Abstract
Community pharmacists worldwide operate with the continual risk of errors (Quality Related Events—QREs) occurring in their dispensing processes. Contemporary analysis of community pharmacy QREs tends to concentrate on the outcomes of the error, such as the degree of patient harm, rather than on the risks associated with the triggering of a QRE. Drawing on risk identification and mapping techniques from the information security sector, we conducted a risk mapping exercise of QREs occurring in Canadian community pharmacies as identified in publicly available accident investigations. The findings from the present study identified relationships and patterns between various risk factors, types of errors, and patient outcomes. For example, the risk factors most associated with errors that result in patient fatality were the “sound-alike/look-alike” medication labeling and the dispensing checking and verification processes in the pharmacy. Study findings support the application of risk identification and mapping techniques to community pharmacy risk and QRE mitigation practices and regulations.
Introduction
Medication dispensing errors cause significant harm to create far-reaching consequences, as seen in a recent Canadian case of a patient who died after she was given the wrong medication by her community pharmacist. 1 Community pharmacies are among several settings having an increased risk of medication-related incidents. A meta-analysis revealed that the community pharmacy dispensing error rate was surprisingly high, ranging between 1.4% and 1.8% of all prescriptions. 2 These incidents, otherwise known as quality-related events (QREs), are defined as “known, alleged, or suspected medication errors that reach the patient as well as those that are intercepted prior to dispensing.” 3 The latter is referred to as a near miss. 3 Due to the multitude of steps involved in prescribing and dispensing medications, there are numerous instances in which QRE can occur. QREs that reach the patient can increase healthcare costs 4 and most importantly, as noted above, cause negative health implications for patients.
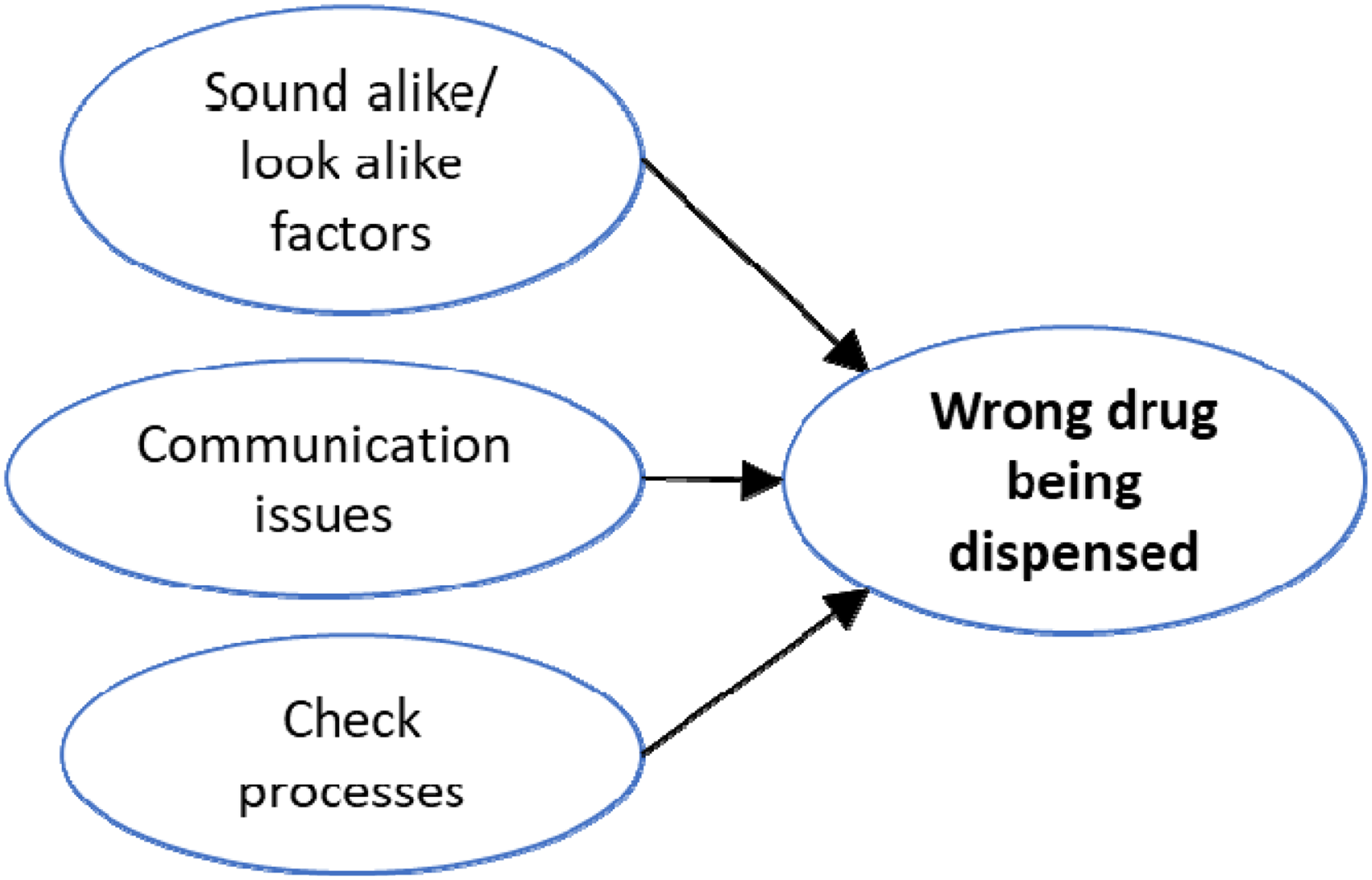
Contributing risk factors—wrong drug being dispensed.
Preventing medication incidents remains a top priority for pharmacy regulatory authorities as they implement systems to heighten awareness and mitigate the causes of medication incidents. In Canada, provincial pharmacy practice regulators in Nova Scotia initially, followed by New Brunswick, Saskatchewan, Ontario, and Manitoba, incorporated mandatory error reporting into continuous quality improvement processes to promote awareness of the types of QREs that are prevalent in community pharmacy practice.3,5,6 Pharmacy practitioners who report QREs contribute to a warehousing database, which allows organizations such as the Institute of Safe Medication Practices (ISMP) Canada to synthesize data, identify root causes, and disseminate the lessons learned to healthcare practitioners. For example, ISMP's newsletters regularly feature one major QRE or several smaller cases that explain the pertinent details including the figures involved, the risk factors present in the case, the steps leading to the incident, and recommendations for practices pharmacies can implement to mitigate similar QREs from occurring. However, the patterns and relationships between the risk factors, types of medication incidents, and the outcome of the incidents have yet to be investigated on a larger scale.
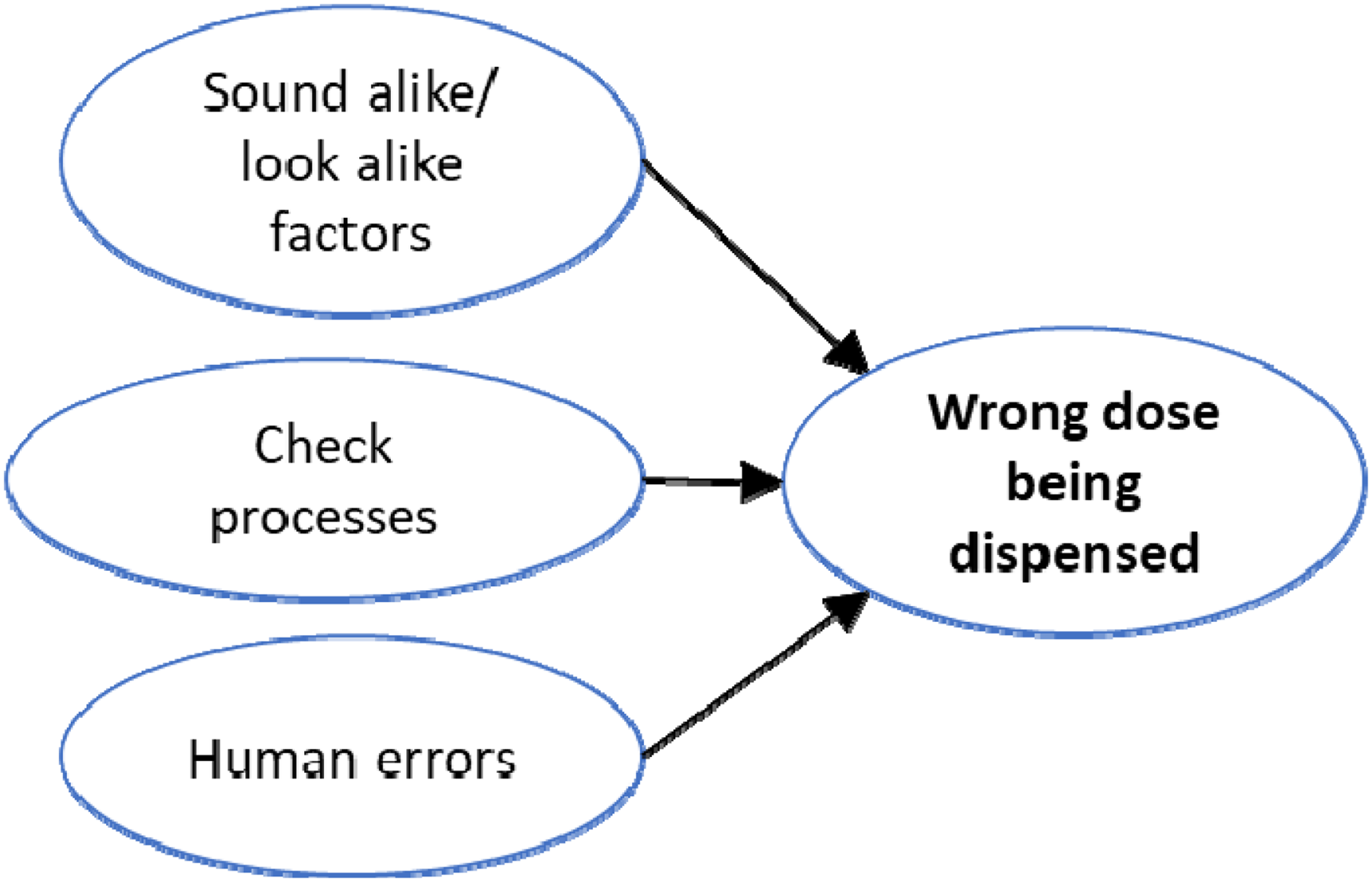
Contributing risk factors—wrong dose being dispensed.
Reporting QREs is a first step toward safer practices. Identifying patterns leading to errors and sharing lessons learned can enhance our improvement efforts, but this is contingent on a structure to analyze events meaningfully. To provide such a structure, the authors applied a risk analysis framework to reported QRE cases to identify and investigate patterns found across these cases. The risk analysis framework provides a clear distinction between “negative events” (the errors), the contributing factors (increasing or decreasing the likelihood of negative events), and the outcomes associated with events. The authors argue that this visualization can foster learning, prevention, and ultimately enable safer practice.
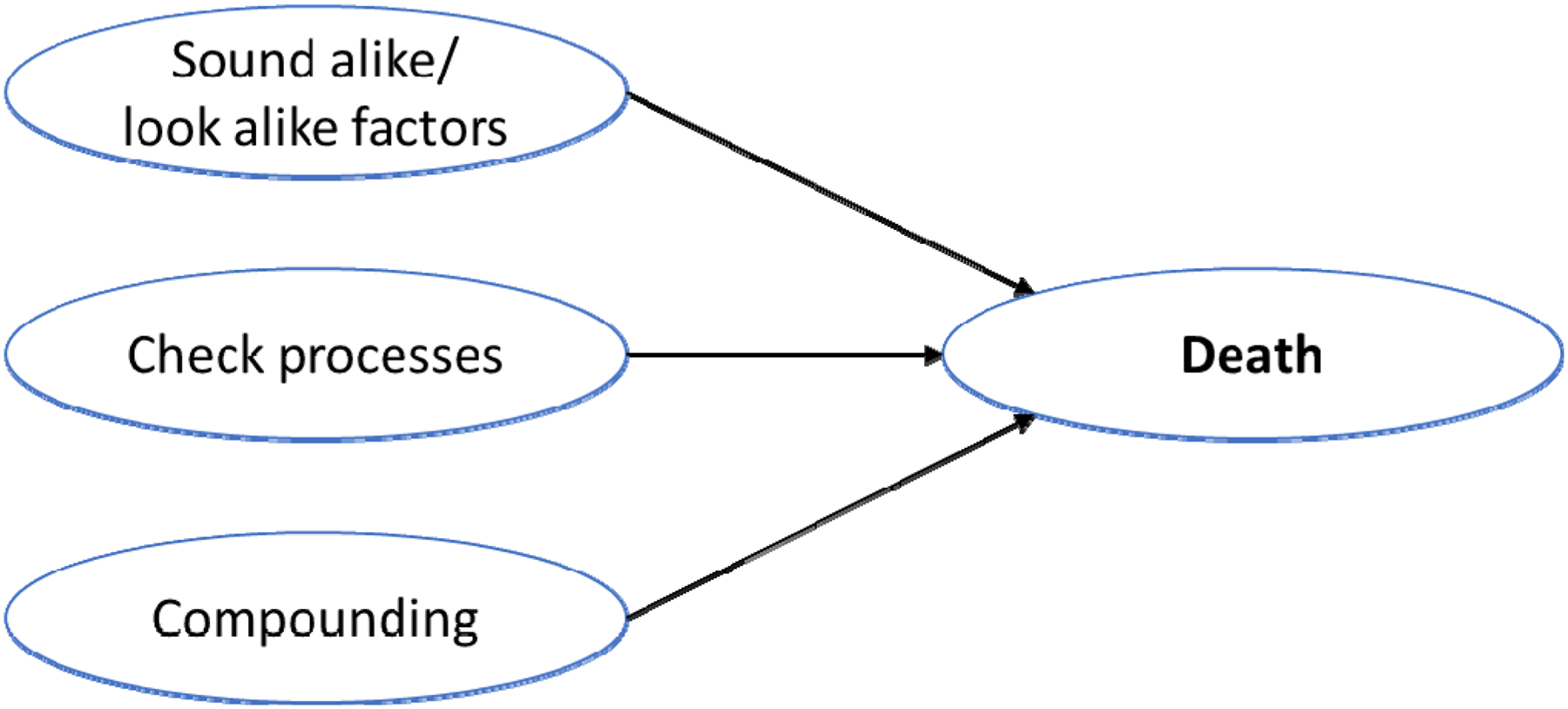
Contributing risk factors—death.
Literature review
Published evidence on the medication errors occurring in North American community pharmacies was relatively rare prior to 2005. The lack of information on QREs in community pharmacies is a striking contrast with the plethora of data on QREs coming from hospital settings, a result of the mandates set by national hospital accreditation bodies. 7 Community pharmacies in Canada are governed by provincial pharmacy regulatory authorities 8 and prior to around 2010, mandatory error reporting was not a requirement. However, findings of medication incident analysis in U.K. community pharmacies 9 emphasized the importance of medication safety in community pharmacies and sparked a cascade of research into medication errors in community settings. In the United Kingdom, the National Patient Safety Agency established the National Reporting and Learning System to allow healthcare practitioners to report adverse events and medication errors and to learn from these errors. 10 In Canada, ISMP Canada introduced the Community Pharmacy Incident Reporting (CPhIR) system in 2010. The CPhIR system was developed in collaboration with a group of university researchers and provincial pharmacy regulatory authorities pursuing the goal of establishing a QRE reporting system for Canadian community pharmacies. 8 The introduction of mandatory reporting into community pharmacy practice created an opportunity for practitioners to report medication incidents, as well as near misses, for analysis and subsequent patient safety practice improvements.
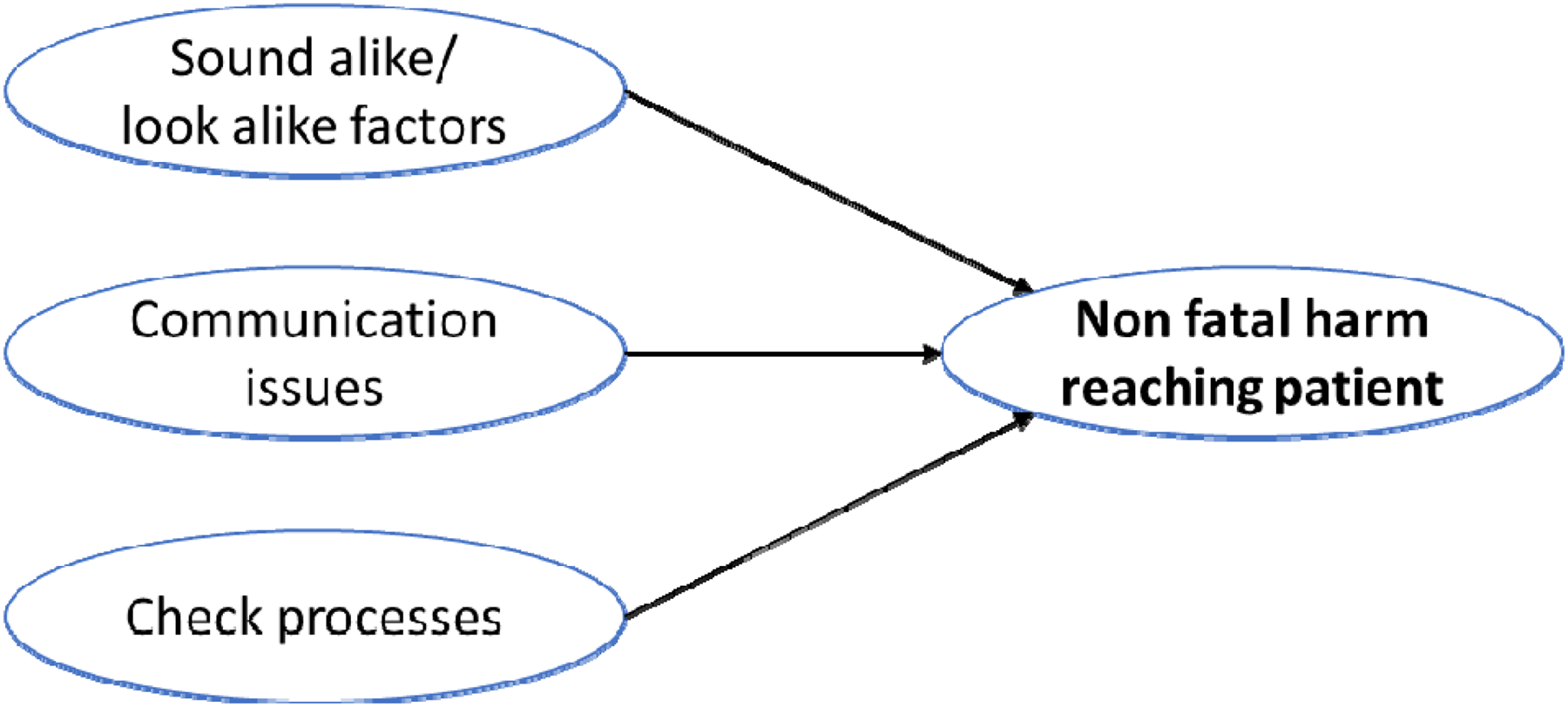
Contributing risk factors—nonfatal harm reaching patient.
Existing research on medication incidents in community pharmacies is often focused on the quantitative aspects of error occurrences, including the frequency of the different types of errors. A study conducted by Boucher et al. in 2018 analyzed the QREs reported by community pharmacies in Nova Scotia 11 and identified the proportion of QREs that inflicted harm to patients and the stage at which the QRE occurred. Despite most of the QREs being caught as a near-miss, this paper brought light to the frequency, types, and patient outcomes of QREs that exist in Nova Scotia community pharmacies. Building from this research, the categorization developed for the types of QREs 11 was adopted in the present research to further analyze the risk factors and their associated patterns in leading to QREs.
Contributing risk factors—no harm but reached patient.
Risk analysis framework
Risk is defined in many different ways depending on the field, research, application, and types of threats in each field. In the community pharmacy context, risk is associated with activities conducted by participants (which include the pharmacist, the pharmacy technicians and staff, the prescriber, and the patient). Risk depends on choices made by participants and system-related factors. This condition of participant choice requires a managerial definition of risk that concentrates on the types of error that should be avoided when conducting medication dispensing activities. 12 In the case of a community pharmacy, the goals of dispensing are to “ensure that an effective form of the correct medicine is delivered to the right patient, in the correct dosage and quantity, with clear instructions, and in a package that maintains the potency of the medicine.” (Garfield, 2013, p. 1152) 13 Therefore, key types of error that must be avoided would include dispensing the wrong drug, in the wrong form, in the wrong quantity or dosage, giving erroneous/unclear instructions, or failing to use appropriate packaging.
To develop an understanding of the risk associated with the dispensing activities of a community pharmacy, we need to assess, for each of the types of error noted in the previous paragraph, the potential outcomes and the likelihood of such outcomes materializing. The resulting risk of the dispensing activities can be defined as the combination of the likelihood of each undesirable outcome multiplied by the severity of the outcome.
Severity of outcomes is typically labeled (in increasing order of severity): “near-miss,” when an error was caught before reaching the patient; “no harm,” when the error reached the patient but without impacting negatively the patient health; “harm,” when the patient's health was negatively impacted by the error; and finally “death,” when the error led to a patient fatality. 14
When assessing likelihood, it is difficult to determine a pure probability as errors in a pharmacy setting are influenced by a number of factors. A useful approach in such situations is to determine relevant risk factors to serve as a proxy for likelihood. Risk factors are factual elements that have been associated in the past with the occurrence of an outcome. 15 Such analysis commonly draws on the results of selected past events and expert judgement. 16
Methods
Data
The data used comes from documented incidents. Each month, ISMP releases a safety bulletin that highlights the month's medication safety incidents. The authors analyzed safety bulletin newsletters from May 2009 to May 2019 and focused on newsletters that highlighted QREs from the community pharmacy setting.
Each medication incident was analyzed and broken down by several elements:
Outcome: death, nonfatal harm, no harm, and near miss Type of Error: wrong dose, wrong drug, wrong instructions given to the patient, and other Risk factors: which were determined by reviewing the information in each case.
Errors and their outcomes.

Risk factors.
This analysis enabled the application of the risk analysis framework to identify the risk factors leading to the negative outcome for each incident.
Coding procedure
In each newsletter, ISMP identifies the factors that are present in each medication incident (even if they are not always formally labeled as “risk factors,” they are described in the text). After assigning the type of error, outcome, and risk factors, the data were pooled into Tables 1 and 2. This coding process was done for all 34 community pharmacy-based incidents that were published in the ISMP Canada Safety Bulletin from May 2009 to May 2019.
For example, a May 2017 ISMP safety bulletin featured an unfortunate medication incident which led to the death of a patient from dispensing the incorrect medication.
17
In this case, in a prescription calling for L-tryptophan for the 18-month-old patient, the pharmacy compounded and dispensed baclofen, which was inadvertently administered, leading to a dose that was 20 times higher than the maximum recommended pediatric dose, ultimately leading to death. Using the information provided in the ISMP bulletin, we identified several risk factors: Both baclofen and L-tryptophan
All incidents were coded the same way. We next compiled incidents into consistent groups: all incidents together, all incidents involving wrong dose, or wrong drug, or involving death or serious harm, etc. Grouping incidents enabled us to assess if patterns were emerging and to assess if risk factors are more prevalent in some situations than others. This provides learning that can improve safety.
Validation procedure
We convened a panel of five pharmacists currently working in different Canadian regions who were asked to assess the findings and offer adjustments drawn from their collective experiences. One of the pharmacists was a current pharmacy manager and two of the panel members also held extensive experience in pharmacy risk mitigation and improvement management. Following expert panel review procedures, 18 the panel members reviewed tables presenting the types of error, risk factors, and outcomes identified in the initial analysis and the apparent linkages between these elements. The panel members were asked to identify any element or linkage they believed was missing or misinterpreted, based on their experience. The panel's input was consolidated and reviewed by the research team to produce a final output of risk factors, types of error, and outcomes.
Results
All 34 cases were analyzed to uncover patterns behind the errors reported.17,19–46 There were an almost equal number of wrong dosage and wrong drugs given. Only two cases reported wrong instructions. Other errors were a misinterpretation of an abbreviation that was caught before leading to problem and a mislabeling of a box. In the cases reported, six led to the death of a patient, 16 caused harm, eight reached the patient but did not cause harm, and four were intercepted before reaching the patient. Table 1 presents the types of errors and outcomes observed.
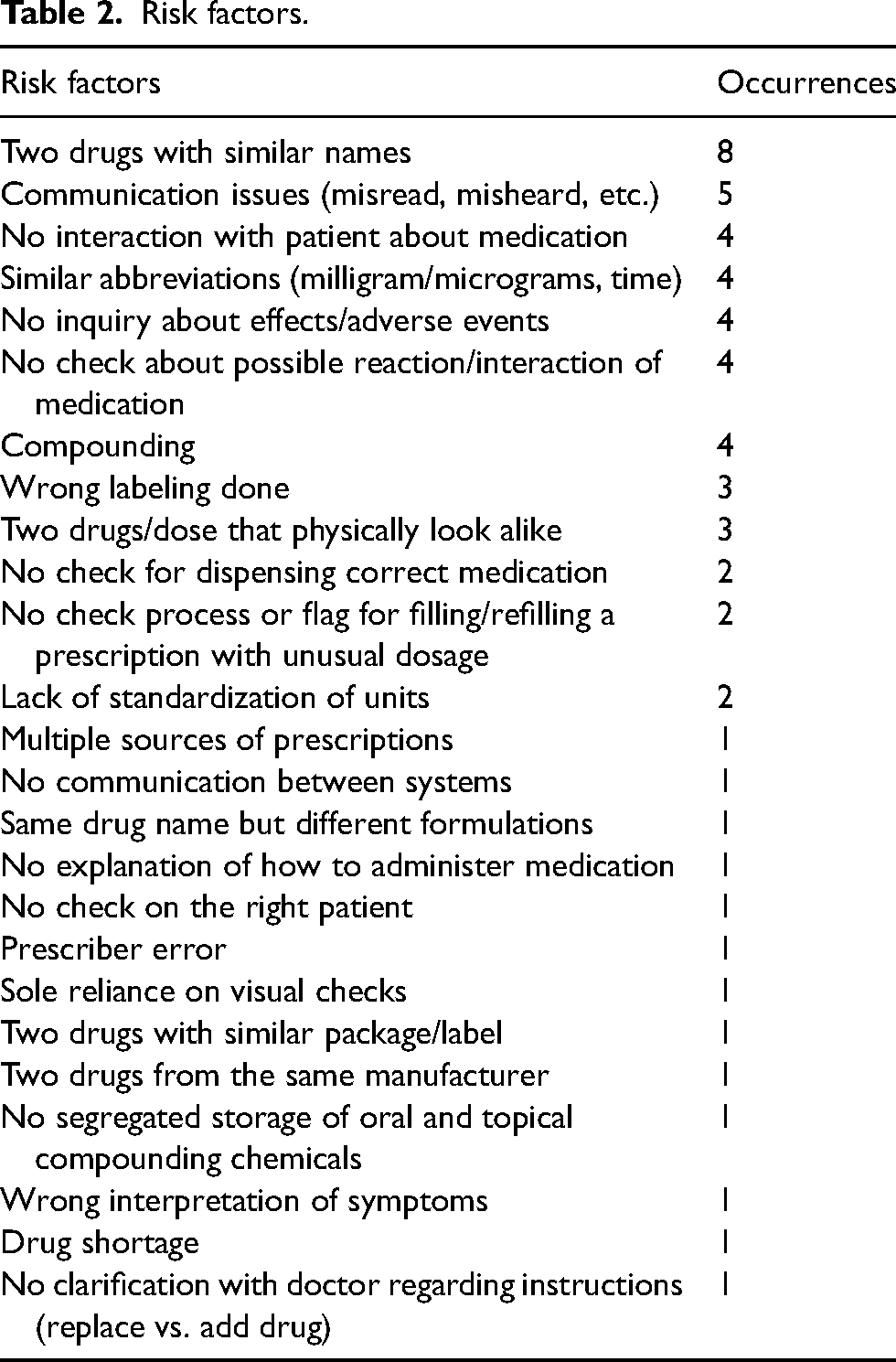
There were numerous risk factors observed in the cases, as shown in Table 2. The most common risk factors involved different medications that sounded alike and communication issues. In total, 25 risk factors were identified.
Notably, in 22 of the 34 cases, more than one risk factor was present at the same time. In 20 cases there were two risk factors identified and two cases had three or more risk factors identified at the same time.
Links between type of error and outcome
Among the six cases leading to the death of the patient, three were cases of wrong dosage, and three were cases of wrong drugs being dispensed. Similarly, when considering the cases in which there was nonfatal harm, there were seven cases of wrong doses and seven cases of wrong drugs being given. Two involved providing wrong instructions. This suggests that the two most common types of errors (wrong drug and wrong dose) can lead to the same negative outcomes.
Links between risk factors and errors
Patterns start to emerge when observing the linkages between the risk factors and the types of errors. Wrong drugs are often dispensed when names are similar or when communication is impeded. Wrong doses are dispensed mostly when checks of various types have been omitted. Understanding these patterns is key to reducing the likelihood of errors occurring. The following sections look at these patterns in more detail.
(a) Wrong drugs
When identifying risk factors that lead to the wrong drug being dispensed, three factors are prevalent in those cases: the similarities (sound-alike/look-alike factors), the communication issues, and the process issues (Figure 1).
The “sound alike/look alike” risk factors are largely present. Eight cases had two drugs with similar names. Two cases had drugs that physically looked alike, had similar packaging, shared the same name, and came from the same manufacturers but had different formulations. Interestingly, in one case the prescribing physician wrote the prescription for the wrong drug because two drugs sounded the same, and the error was detected and intercepted by the pharmacist.
Five cases involving wrong drugs showed communication issues of some form between the pharmacist and the patient: two cases had no communication about the effects and side effects of the drug, one case had no communication of a brand change, one case had no communication about the purpose of the medication, and one case had simply no interaction with the patient about the medication.
The other group of risk factors contributing to wrong drug errors centers around process issues, specifically involving verification.
47
Three cases showed no checks done about possible reactions or interactions associated with the medication, two cases featured a lack of double check of the physical product packaged when dispensing the prescription by the pharmacist, and one case showed that a patient's second identifier (date of birth, phone number, etc.) was not confirmed before dispensing the medication to the correct patient. Finally, a few other unrelated factors were observed such as issues revolving around the transmission or transcription of the prescription.
(b) Wrong dose
Interestingly, sound-alike/look-alike factors and process issues lead to the wrong dose being dispensed, which is similar as what is observed when the wrong drug is being dispensed (Figure 2).
When examining cases where the wrong dosage was dispensed, it can be observed that wrong dosage seems to be caused in large part by the lack of verifications: lack of double checks between dispensed medication and prescription, erroneous calculations during compounding, lack of checks of possible reaction or interaction, or for refilling prescriptions with unusual dosages. Two cases showed no alert in place for confirming unusual dosages.
The look alike, sound alike cases involved similar abbreviations, numbers that looked alike, or different doses of the same medication that looked similar.
Human errors were observed in four cases. Incorrect transcription of a prescription, wrong interpretation of a patient's symptoms, wrong label attached to a vial, and wrong choice of correct dose all led to the wrong dosage being delivered.
Communication issues were less frequent drivers to wrong dosing than in the cases in which the wrong drugs were dispensed. Only two cases showed communication challenges. Specifically the lack of inquiry about effects or side effects. One case was linked to the presence of multiple sources of prescriptions in various electronic systems with interoperability between each.
Links between risk factors and outcomes
The data also provided information about the types of risk factors that were associated with different outcomes.
(a) Death
Look-alike sound-alike medications, compounding challenges, and failures in the verification process were prominent risk factors in errors that result in patient deaths(Figure 3).
Three of the six cases involving a patient death involved an absence of checks about possible adverse reactions or drug interactions associated with the medication. In one case, absent checks led to the wrong drug being given and, in another case, to the wrong dosage. Three cases involved compounding activities.
Sound-alike/look-alike issues are also one of the risk factors leading to the death of a patient. Examples of sound-alike/look-alike risk factors were: similar abbreviations, similar names of drugs, similar packaging, physical similarity, and same manufacturer.
(b) Nonfatal harm
Risk factors associated with similarity also characterize nonfatal harm events. Sound-alike/look-alike factors are the most numerous, with three cases involving similar abbreviations and three cases involving drugs having similar names. Other types of similarities were different formulations of drugs sharing the same name and two look-alike numbers (Figure 4).
There were numerous communication issues between pharmacists and patients. These included the absence of communication regarding side effects, lack of explanation behind medication administration communication, no clarification regarding instructions, lack of follow-up about the proper use of medication, and lack of communication with the patient about the medication.
Process elements behind dispensing were also observed to be missed in this group. There were two cases for which there was no check to ensure the appropriate medication was dispensed. In four instances double checks were missing during compounding calculations when assessing for possible adverse reactions or interactions, and to ensure the medication dispensed matched the prescription and the patient.
(c) No harm (but reached patient)
Eight errors were made in the sample that reached the patient without causing harm. Three errors were associated with medications sharing similar names and three errors were associated with other forms of similarities, including the similar appearance of medication with different strengths, different doses that looked alike, and different formulations that sound or looked alike (Figure 5).
A few process issues were observed that did not contribute to harm, although these were not as prevalent as they were in the cases in which harm was observed. Examples include mislabeling.
Discussion
The objective of this paper was to apply a risk analysis framework to QREs to uncover qualitative patterns contributing to medication errors in community pharmacies. Current literature focuses on the quantitative aspects of QREs including the incidence of the types of QREs, aspects of the pharmacy workflow that are highly susceptible to errors, and the economic implications of medication-related errors to the healthcare system.11,48 To complement and further advance this area of research, the authors were able to synthesize qualitative data from QREs published by ISMP Canada by applying a risk analysis framework to identify relationships between risk factors, types of errors, and their outcomes.
(a) Identifying patterns between risk factors and outcomes
In the cases reported, six led to the death of a patient, 16 caused harm, eight reached the patient but did not cause harm, and four were found before reaching the patient. Half of the deaths discussed in the bulletins were due to the absence of verification regarding possible adverse reactions, drug interactions, and indications. This is a single factor with dire outcomes. However, in the cases in which the error led to death, the pharmacist was unable to assess the all-around appropriateness of the prescription. Unfortunately, we are unable to assess the potentially additional intrinsic and extrinsic factors that may have contributed to the pharmacist's inability to intercept the medication incident.
11
However, a busy shift, numerous interruptions, or lack of knowledge are all known potential risk factors that increase the likelihood of error.
When discussing the most common risk factor in all cases as presented in Table 2, similarities (sound-alike/look-alike) were present in all situations. Sound-alike and look-alike risk factors pertain both to the name of the medication and to the physical appearance of the medication. For instance, one of the cases that led to a near miss involved a student being instructed to fill up an automated dispensing machine canister with metformin; however, the student accidentally grabbed a bottle of acetaminophen with codeine, which is similar in physical appearance. Another case featured a medication incident in which a patient was dispensed lorazepam, a benzodiazepine commonly used for reducing anxiety, instead of loperamide, a medication used to stop diarrhea, which was the patient's primary complaint. The aforementioned case involves the visual misinterpretation of the prescription, likely due to the similarities between the name of the medication and the dose of the medication (loperamide 2 mg tablets and lorazepam 2 mg tablets). In the latter case, the incident was missed by the pharmacy staff as well as the pharmacist. Sound- and look-alike risk factors have high susceptibility, which affects all members of the pharmacy at all steps in the dispensing process. This has been documented in the literature14,49,50 and remains a challenge for community pharmacists. Solutions range from relatively simple interventions, such as creating storage rules that do not rely on alphabetic order to ensure different drugs with similar names are separated, to very complex interventions, such as bringing together industry members and regulators to reassess naming logic and packaging used. 14 Such an approach requires a level of coordination that goes beyond what pharmacists alone can achieve.
When evaluating the QREs that involved the incorrect drug being dispensed, a number of communication issues between the pharmacist and patient were identified. In a case in which the wrong medication was dispensed, the lack of pharmacist intervention and inquiry was evident. The pharmacy had unintentionally ordered propylene glycol, a compounding agent, instead of polyethylene glycol, which is used as a laxative. With all new prescriptions, the pharmacist bears the responsibility to counsel and provide information to the patient; however, in this situation, the lack of pharmacist intervention in assessing the indication and providing critical information regarding the medication (side effects, efficacy, monitoring, etc.) enabled the error to reach the patient and cause harm (metabolic acidosis). Had the pharmacist been given the chance to assess the patient and provide counseling to the patient regarding their prescription, the pharmacist would have had the opportunity to determine that the polyethylene glycol would be inappropriate for the patient's indication. Unfortunately, we cannot determine whether other extrinsic and intrinsic factors were present at the time of the incident, such as busy workflow in the pharmacy which prevented the pharmacist from counseling, patient refusing counseling, or if lack clinical knowledge was at play, based on the information provided in the safety bulletin from ISMP, Canada. Time for such communication needs to be taken, which can be difficult considering the multiple demands put on community pharmacies. Communication can be even more challenging in pandemic situations, notably when masks are worn, which limits nonverbal cues.
Lack of verification processes was the strongest driver of wrong dose errors. We see several similarities between cases in which the pharmacist did not adhere to proper checking processes. In one of the medication incidents involving the wrong dose being dispensed, a pharmacist relied on a visual check of the quantity of selenium powder weighed; however, even in the presence of the necessary technology and checking processes, the pharmacist did not utilize the available equipment and technology to properly check the units of measurement (micrograms vs. milligrams). This error led to the death of a patient from one 1000-fold overdosage. Along with improper check processes, we found elements of human error when dispensing the incorrect dose, which entails incorrect transcription and interpretation of prescriptions and calculation errors. While there are resources in place to raise awareness to prevent errors during transcription and interpretation of prescriptions, such as tall man lettering 51 and lists of error-prone abbreviations 52 from ISMP, errors still exist.
Despite proper verification processes, human error and system-based errors were still present. Further research will have to investigate in which situations those errors happen (fatigue, noise, etc.). Systematic check processes are key to avoid harming at any level. Embedding verification checks into established routines are useful steps for ensuring their use is constant.
49
(a) Complementarity with other approaches
As mentioned earlier, previous approaches looked at quantifying error occurrences
11
or learning from past incidents reported,
6
which led to the development of tools such as the root-cause analysis tool.
53
The quantification is important to assess the frequent types of errors and the most common situations leading to error. Root cause analysis provides insights into the process and the possible process improvements that can be introduced to improve safety.
Risk mapping, as presented in this paper, provides a complementary view to established approaches. It is about understanding patterns. By looking at an ensemble of risk factors and outcomes, we can identify how different risk factors tend to be present together and lead to outcomes. The approach does not seek to assign a fault but rather to identify the various types of situations for which risk will be higher. For example, in our sample, half of the cases leading to the death of the patient involved compounding activities. However, compounding was much less frequent in the other types of incidents. This suggests that there are higher risk levels when compounding is involved. If such situations are recognized, appropriate mitigation measures can be developed. If we analyze cases individually, this element could be missed. The more details are available in a case description, the more refine the risk mapping analysis will be.
Limitations
Our findings apply to a small sample size of QREs that were selectively published by ISMP Canada. The authors of this paper chose to use the safety bulletin newsletters from ISMP, despite the small sample size, because it provides a source of incidents with detailed information, validated by a recognized organization, and for which reporting was done in a consistent way and language from one incident to the other. With increased consistency, we sacrificed the generalizability of the findings due to the small sample size. However, by successfully applying the risk mapping model, we highlight the contribution of the approach.
As noted above, the authors analyzed QRE safety bulletin newsletters from May 2009 to May 2019 that were focused on the community pharmacy setting. This is both an advantage and a limitation. On the one hand, it ensures that there is consistency in the reporting of these incidents, as it is done solely by one organization (ISMP), which is dedicated to the prevention of medication errors. On the other hand, it means that researchers have no control over the selection of incidents to report and that we must rely on ISMP to decide what is worth reporting.
Conclusion
Our goal was to apply a risk analysis framework to QREs that occurred in community pharmacy settings to provide a way for enhancing research on community pharmacy error reporting. The findings from the present study identified relationships and patterns between various risk factors, types of errors, and patient outcomes. Although we analyzed a smaller dataset, our findings provide guidance on applying a risk analysis framework to a larger database.
Present research on the quantitative aspects of community pharmacy error reporting primarily concerns descriptive statistical analyses. 11 Such analyses are useful in terms of identifying key error patterns (e.g., wrong drug dispensed frequencies) and outcomes (e.g., number of specific patient harm levels), but less useful for identifying and assessing the relationships among the risk factors and deriving practice recommendations for risk mitigation. Taking a risk analysis approach offers a useful next step to progress the usefulness of community pharmacy safety data and to form the basis for enhanced risk mitigation policy and practice benefiting both pharmacists and patients.
Future research is needed on a larger sample size of medication incidents to increase the validity and generalizability which will provide regulatory bodies and community pharmacies with the tools and knowledge to identify specific risk factors that have the capability to compromise the safety and health of the patients.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Dalhousie Pharmacy Endowment Fund (grant number 2018).
Ethical approval
Analysis was conducted on publicly available, open-access documents. No data were collected from human subjects.
Guarantor
James R. Barker
j.barker@dal.ca.
