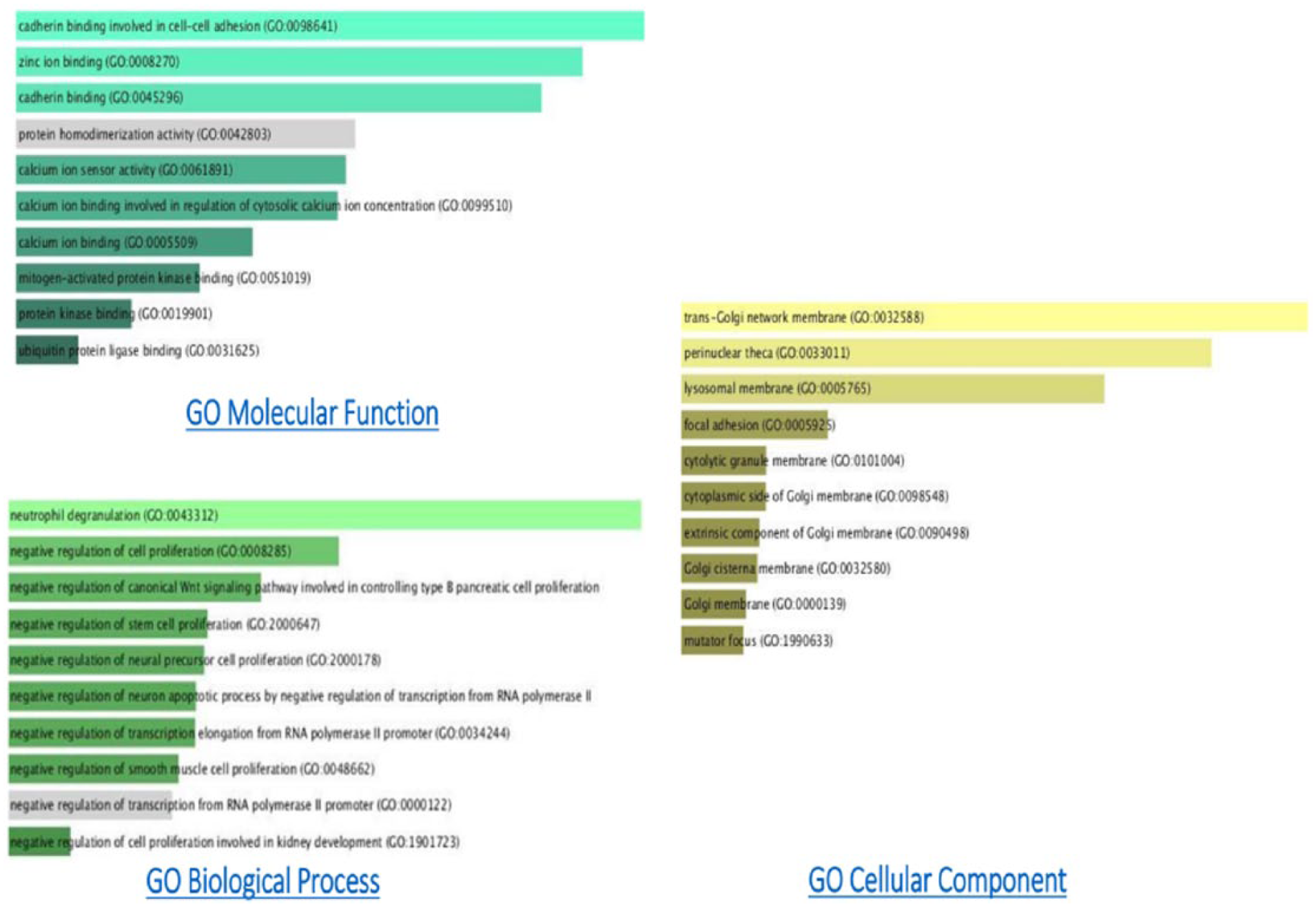
Annotation statistics of downregulated GO terms of the N4-Eru-treated Jurkat cells. The length of the bar represents the significance of that specific gene set or term. In addition, the degree of the brightness of the color denotes the significance (P < .01) of the differentially expressed genes.

Annotation statistics of upregulated GO terms of the N4-Eru-treated Jurkat cells. The length of the bar represents the significance of that specific gene set or term. In addition, the degree of the brightness of the color denotes the significance (P < .01) of the differentially expressed genes.
KEGG/Biocarta pathway analysis
The KEGG/Biocarta pathway analysis showed the possibility of the involvement of NOTCH, JAK-STAT, and Wnt signaling pathways (Figures 3 to 5). There was a significant decrease in the levels of STAT5 which overexpression is known to be involved in cell survival. The important epigenetic modulators such as HDACs 1 and 3, DNA methyltransferase (DNMT1), and lysine demethylases (KDM3A, 4B, 4C), which regulate gene expression, by modifying histone posttranslational marks, were significantly downregulated (LogFC < 1.3; P < .01; Table 3).

KEGG pathway analysis of differentially regulated genes in Jurkat cells after treatment with N4-Eru, depicting the affected genes in Notch signaling pathway.

Significant deregulated key signaling nodes of JAK-STAT signaling pathway are represented in Jurkat cells after treatment with N4-Eru. Biocarta pathway analysis of critical differentially expressed genes is depicted with an asterisk.

Significant deregulated key signaling nodes of JAK-Wnt signaling axis are represented in Jurkat cells after treatment with N4-Eru. KEGG pathway analysis of differentially expressed genes is depicted with an asterisk.
Downregulated genes in N4-Eru-treated Jurkat cells as compared with untreated cells.
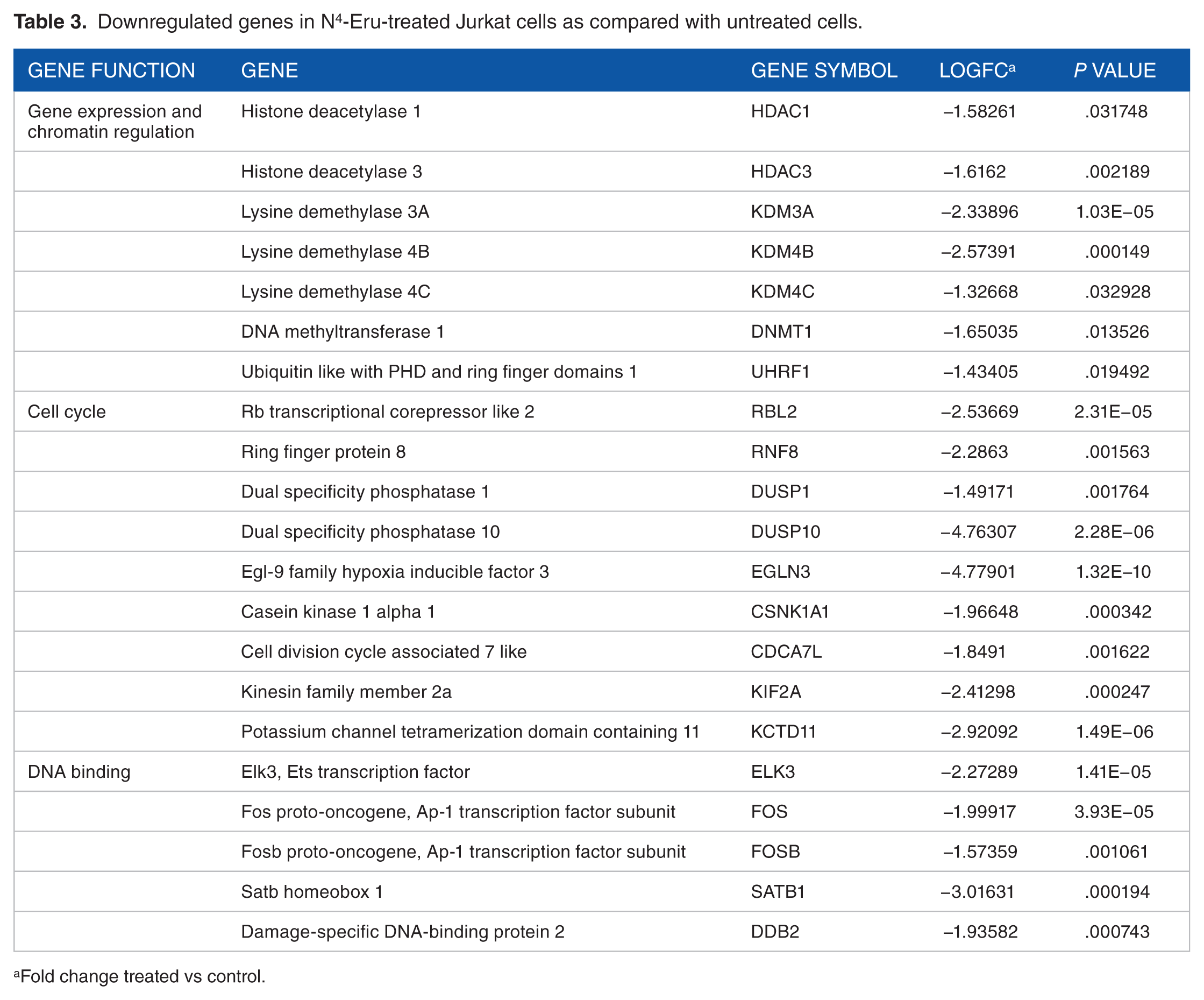
Fold change treated vs control.
Moreover, NOTCH1, NOTCH2, and NOTCH3 were downregulated with the subsequent downregulation of notch-related genes inhibiting cell growth and survival (Table 4). Furthermore, several genes such as RBL2, RNF8, DUSP1, DUSP10, EGLN3, CSNK1A1, CDCA7L, KIF2A, and KCTD11 involved in cell cycling were significantly downregulated. In addition, FOS and FOSB transcription factors which are proto-oncogenes were downregulated indicating a possible event of inhibition of cell growth in response to N4-Eru treatment. The crucial gene NDRG1 is an important component of the Wnt/Hedgehog/Notch super pathway involved in apoptosis and regulation of TP53 activity. Our results showed that NDRG1 was downregulated in N4-Eru-treated cells suggesting that NDRG1 acts as an oncogene in acute leukemia. The tumor suppressor genes KISS1, FOXP1, PPARG, E2F1, TGFB2, and DDIT3 were upregulated (Table 5). The gene interactions of the different transcriptional regulators and tumor suppressors are shown in Figure 6. Figure 6 represents a brief overview of the different gene interactions indicated by different line colors. The coexpression of the genes was highest (50.73%), the physical interactions between the genes were found to be 24.25%, the predicted interactions were 11.20%, followed by genes involved in the pathway at 6.01% and shared protein domains at 4.17%, and co-localization of the genes was found to be 2.42%. The genetic interactions stood at mere 1.2%. The colors in the circle of the gene point resemble a pie chart distribution which helps to identify the role of that particular gene in the different biochemical reactions such as histone modification, chromatin binding, cell growth, and response to hypoxia in the JK cells treated with N4-Eru.
Downregulated transcriptional and nuclear oncogenic cofactors in N4-Eru-treated Jurkat cells as compared with untreated cells.

Fold change treated vs control.
Upregulated tumor suppressor genes in N4-Eru-treated Jurkat cells as compared with untreated cells.
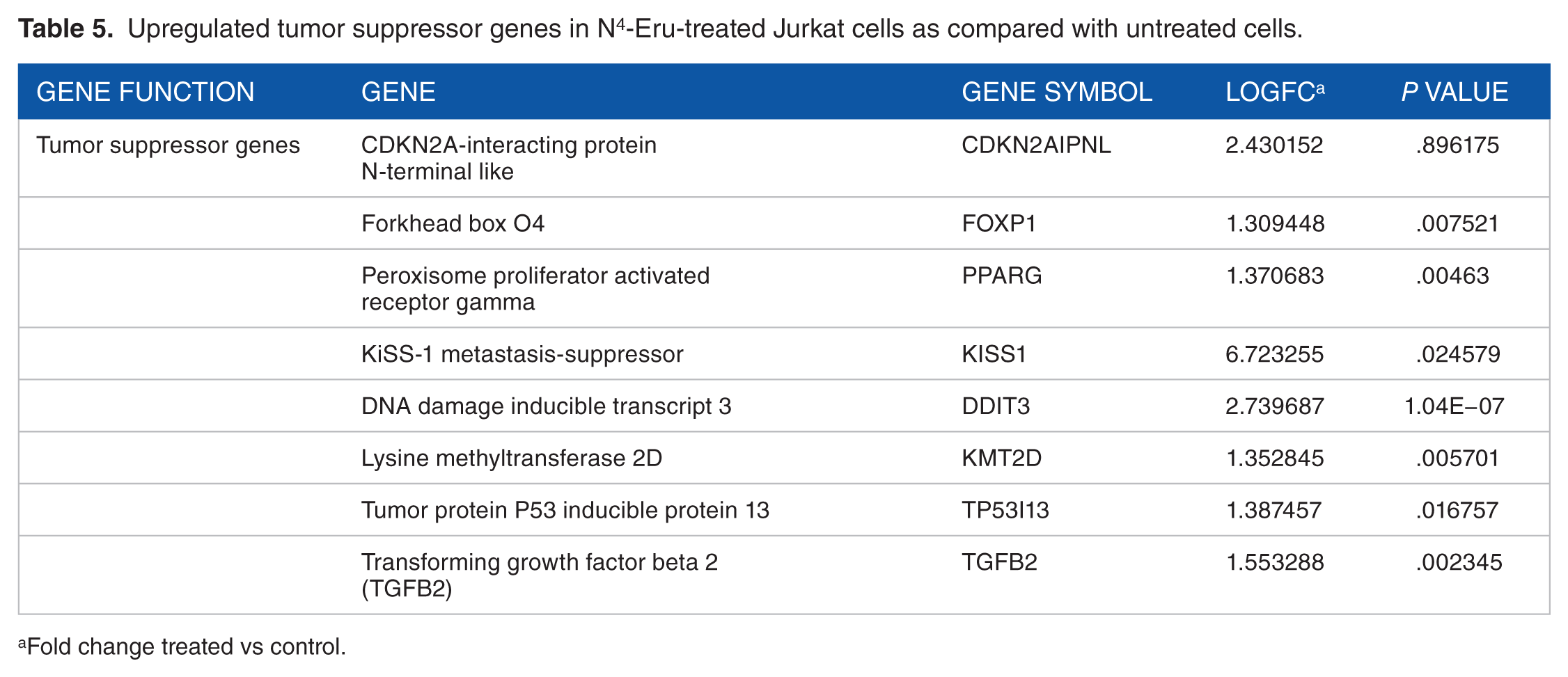
Fold change treated vs control.
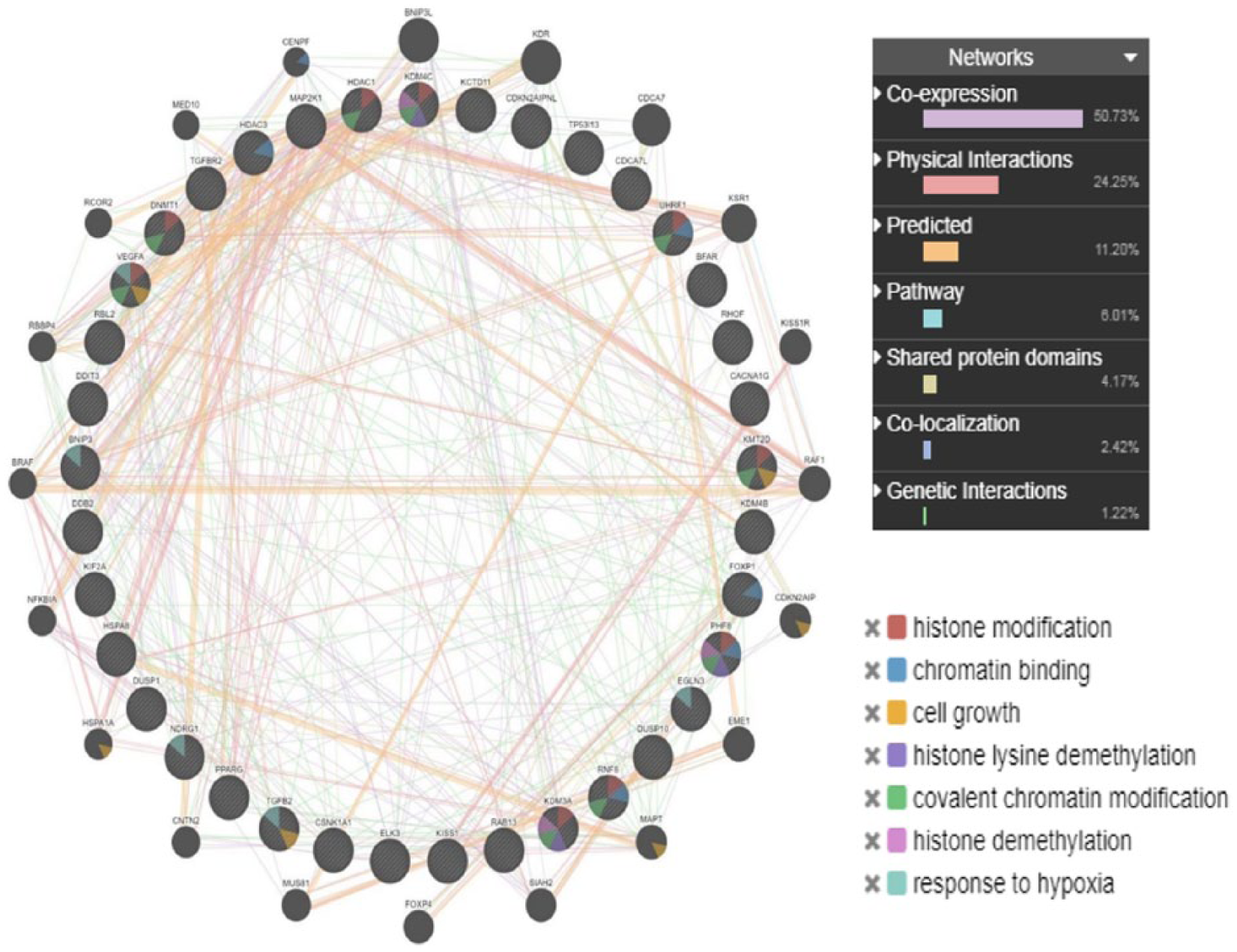
Different gene interactions of the most significantly deregulated differentially expressed genes are represented as strings depicting cell death and tumor suppressor genes in N4-Eru-treated Jurkat cells as compared with untreated cells.
The heat map presented in Figure 7 gives an overall overview of the modulated gene expression with respect to both LogFC and “P” value.

The heat map of the significantly altered differentially expressed genes shows the change with the intensity of the color varying with the alteration of LogFC (fold change) from −6 to +6 in N4-Eru-treated Jurkat cells as compared with untreated cells.
Discussion
Most of the polyamine analogues carry an overall positive charge on the complex and hence increase the affinity towards the negatively charged DNA.39,40 One of the most important roles of the biogenic polyamines is to maintain DNA stability for normal cell function by optimal gene expression. 41 As a matter of fact, polyamine analogues are designed to correct abnormal gene expression occurring in different diseases such as cancer. Inactivation of TSGs is a common characteristic in human cancer cells and is caused by several mechanisms. Epigenetic silencing of TSGs is one of the chief mechanisms involved in the regulation of TSGs and is ensured through a coordinated dialogue between DNA methylation and histone posttranslational modifications such as acetylation and methylation.42–44
In cancer cells, besides promoter hypermethylation, histone proteins are hypoacetylated by HDAC and either hypomethylated or hypermethylated at specific amino acids by histone demethylase (HDM) and histone methyltransferase enzymes, respectively, leading to reduced expression of TSGs. 45 Therefore, many new polyamine analogues have been developed recently which act as HDAC/demethylase inhibitors leading to the reexpression of TSGs.46,47 The data obtained from RNA-sequencing in this study showed that treating JK cells with the polyamine analogue N4-Eru induced a significant increase in a panel of TSGs such as p16INK4a, FOXP1, PPARG, and KISS1 (Table 5) and a significant decrease in the expression of DNMT1 and UHRF1, HDACs HDAC1 and HDAC3 and lysine demethylases 3A, 4B, and 4C (Table 3). These results are consistent with a regulatory positive role of UHRF1 on TSGs, p16INK4A, PPARG, and KISS1.29,48–50 This suggests that N4-Eru-induced upregulation of TSGs could be challenged by epigenetic mechanisms involving DNA hypomethylation or histone acetylation and methylation through downregulation of DNMTs, HDACs, and HDM enzymes, respectively.
The polyamine analogues play a potential role like demethylase inhibitors in the expression of the aberrantly silenced genes by chromatin remodeling. Interestingly, lysine demethylases such as KDM3A, KDM4B, and KDM4C were significantly downregulated in response to N4-Eru which is in agreement with previous studies on polyaminoguanidines and polyaminobiguanides as noncompetitive inhibitors of LSD1. 25 These findings are also in line with previous studies on polyamine analogues (trichostatin (TSA), SAHA (suberoylanilide hydroxamic acid), MS-275 {N-(2-aminophenyl)-4-[N-(pyridine-3-methoxycarbonyl)-aminomethyl] benzamide} as HDAC inhibitors. There is a considerable evidence that HDAC inhibitors TSA, SAHA, and MS-275 are effective in chemotherapeutic regimens in many cancers; however, dose-limiting toxicity remains challenging.23,24,51,52
The data obtained also showed that many oncogenes and genes responsible for cell proliferation and survival such as c-MYC were downregulated in JK cells in response to N4-Eru treatment. In addition, many transcription factors associated with the NOTCH/Wnt/JAK-STAT axis were also significantly downregulated. For instance, signal transducer and activator of transcription 5 (STAT5) activity is induced by a plethora of cytokines and growth factors, resulting in transcriptional activation of specific target genes. STAT5 plays an important role in a variety of cellular processes, including proliferation, differentiation, and apoptosis. Aberrant regulation of STAT5 has been observed in solid tumors as well as in patients with either chronic or acute myeloid leukemia. 53 This study showed that the levels of STAT5 mRNA decreased to nearly 6-fold when compared with the control (Table 4) suggesting an important role of STAT5 in N4-Eru-induced apoptosis.
The Wnt signaling pathway has been reported to be hyperactive in leukemia by the constituent overexpression of an important regulator, β-catenin, a free monomeric protein which is well known for its transcriptional regulation in solid as well as hematopoietic tumors. Many previous studies have identified β-catenin as a substrate for apoptosis induced by proteolysis.54–57 Interestingly, while normal resting hematopoietic cells do not express β-catenin protein, 55 leukemic cells abundantly express β-catenin. 58 This study showed that N4-Eru might cause the repression of Wnt signaling axis and induce apoptosis. In addition, several genes of Notch signaling pathway were also downregulated suggesting a repression of Notch pathway in N4-Eru-treated JK cells. Of note, hypoxia-related key genes such as EGLN3 and HMOX1 were also downregulated which could also contribute in inducing apoptosis.
Conclusions
In conclusion, the polyamine analogue-N4-Eru can target several key pathways in leukemia cells. Therefore, to get a deeper insight into the molecular mechanisms, the advanced fields such as metabolomics, microarray array, and proteomics could reveal the specific targets with the integration of efforts of biologists and chemists to reduce the off-target effects and overcome the problem of dose-limiting toxicities.
Footnotes
Acknowledgements
The authors acknowledge the technical support from Deanship of Scientific Research, King Abdulaziz University, Jeddah, and KFMRC for providing RNA-Seq facility. The data and material presented in this manuscript have neither been published before nor has been submitted for publication to another scientific journal or is being considered for publication elsewhere. All the co-authors have read this manuscript and approved it for submission.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded and supported by Deanship of scientific research, King Abdulaziz University, Jeddah, under grant no. 1-130-36-HiCi.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership, grants, or patents received or pending and royalties. No writing assistance was used in the production of this manuscript.
Author Contributions
TA, CB, MA, and KA designed the project. SR, HC, AH, CB, and MA performed research, analyzed data and wrote the paper. SM, MAH, MZ, and TK helped with experimental design, data interpretation, and drafting of the paper. MH and MAH generated and contributed the data. DH also performed quality control analysis.
