Abstract
Objective:
To summarize evidence-based indications of topical 0.1% tacrolimus in treating ophthalmological diseases.
Methods:
A comprehensive literature review was conducted using PubMed, Embase, and Wanfang databases. Two independent reviewers screened studies according to predefined criteria: original human research (case reports/series, prospective/retrospective studies, RCTs) on ophthalmic applications of 0.1% tacrolimus were included, while reviews, conference abstracts, non-ophthalmic studies, alternative formulations/routes, and preclinical studies were excluded. Two reviewers independently extracted data using a piloted form, resolving discrepancies through thorough discussion. Extracted variables included study details (author, year, design, location), population characteristics (patients/eyes), and intervention protocols. Data were synthesized thematically by ocular disease category, with comparative analysis of study designs, treatment regimens, and clinical outcomes.
Results:
0.1% topical tacrolimus significantly improves the signs and symptoms of a wide spectrum of inflammatory eye conditions of the anterior segment, namely allergic conjunctivitis, viral conjunctivitis, immune checkpoint inhibitor-related conjunctivitis, ocular cicatricial pemphigoid, nodular episcleritis, graft rejection, graft-versus-host diseases, dry eye disease, autoimmune keratitis, blepharitis, and periorbital dermatitis. In addition to monotherapy, 0.1% tacrolimus can be used as an adjunct alongside other medications such as topical steroids and topical cyclosporine. The medication is generally well-tolerated with minimal side effects, with the most common complaint being local stinging, which dampens over time. Its reassuring safety profile in the ocular application is evidenced by its minimal systemic absorption post-administration.
Conclusion:
0.1% tacrolimus is effective and safe for a wide spectrum of eye diseases involving the anterior segment and adnexa. Not only does 0.1% topical tacrolimus spare patients from the side effects of chronic topical corticosteroid use but it also emerges as an option for patients inadequately managed by other immunosuppressants, highlighting the growing significance of 0.1% tacrolimus in ophthalmic practice.
Keywords
Introduction
First isolated from Streptomyces Tsukabenesis 1987, 1 tacrolimus is a macrolide lactone that is widely used in medicine as an immunosuppressive. It has a long history of safety in medicine for indications such as the prevention of post-organ transplant rejection in oral preparation and the treatment of atopic dermatitis in topical ointment.
Over the past decades, ophthalmologists have gradually incorporated topical tacrolimus in their clinical practice, particularly in the treatment of ocular inflammation, which has been treated traditionally with topical corticosteroids, in the hope of reducing ocular side effects such as steroid-induced glaucoma, cataract, increased risk of infection, perforating corneal ulcer, and delayed wound healing. Despite tacrolimus’ proven efficacy and safety, it remains an off-label drug in the treatment of ocular diseases. 2
This review aims to explore the indications of topical 0.1% tacrolimus in ophthalmology, which has been underreported in previous literature. While other literature reviews have been conducted to evaluate the general use of topical tacrolimus in ophthalmology or a specific eye disorder, none of them specifically focused on the indication of 0.1% concentration of tacrolimus.
Methods
Protocol
Our protocol was drafted using the Preferred Reporting Items for Systematic Reviews and Meta-analysis for Scoping Reviews (PRISMA-ScR). To identify potentially relevant documents, bibliographic databases of PubMed, Embase, and Wanfang were searched up to May 2025, with keywords of “topical,” “tacrolimus,” “0.1%” in the abstract and/or title. The final search strategy for Embase is attached in Supplemental Appendix 1. The final search results were exported into EndNote with duplicates removed.
Eligibility criteria
Original peer-reviewed research was included if they involved human participants using topical 0.1% tacrolimus in treating ophthalmological disorders. The following study designs were eligible for inclusion: case reports and series, prospective and retrospective studies, as well as randomized controlled trials. No language restriction had been applied, and all published studies up to May 2025 were eligible.
Papers were excluded if they met any of the following criteria: (1) review papers, (2) conference abstracts (3) Letter-to-editor (4) not relevant to ophthalmology, (5) focus on topical tacrolimus of preparations other than 0.1%, (6) non-topical administration routes, (7) animal studies or in vivo preclinical studies, (8) in vitro studies.
Selection of sources of evidence
By applying the eligibility criteria, two authors (P.D.L and J.Y.Y.C) carried out data extraction independently. Titles and abstracts were first screened, followed by full-text evaluation. All conflicts arising during the screening process were discussed to reach a consensus. In case of conflicting data, a third independent author (V.W.S.C) reviews the data for any error (Figure 1).

PRISMA flowchart of the manuscript.
The risk of bias of included observational studies was assessed by the Risk Of Bias In Non-randomized Studies – of Interventions (ROBINS-I) tool 3 (Table 1) and Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) 4 (Table 2). Two independent authors (J.Y.Y.C and T.P.H.L) evaluated the risk of bias of each study, and a third independent author (V.W.S.C) was consulted to reach a consensus.
Risk of Bias in Non-Randomized Studies of Interventions (ROBINS-I) tool assessment.

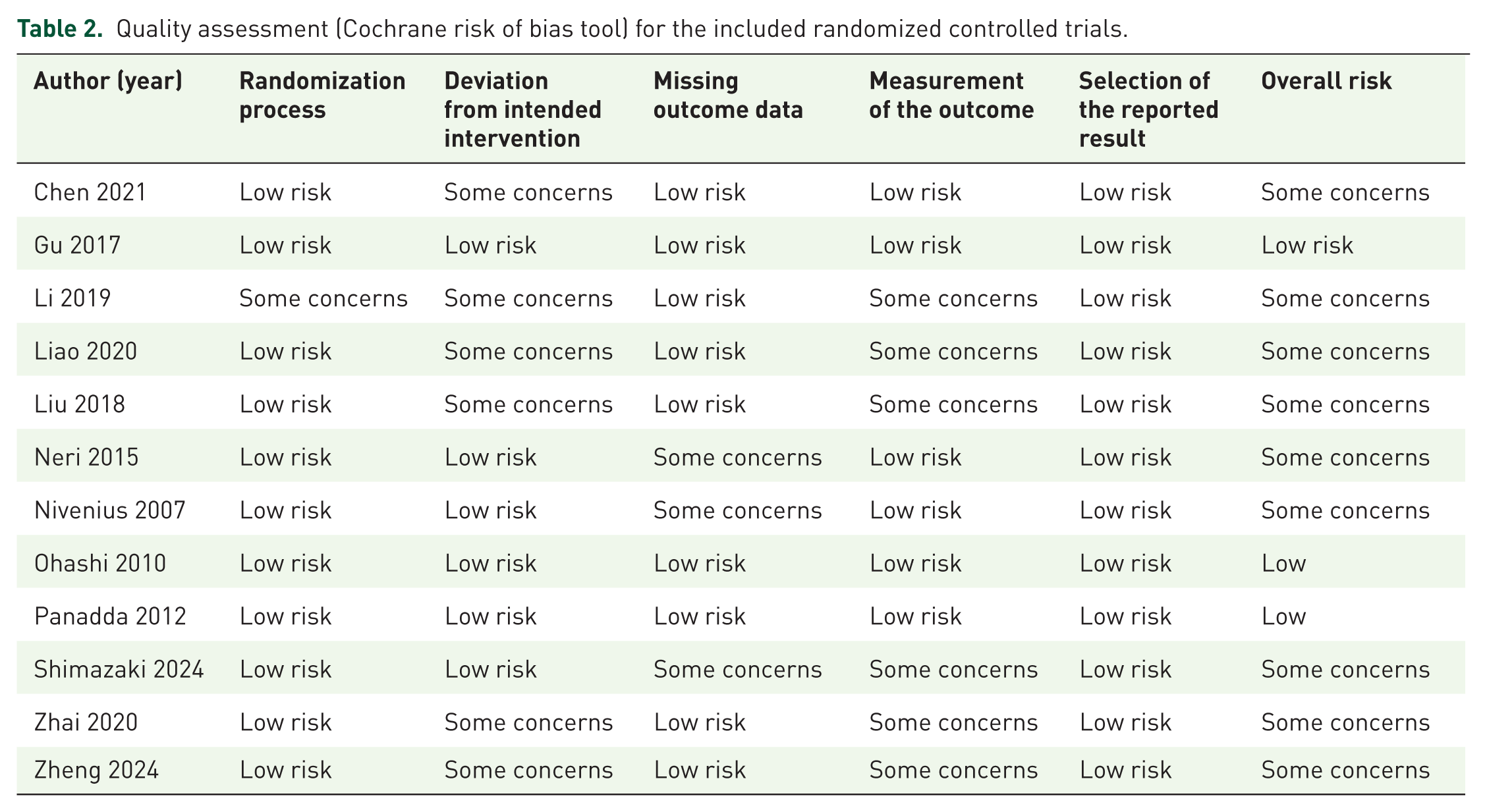
Quality assessment (Cochrane risk of bias tool) for the included randomized controlled trials.
Data extraction and synthesis of results from included studies
Two reviewers collaboratively created a data-charting form to identify the variables for extraction. They independently extracted the data, compared their findings, and refined the form through discussion.
The baseline characteristics extracted include: the first author of the published study, article title, year of publication, study type and design, city and country, number of eyes, number of patients, and patient characteristics.
The results were synthesized by first grouping studies according to the reported ocular disease. Within each disease category, we then analyzed and summarized the study populations, research designs, treatment regimens, and primary clinical outcomes.
Results
We have identified a total of 170 records in our literature search, after screening, and examination for eligibility, a total of 56 articles fulfilled the selection criteria for further analysis. The PRISMA flowchart is illustrated in Figure 1.
Indications of 0.1% tacrolimus in conjunctiva
Allergic conjunctivitis
Allergic conjunctivitis refers to the inflammation of the conjunctiva in response in an allergen. The spectrum of allergic conjunctivitis comprises of milder versions including seasonal allergic conjunctivitis and perennial allergic conjunctivitis, as well as more chronic and severe counterparts, such as vernal keratoconjunctivitis (VKC) and atopic keratoconjunctivitis (AKC).
Traditionally, the treatment modality of allergic conjunctivitis depends on its severity. First-line treatment for mild cases includes lifestyle/ environmental modification, topical antihistamines, and mast cell stabilizers. As severity increases, topical cyclosporine followed by topical corticosteroid can then be added. 5
Vichyanond first described the potentially effective use of 0.1% topical tacrolimus ointment in refractory VKC through an open-label trial. 6 Since then, multiple studies have indicated the clinical value of 0.1% tacrolimus in allergic conjunctivitis. An ex vivo study 7 demonstrated that 0.1% has the least effect on human corneal thickness compared to 0.005%, 0.01%, 0.05%, and 0.2%, while also exhibiting a better corneal viability profile and the highest cell density. The same study also showed that tacrolimus eye suspension is safe and effective for severe VKC, with drug concentration free to be titrated based on patients’ needs. Moreover, a dose-ranging study conducted by Astellas Pharma Inc. comparing the efficacy of 0.01%, 0.03%, and 0.1% tacrolimus on severe allergic conjunctivitis shows 0.1% exhibits superior effect while displaying a similar safety profile as other suspensions of other concentration. 8 Furthermore, Saha et al. conducted a retrospective study to compare 0.03% and 0.1% tacrolimus ointment in children with VKC. Results show that although signs and symptoms were significantly reduced in both groups, the size of giant papillae showed more significant improvement in patients treated with 0.1% tacrolimus, at the expense of experiencing more side effects such as burning and stinging. 9
In the recent decade, 21 studies have been conducted to evaluate the effectiveness and safety of 0.1% topical tacrolimus in treating allergic conjunctivitis (Table 3), with all the results showing that tacrolimus is an effective steroid-sparing agent. Wakamatsu T conducted a prospective study showing that eosinophil cationic protein decreases after 0.1% tacrolimus eye drops treatment, 10 whereas Wan Q recorded in vivo microscopic images of dendritic cells during tacrolimus eye drops treatment, with the results showing dendritic cells have lost their dendritic processes by week 8. However, the negative cells remaining at the palpebral conjunctiva may be prone to reactivation, suggesting that patients should continue a low-concentration tacrolimus treatment despite the improvement of symptoms. 11 The importance of low-dose maintenance therapy is exemplified in Al-Amri A’s prospective study, where all patients who discontinued 0.1% tacrolimus ointment in the 24-month follow-up period had recurrence. 12
0.1% Tacrolimus in the treatment of allergic conjunctivitis.

Itching, discharge, lacrimation, foreign body sensation, photophobia, and eye pain.
The efficacy of topical tacrolimus treatment is affected by the baseline characteristics of individual patients. 13 Patients without or have mild giant papillae, palpebral conjunctival hyperemia, and edema display better response to topical tacrolimus.
The effectiveness of tacrolimus as a conjoint treatment with other topical medications for allergic conjunctivitis was also further examined. Chen M conducted a prospective study to compare the efficacy of 0.05% azelastine and 0.1% tacrolimus eye drops with 0.1% tacrolimus monotherapy among 76 pediatric patients. 14 Results at the end of the 6-week study show that the combination group displays a more rapid improvement than monotherapy. This is accounted for by how azelastine has a more rapid onset of action from blocking histamine receptors when compared to tacrolimus, which typically requires 1–2 weeks to be effective. Improvement of palpebral conjunctiva papillae and conjunctiva hyperemia in the combined therapy is also superior, while corneal involvement in both groups is comparable.
Due to the mechanism of immunomodulation, 1–2 weeks are required for tacrolimus to exhibit its effect. Topical steroids, being fast-acting, can be used for prompt symptom relief. After 1–2 weeks, treatment can then be continued with tacrolimus monotherapy, with the tapering of steroids having minimal impact on treatment efficacy. 13
For the route of administration, tacrolimus is conventionally applied directly on the conjunctiva as an eye drop or ointment. Bardoloi et al. evaluated the possibility of cutaneous application of tacrolimus 0.1% ointment over the upper eyelid skin as an alternative. 15 Tear tacrolimus level was assessed to prove its bioavailability, whereas the clinical disease severity of vernal keratoconjunctivitis was scored to reveal an effective clinical response. This suggests that the cutaneous application of tacrolimus 0.1% ointment is an efficacious alternative.
Viral conjunctivitis
Viral conjunctivitis can lead to persistent subepithelial infiltrates (SEIs), which may cause discomfort and visual impairment. Such complication necessitates treatment beyond topical steroids. The efficacy of topical 0.1% tacrolimus ointment with and without prednisolone for treating SEIs was evaluated in a retrospective observational study involving 102 eyes conducted by Karaca et al. 22 Results showed significant improvements in best-corrected visual acuity, corneal subepithelial infiltrate score, and subjective symptom score, and Fantes corneal haze grading score in both groups at 18 months. Four patients in the combination group experienced an increase in intraocular pressure, suggesting that close monitoring of intraocular pressure is necessary if combination therapy were to be adopted for faster recovery.
To improve the detection of irregular corneal surfaces caused by SEIs following viral conjunctivitis, Toyokawa et al. 23 recommended using Placido ring-based imaging to identify irregular astigmatism after infection. Additionally, their case report demonstrated that topical 0.1% tacrolimus eye drops can effectively treat such corneal irregularities.
Immune checkpoint inhibitor-related conjunctivitis
Kawaguchi et al. reported a case of a 61-year-old female with a history of gastric cancer treated with nivolumab, who complained of conjunctival hyperemia and eyelid swelling post-treatment. 24 After the failure of treatment with 27 weeks of 0.1% fluorometholone eye drops, the patient was prescribed 0.1% tacrolimus eye drops twice daily. The patient showed complete recovery after 3 months and continued to be in remission with a daily administration of 0.1% tacrolimus once daily. This suggests the possibility that tacrolimus may be able to suppress immune checkpoint inhibitor-related conjunctivitis.
Ocular cicatricial pemphigoid
Ocular cicatricial pemphigoid (OCP) is a rare condition that leads to the cicatrization of the conjunctiva. Poorly managed OCP may lead to irreversible sequelae according to Foster’s Classification System such as symblepharon (stage III), keratinization of the ocular surface, and ankyloblepharon (Stage IV).
Michel et al described a case of a 31-year-old woman with OCP who showed a suboptimal response to systemic cyclophosphamide and cortisone eye drops. 25 The patient opted for a trial of topical tacrolimus ointment to treat the persisting symblepharons in both eyes. 0.03% tacrolimus ointment was first administered in the evening, which was then stepped up to two times a day, and finally 0.1% tacrolimus was adopted. Marked improvement in symptoms was shown, and no relapse occurred within 12 months of treatment despite slowing tapering off of treatment.
Indications of 0.1% tacrolimus in sclera
Non-infective non-necrotizing anterior scleritis
Scleritis, commonly linked with systemic rheumatic disease and vasculitis, refers to the inflammation of the sclera. This vision-threatening condition is categorized by the location of inflammation into anterior and posterior scleritis. Specifically, within anterior scleritis, it can be subdivided into necrotizing and non-necrotizing subtypes.
The current first-line treatment for non-necrotizing scleritis includes oral non-steroidal anti-inflammatory drugs and topical corticosteroids. Despite the proven efficacy of such treatment in non-necrotizing scleritis, a significant proportion of patients would need an alternative treatment regimen eventually. 26
A prospective, single-arm study was performed by Yazu et al to evaluate the efficacy of 0.1% tacrolimus eye drops in alleviating symptoms and signs of non-infectious, non-necrotizing anterior scleritis. 27 The patients were first put on a 1-month course of topical steroids, with results showing no significant improvement in symptoms. They were then treated with 0.1% tacrolimus eye drops, which resulted in significant improvement in scores of hyperemia and pain. No infectious adverse events were reported.
Nodular episcleritis
Episcleritis is defined by the inflammation of the episclera, which can be further classified into diffuse, sectoral, or nodular. Diffuse episcleritis is more common and may be self-limiting. Conventional treatment includes topical steroids and oral NSAIDs. Nodular episcleritis has a more severe presentation and higher treatment failure rate than its diffuse counterpart.
Alghamdi A. reported a case of steroid-resistant nodular episcleritis that is successfully treated with 0.1% tacrolimus eye drops four times a day for 6 weeks, after which the medication was gradually tapered off over another course of 6 weeks. 28 After treatment, the episcleritis showed complete resolution, and the patient went into remission.
Indications of 0.1% tacrolimus in cornea
Graft rejection
Corneal disorders can lead to blindness, with corneal graft being the only curative treatment. Despite the cornea being an immunologically privileged site, the leading cause of corneal graft failure is immune-mediated. High-risk graft recipients, which are categorized by factors such as a history of graft failure, corneal neovascularization, history of glaucoma, and infectious cause of corneal opacity, are especially prone to graft failure. 29 Strategies to prevent corneal transplant rejection differ widely, but the most commonly used immunosuppressive drugs are corticosteroids and cyclosporin topically and systemically. These treatment methods are accompanied by side effects such as glaucoma, cataracts, and delayed wound healing. Tacrolimus has also been used in preventing graft rejection in high-risk corneal transplantation. Studies have explored the efficacy and safety of 0.1% tacrolimus in preventing graft rejection.
To assess the ocular safety profile of different concentrations of tacrolimus, Yuan et al. administered topical tacrolimus of up to 0.1% to rabbit eyes. 30 Histological examination shows that there is no ocular abnormality at any of the administered concentrations of tacrolimus, preliminarily suggesting that up to 0.1% topical tacrolimus four times a day is safe for the rabbit eye.
The tolerability of 0.1% tacrolimus suspension is also demonstrated in a single-center, single-blinded, randomized controlled trial conducted by Shimazaki et al. 31 Twenty-five patients were followed up to 52 weeks post-keratoplasty, with 12 eyes receiving 0.1% tacrolimus suspension and 13 eyes receiving artificial tears instead. No serious adverse effects were developed in patients receiving 0.1% tacrolimus suspension.
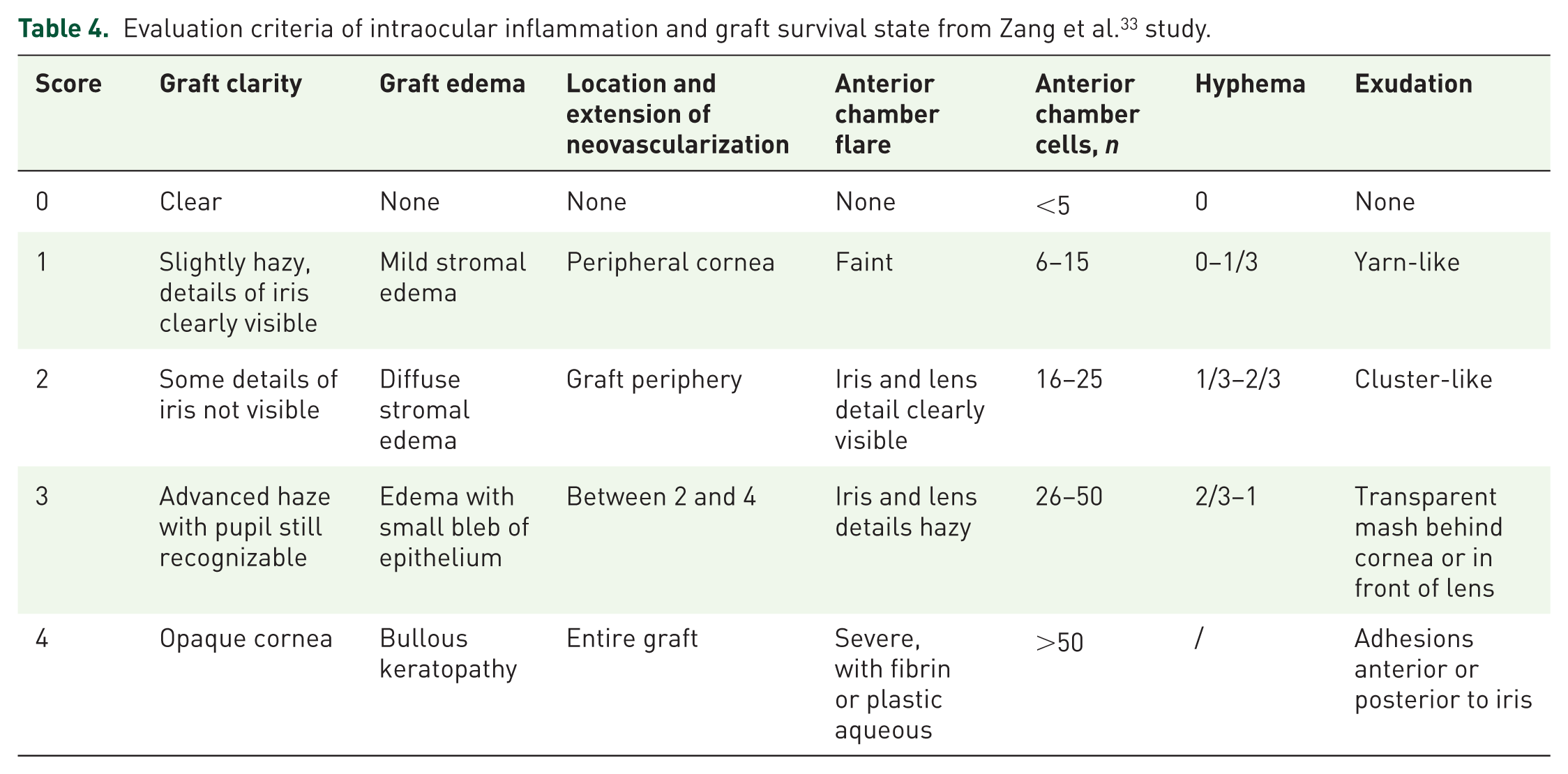
Early postoperative intraocular inflammation is one of the determining factors of the long-term survival of grafts. Zang et al. investigated retrospectively the anti-inflammatory effect of 0.1% tacrolimus eye drops on 20 patients who underwent therapeutic penetrating keratoplasty due to severe keratitis. 32 The patients were treated with antibiotics if there was any evidence of infection. All the patients were also prescribed with 5 mg 0.1% tacrolimus eye drops four times a day for 2 months. Steroid eye drops were administered 1 month after the surgery. Results show that the intraocular inflammation score (Table 4) improved from 7.4 ± 2.06 on the first day to 2.0 ± 2.47 after one month, with the mean post-operative early intraocular inflammation absorption fraction being 5.4 ± 2.13 (p < 0.01). As for long-term follow-up, there are six cases of transplant failure. The degree of inflammatory exudation post-op and long-term immune rejection is found to be positively associated. Hence, this study demonstrates that tacrolimus eye drop facilitates the absorption of early post-operative intraocular inflammation, which potentially improves corneal graft outcome.
Evaluation criteria of intraocular inflammation and graft survival state from Zang et al. 33 study.
Zhai et al., 34 Qi et al., 35 Li et al., 36 and Liao et al. 37 conducted clinical trials, while Zhang et al. 38 conducted a retrospective study to compare the efficacy of 0.1% topical tacrolimus and 1% cyclosporine in preventing immune rejection following keratoplasty. All the studies demonstrated tacrolimus’ superior effect in suppressing graft rejection compared to cyclosporine.
In Zhai et al.’s study, patients were followed up for 6–54 months (averaging 24 months). 34 11 out of 24 (45.8%) high-risk patients administering topical cyclosporine 1% eye drops experienced rejection. As for the 0.1% tacrolimus suspension group, rejection occurs in four eyes, constituting a rejection rate of 16%.
Qi et al. recruited 106 patients undergoing high-risk keratoplasty, with 50 eyes treated with 0.1% tacrolimus eye drops four times a day and 56 with 1% cyclosporine eye drops four times a day. All patients are concomitantly treated with corticosteroid eye drops and are followed up for 5 years. Results show that the rate of rejection episodes in the tacrolimus and cyclosporine group are 14.0% and 37.5% respectively, while the irreversible rejection rate is 5% and 7.1% respectively. A major influential factor for graft survival is medication compliance, which could be accounted for by side effects such as redness, burning, and stinging sensations more often experienced by the cyclosporine group. The relatively lower percentage of rejection rate in this study when compared by Zhai et al’s study may also be explained by the concomitant corticosteroid eye drops, suggesting that a maintenance dose of corticosteroid eye drops is indicated.
Similar findings were seen in Li et al.’s study where 25 eyes were treated with 0.1% tacrolimus and 25 eyes in the control group were treated with topical 1% cyclosporin A. After 1 year post-treatment, the rejection rate was 4.54% and 27.23% in the tacrolimus and control group, respectively. Both the degree of corneal edema and neovascularization in the control group were more severe.
Graft-versus-host disease
Graft-versus-host disease, commonly associated with bone marrow or stem cell transplant, is a systemic order causing inflammation of multiple organs. Severe dry eye is the most common ocular manifestation with the underlying mechanism being lacrimal gland damage as well as meibomian gland dysfunction. Topical glucocorticoid is currently the first-line treatment, but it also comes with long-term side effects. Several studies show topical tacrolimus39–42 and cyclosporine are other possible treatments.
Gu et al. conducted a randomized double-blind study to evaluate the therapeutic effect of 0.1% tacrolimus on dry eye associated with graft-versus-host disease. 39 14 patients were divided into two groups; group A being treated with 0.1% tacrolimus eye drops, artificial tears, and corneal bandages, while group B was treated with only the latter two methods. Ocular surface disease index (OSDI), Schirmer’s test (SIt), corneal fluorescein staining (CFS) score, and fluorescein tear break-up time (BUT) are compared between the two groups. Results show that 0.1% tacrolimus exhibit a more significant improvement in all parameters (Table 5). It is worth noting that only group A shows statistically significant improvement in Slt, suggesting that tacrolimus may be able to alter the tear secretion. No patient experienced significant side effects.
Results from Gu et al. study. 43
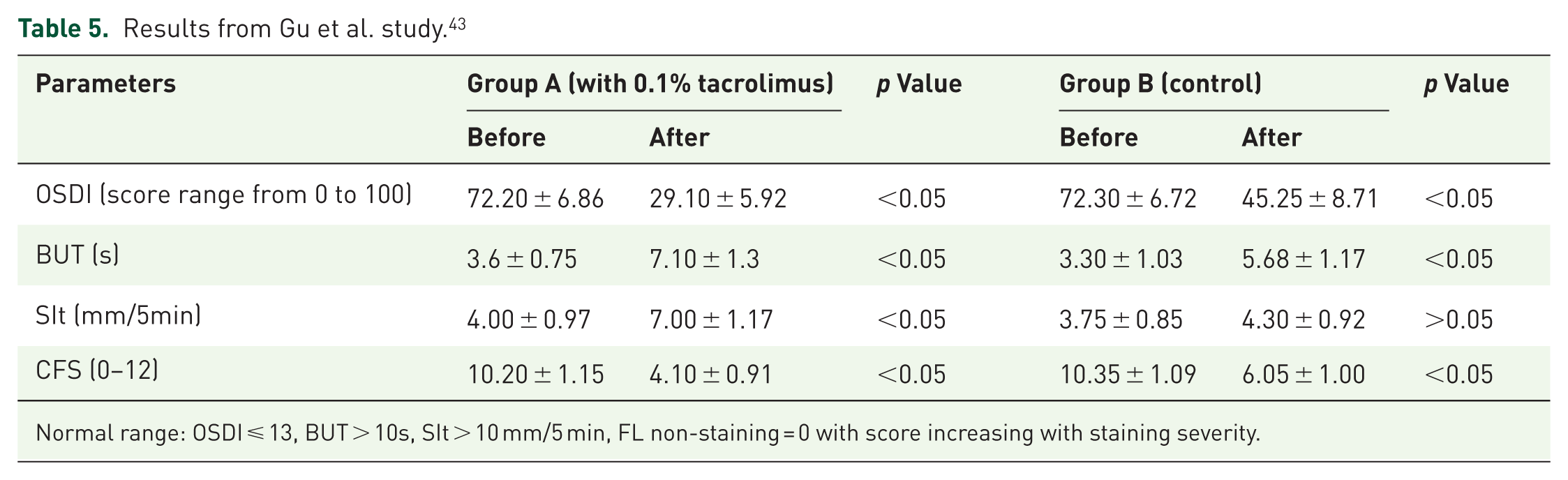
Normal range: OSDI ⩽ 13, BUT > 10s, SIt > 10 mm/5 min, FL non-staining = 0 with score increasing with staining severity.
To compare the effectiveness and safety of 0.1% tacrolimus and 0.05% cyclosporine A eye drops in patients with dry eye manifestation of chronic graft-versus-host disease, Liu et al. conducted a non-randomized concurrent control trial with 84 patients. 40 In the first phase of the study (0–3 months), 44 patients received 0.05 cyclosporine A eye drops four times a day, while 39 patients received 0.1% tacrolimus eye drops twice a day. In the second phase (3–6 months), both groups used 0.05% cyclosporine eye drops and sodium hyaluronate. Parameters assessed in this 6-month study include OSDI, CFS score, and BUT. Results show that despite a slower effect in cyclosporine A, both eye drops are effective in treating dry eye. 0.05% cyclosporine A also provides better symptom and visual acuity improvement in the long term. Irritation was self-resolving in both groups (Table 6).
Treatment regimen of 0.1% topical tacrolimus therapy of various ocular diseases as reflected in literature.

Dry eye disease
Currently, there is no study evaluating the efficacy of 0.1% tacrolimus on dry eye that is not associated with graft-versus-host disease. There are studies, however, that show 0.03% tacrolimus eye drops are effective in alleviating dry eye. 52 Conducting studies using 0.1% tacrolimus eye drops as the intervention can be a direction for future research.
Autoimmune keratitis
The clinical presentation of autoimmune keratitis ranges from mild infiltrates to necrotizing ulceration of the cornea, with the course of the disease being progressive and recurrent. The mainstay of medical therapy is topical glucocorticoid eye drops and cyclosporine eye drops, whereas surgical intervention such as keratoplasty is considered for cases with severe ulceration. However, some cases are refractory to the traditional therapy of glucocorticoid or cyclosporin. A number of studies demonstrated the efficacy of topical 0.1% tacrolimus in treating refractory immune-mediated keratitis.53–56
In a retrospective study to evaluate the efficacy of 0.1% tacrolimus eye drops in refractory cases, Zhang et al. recruited 13 patients diagnosed with refractory immunologic keratitis. 55 Eight of the patients were given 0.1% tacrolimus eye drops four times a day after the second attempt of lamellar keratoplasty, while the other five patients with seasonal keratoconjunctivitis were treated with 0.1% tacrolimus eye drops four times a day. The medication is then slowly tapered down to 1–2 times per day. In the 1-year follow-up period, all patients achieved remission with continuous tacrolimus.
Specifically for recurrent Mooren’s ulcer associated with autoimmune keratitis, Xie et al. conducted a retrospective interventional consecutive case series on nine patients to evaluate the efficacy of topical 0.1% tacrolimus eye drops used alone or combined with keratoplasty. 56 Two eyes with Mooren’s ulcer smaller than half the corneal limbus were treated with 0.1% tacrolimus four times per day, which was then slowly tapered off over a course of 1 year. For the other 13 eyes with larger ulcers, they are treated with both keratoplasty and tacrolimus. The tacrolimus regimen is four times per day for 6 months followed by two times per day for another 6 months. Treatment was successful with patients experiencing a reduction in pain and photophobia within 2 weeks, as well as demonstrating no recurrence.
Indications of 0.1% tacrolimus in eyelid and neighboring skin
Blepharitis
The global prevalence of atopic dermatitis is quoted to be 2.6%, affecting more than 200 million people worldwide. 57 Symptoms include redness, edema, scaling, excoriations, and exudation. In serious cases, chronic inflammation of the eyelid may lead to conjunctivitis and poor corneal surface. Due to the risk of skin atrophy, corticosteroid is not recommended for long-term use. Systemic cyclosporine is used if severe corneal or conjunctival changes are observed; however, topical cyclosporine shows a suboptimal effect in treating blepharitis with associated atopic dermatitis. 58
In a prospective study, 0.1% tacrolimus ointment was prescribed twice a day with gradual tapering over 5 months on 14 patients with severe atopic blepharitis. Results showed signs and symptoms, including itching, erythema, lichenification, exudation, excoriation, and scaling, improved significantly. 59 Around half of the patients experienced burning, which subsided gradually as eyelid inflammation was calmed down. Skin atrophy is unlikely as evidenced by a randomized controlled trial to assess the effect of tacrolimus ointment on collagen synthesis. 60 It is, however, advised that patients should adopt sun protection during treatment.
Apart from blepharitis associated with atopic dermatitis, Zhu et al reported a rare case of severe psoriasiform blepharitis being effectively treated by topical 0.1% tacrolimus ointment. 61 The original dosage is tacrolimus 0.1% ointment two times a day, which effectively relieves eyelid redness and edema. However, eye irritation was reported, resulting in a change of regimen to apply the ointment away from the eyelid margin, which still resulted in satisfactory improvement. The case highlights apart from conventional treatment of low-dose steroid, ultraviolet B phototherapy, and retinoid therapy, 0.1% tacrolimus could be of therapeutic effect.
Periorbital dermatitis
Periorbital atopic dermatitis is a common disease characterized by scaly, erythematous changes around the eyes. Common topical treatments include corticosteroids and topical calcineurin inhibitors such as pimecrolimus and tacrolimus. There is yet to be a large randomized trial evaluating the efficacy and safety of the various agents for periorbital dermatitis.
Freeman et al. first conducted a clinical trial to evaluate the safety and efficacy of 0.1% tacrolimus ointment in treating atopic dermatitis of the eyelids. 62 Twenty-one patients with moderate to severe eyelid dermatitis were given 0.1% tacrolimus ointment twice daily for 8 weeks, and were then followed up for 2 weeks after the last day of treatment. Sixteen patients experienced marked improvement in the physician global assessment score. Apart from local burning and itching, no other adverse events were reported. Similar findings were also observed in Katsarou et al.’s clinical study of 20 adults using 0.1% tacrolimus ointment in the treatment of allergic contact eyelid dermatitis. 63 The non-inferiority of 0.1% tacrolimus ointment to 0.05% clobetasone butyrate ointment was demonstrated in a double-masked explorative crossover study conducted by Nivenius et al. 64 25 patients with both eyelid eczema and atopic keratoconjunctivitis were treated with either ointment twice daily for 3 weeks. Results show that both treatments were equally effective in reducing signs and symptoms of eyelid eczema, suggesting that 0.1% tacrolimus ointment is a promising alternative to steroids in treating eyelid eczema.
Alzahrani et al. conducted a case series presenting the use of tacrolimus ointment in periorbital atopic dermatitis. 65 Two patients were treated with 0.1% tacrolimus ointment on the periorbital region twice per day after refractory response to topical steroid. Within 2 weeks of treatment, both eyes showed a significant reduction in inflammation and became asymptomatic.
A retrospective review of possible treatments for refractory periorbital dermatitis was conducted by Meer et al., where they characterized and compared the wide range of potential therapies. 66 The study suggests that if patients show a lack of response to triamcinolone 0.025% or 0.1%, they can consider treatments such as 0.1% tacrolimus ointment four times per day as evidenced by the positive response by patients in the study. Other treatment options such as tobramycin-dexamethasone, also show positive results.
Discussion
Mechanism of action
Tacrolimus exhibits its anti-inflammatory effect by suppressing the production of cytokines from T cells. Tacrolimus suppresses Th1 cytokines (Interleukin(IL)-2, Interferon (IFN)-gamma), Th2 cytokines (IL-4, IL-5) and IL-3, granulocyte-macrophage colony-stimulating factor (GM-CSF), which are produced by both Th1 and Th2. 67 Such suppressive effect on cytokine production was comparable to or even stronger than certain corticosteroids such as betamethasone valerate and alclometasone. The anti-inflammatory effect of topical ophthalmic treatment was demonstrated in vitro corneal epithelial cells. 68 Tacrolimus is shown to exhibit a similar effect as cyclosporine, another immunosuppressant widely used in ophthalmology, but at a concentration 100 times lower, thus demonstrating its superior potency. 69
Pharmacokinetics and safety profile
After topical ocular instillation of 1% tacrolimus suspension, the concentrations in the conjunctiva and the anterior segment of the eye are much higher than blood concentration in a rabbit model, favoring effective local pharmacologic effect. 70 The difference between blood concentration after instillation of 0.1, 0.3, and 1% was not significant.
In a human study, the blood concentration of tacrolimus in patients administered with 0.1% tacrolimus ophthalmic suspension demonstrated a safe maximum concentration being lower than 2 ng/mL, 18 suggesting that the risk of adverse effects due to serum accumulation is theoretically low.
Side effects
Common side effects of 0.1% topical tacrolimus include local irritation such as stinging and itching. These local side effects generally resolve within weeks of continued use.
More serious side effects include an increase of infection risk. A severe example is a case reported by Miyake-Kashima et al. of a 20-year-old male developing Kaposi varicelliform eruption after 28 days of 0.1% tacrolimus ointment for severe atopic blepharitis. 71 Ocular surface toxicity may also be presented as punctate keratitis or corneal erosions. 19
Despite these side effects, the key advantage of using topical tacrolimus is that it spares the eyes from steroid-related ocular manifestations such as glaucoma and cataracts. 72
Treatment regimen
The duration of therapy of topical 0.1% tacrolimus depends on the specific ocular disease, its severity as well as the patient’s response to treatment. Below is the typical duration of therapy for various ocular diseases as reflected in the literature.
Long-term use
The ocular indications of topical tacrolimus may carry a chronic and relapsing disease course, resulting in the requirement for long-term treatment. Caputo et al., 49 Hirota et al., 51 and Yazu et a. 29 have assessed the clinical outcome of long-term application of 0.1% topical tacrolimus in allergic conjunctivitis. Hirota et al. retrospectively evaluated the efficacy and safety of using 0.1% topical tacrolimus in the treatment of vernal and atopic keratoconjunctivitis for up to 2 years, 51 while Yazu et al. evaluated its long-term outcome of 10 years. 50 In both studies, the adjusted mean clinical symptom score shows significant improvement, whereas the side effect profile of long-term topical tacrolimus use was comparable to that of short-term application.
Possible complication
Yokogawa et al. 73 reported the development of cytomegalovirus corneal endotheliitis in three patients with ocular surface inflammatory disease treated with long-term topical 0.1% tacrolimus and steroid. 74 Despite successful treatment with topical 0.5% ganciclovir, all patients developed irreversible mydriasis post-infection.
In addition, Chatzea et al. 75 reported a case of a 20-year-old patient with prolonged use of topical tacrolimus ointment 0.03% of 5 years due to longstanding atopic keratoconjunctivitis. 75 She was presented with an insidious onset of bilateral blurring of vision, which was later discovered to be tacrolimus-induced limbal stem cell deficiency. The signs and symptoms improved after discontinuation of the offending agent. Hence, the risk of drug-induced limbal stem cell deficiency may be equal or potentially increased with an increase in dosage, it is imperative to continuously monitor patients during tacrolimus therapy.
Limitation
The current review has several limitations that should be considered when interpreting the findings. First, our literature search was limited to the three databases from which other conference proceedings or yet-to-be published trial protocols were not included. Second, there is a wide variability on the research methods and presentation of results among studies, thus making generalization of results difficult. While these limitations affect the comprehensiveness of our review, they do not diminish the value of this review in showing a broad overview of the therapeutic value of 0.1% tacrolimus in ophthalmology.
Conclusion
This review is the first in the literature to offer a broad overview of the indications of 0.1% tacrolimus in various ophthalmic conditions, which has never been reported.
0.1% tacrolimus is effective and safe for various eye diseases involving both the anterior segment as well as the adnexa. Its pharmacological property of low systemic absorption and locally limiting side effects such as burning and stinging allows it to be an effective steroid-sparing agent for chronic and recurrent inflammation, not to mention the dampening of side effects with the buildup of tolerance. It is also useful in patients non-responsive or responding poorly to other immunosuppressives such as topical cyclosporine. Long-term use is generally safe across the literature. In cases where patients opt for stopping regular tacrolimus use after the resolution of symptoms, symptoms can still be promptly controlled with the resumption of tacrolimus.
Supplemental Material
sj-docx-1-oed-10.1177_25158414251405461 – Supplemental material for The evidence-based indications of 0.1% topical tacrolimus in ophthalmology: a scoping review
Supplemental material, sj-docx-1-oed-10.1177_25158414251405461 for The evidence-based indications of 0.1% topical tacrolimus in ophthalmology: a scoping review by Patricia Dorothy Lam, Jacqueline W. T. Chan, Rachel W. Y. Tsui, Ke Liu, Vanissa W. S. Chow and Julia Y. Y. Chan in Therapeutic Advances in Ophthalmology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
