Abstract
Central retinal artery occlusion (CRAO) is a rare emergency, often affecting individuals over 60, with risk factors such as hypertension, diabetes, and smoking. Hyperbaric oxygen therapy (HBOT), classified as level IIb by the American Heart Association for CRAO, helps maintain retinal oxygenation during ischemic events by diffusing oxygen through choroidal capillaries. While HBOT appears promising for addressing various vision-threatening conditions, including retinal occlusions and diabetic macular edema, it has not been officially approved for these indications. A 70-year-old male presented with painless vision loss in his right eye, noticed upon waking, accompanied by high blood pressure. On examination, light perception was detected in the right eye, and visual acuity in the left was 0.6. Biomicroscopy revealed CRAO in the right eye, with a cherry-red spot, retinal edema, and absent circulation. Optical coherence tomography (OCT) confirmed retinal edema and subretinal fluid. Given that the thrombolysis window had passed, HBOT was initiated within 48 h of presentation. Following the first treatment, the patient experienced improvement in vision, with light perception expanding beyond the peripheral area. CRAO is an ocular emergency, and early treatment is crucial for improving visual outcomes. HBOT, recommended within 6–12 h of diagnosis, has shown promise in restoring vision. In our case, HBOT administered within 48 h led to improved peripheral vision, with the presence of a cherry-red spot on the macula associated with better recovery. While HBOT can have side effects, none were observed here. Despite challenges in conducting large-scale trials, HBOT remains a potentially effective treatment for CRAO, especially if started early.
Plain language summary
This report describes a rare eye emergency called central retinal artery occlusion (CRAO), which can cause sudden vision loss. It usually affects older adults and is linked to conditions like high blood pressure, diabetes, and smoking.
The patient was treated with hyperbaric oxygen therapy (HBOT). This involves breathing pure oxygen in a pressurized chamber to help deliver oxygen to the retina, even when blood flow is blocked.
A 70-year-old man woke up with vision loss in one eye. Tests showed CRAO, and since the usual treatment window had passed, doctors started HBOT within 48 hours. After the first session, the patient noticed some improvement in his vision.
Although HBOT is not officially approved for CRAO, this case shows it may help if started early. The patient’s vision improved without any side effects. More research is needed, but HBOT could be a useful option for CRAO when other treatments are not possible.
Introduction
Central retinal artery occlusion (CRAO) is a critical ophthalmological emergency that mirrors a cerebral stroke in its impact on vision.1,2 It is a rare condition, affecting approximately 1–12 in every 100,000 patients annually.1,2 Patients typically experience rapid, one-sided, painless vision loss, often resulting in severe visual acuity of 20/400 or worse in 80% of cases if left untreated. Symptoms can range from finger counting to light perception. When blood flow to the central retinal artery is blocked, it leads to ischemia and retinal infarction, causing irreversible damage if not promptly addressed. The retina’s high oxygen consumption rate makes it particularly vulnerable to ischemia, with significant tissue damage occurring after prolonged oxygen deprivation. Prolonged interruptions in blood supply can lead to severe and lasting retinal damage, resulting in permanent vision loss. The exact timeframe for irreversible retinal damage in humans remains uncertain, varying based on individual factors and the nature of the occlusion.1,2 Prolonged interruptions in blood supply exceeding 4 h typically result in severe and enduring retinal damage, leading to permanent vision loss. The timing of irreversible anoxic retinal damage in humans remains uncertain, with some suggesting around 6–6.5 h; however, this estimate lacks reliability due to the diverse factors among patients, occlusion types, and residual perfusion levels. 1 The narrow window of time for effective intervention in CRAO, along with the high prevalence of serious comorbid conditions, complicates treatment decisions. Therefore, when CRAO is diagnosed in settings such as ophthalmology, neurology, or primary care clinics, immediate referral to an emergency department is crucial. Delays in seeking emergency care to pursue further outpatient evaluation or initiate alternative treatments should be avoided. 3 When the central retinal artery is completely blocked, it can cause retinal ischemia and infarction, leading to temporary vision impairment. Emboli, which can originate from plaque in the carotid artery or the heart, particularly due to atrial fibrillation, contribute to this condition. This mechanism is similar to that of an ischemic cerebrovascular accident, often triggered by plaque in the carotid artery or the heart.2,4 Patients with CRAO are more likely to have atrial fibrillation compared to age- and sex-matched individuals. In addition, CRAO signifies an elevated risk of recurrent stroke in patients already diagnosed with atrial fibrillation. Extended cardiac monitoring enhances the probability of detecting atrial fibrillation in CRAO patients.2,4
A single-center study of 103 cases of CRAO at a single center found that 37% of patients had significant carotid artery disease on the same side as the affected eye. 4 This significant carotid disease was defined in the study as ⩾70% narrowing of the artery, arterial dissection, or the presence of an intra-arterial thrombus. In the EAGLE study (European Assessment Group for Lysis in the Eye), which thoroughly examined 84 patients for potential causes, 40% were found to have carotid artery stenosis of ⩾70%.2,5 Emboli causing CRAO can originate not only from the carotid artery but also from the heart, including the aortic and mitral valves, the aortic arch, or the great vessels. This is reflected in the risk factor profile of CRAO patients. The EAGLE study revealed a notable prevalence of cardiovascular risk factors among the 77 patients evaluated: obesity (82%), hypertension (73%), tobacco use (49%), hypercholesterolemia (49%), and diabetes (14%). Overall, 67% of the patients had at least one cardiovascular risk factor. In addition, 20% had cardiac arrhythmias, 17% had valvular heart disease, and 5% had heart failure. Patients with CRAO are more likely to have atrial fibrillation compared to age- and sex-matched individuals. Furthermore, CRAO signifies an increased risk of recurrent stroke in those already diagnosed with atrial fibrillation.2,5 Prolonged cardiac monitoring improves the chances of identifying atrial fibrillation in CRAO patients. 6 As a result, both the American Heart Association (AHA) and the National Stroke Association (NSA) recommend a thorough cardiovascular evaluation for all patients presenting with retinal ischemia, particularly CRAO. Various studies have identified significant risk factors for retinal artery occlusions, such as nicotine use, hypercholesterolemia, arterial hypertension, and internal carotid artery stenosis, among others, even post-COVID-vaccine or failed chiropractor neck manipulation. While individuals with hypertensive arteriosclerosis are more likely to develop CRAO, cases have also been reported, though rarely, in patients with endocarditis.2,7–12
When hyperbaric oxygen therapy (HBOT) is administered, the excessively constricted blood vessels stop contracting further, contrary to the usual vasoconstrictive response. This occurs because the vessels receive only a limited amount of oxygen, as they are already nearly fully constricted. As a result, the high concentration of oxygen has a pronounced effect, leading to a significantly greater increase in oxygen delivery to the ischemic retinal tissue in patients with hypertension compared to those without hypertension. 1 HBOT is one of the treatment options for CRAO patients and has demonstrated clinical effectiveness when administered within an appropriate time frame, typically less than 24 h after the initial onset of symptoms. The main advantages of HBOT are hyperoxygenation and a reduction in the size of gas bubbles in the bloodstream. Oxygen, which makes up 21% of the air we breathe, is crucial for metabolic processes. A key feature of HBOT is the administration of 100% oxygen at pressures two to three times higher than atmospheric pressure at sea level. During HBOT, arterial oxygen pressure and tissue oxygen pressure can reach up to 2000 mm of mercury and 400 mm of mercury, respectively. This increase in pressure significantly enhances the oxygen gradient, or the transfer of oxygen into tissues, by a factor of 20. Such a boost can positively affect various biochemical, cellular, and physiological processes. In the healing process, oxidants can function as cellular messengers.13,14 A higher concentration of oxygen in the bloodstream enhances the tissues’ ability to heal effectively. In a controlled environment, normal breathing with room air can achieve nearly 100% oxygen saturation in hemoglobin. However, inhaling 100% oxygen under high atmospheric pressure increases the solubility of oxygen in the blood, raising it from 0.3% to 6% by volume. Although this amount of oxygen dissolved in plasma is relatively small, it is adequate to meet the oxygen needs of the body’s tissues. It has been established that increasing the partial pressure of oxygen in the ocular tissues, including the vitreous body and inner retina, enables the choroidal vasculature to deliver more oxygen to these tissues. Administering hyperbaric oxygen using medical compression chambers is a viable approach for preserving tissue viability in cases of retinal vascular obstruction until reperfusion can be achieved. 1
HBOT in CRAO
The constriction of retinal arteries during this treatment is most likely caused by the interaction between free oxygen radicals and nitric oxide, combined with the autoregulatory processes involved. Within the first 10 min of starting HBOT, there is a significant reduction in blood flow. However, following the completion of hyperbaric oxygen therapy, rapid vasodilation occurs due to increased nitric oxide production and the immediate utilization of free oxygen after the treatment ends. 15
While HBOT can cause retinal arteries to constrict, oxygen saturation can increase up to 23% without harming the retina. Specifically, the beneficial effect of HBOT in treating retinal vascular diseases is attributed to improvements in retinal and macular oxygenation due to hyperoxia. This is particularly effective in areas with poor perfusion, interstitial edema, or a thickened basal membrane, as well as in reducing fluid leakage that contributes to retinopathy. However, there is ongoing debate about the effectiveness of HBOT in treating retinal vascular occlusions associated with macular edema, despite current clinical evidence supporting its use. Concerns include the potential production of excess free radicals and the placebo effect. Under normal conditions, choroidal circulation supplies about 60% of the oxygen needed for retinal function, which increases to 100% under hyperbaric conditions. Theoretically, using HBOT to treat CRAO could enhance blood flow from collateral and choroidal circulations, potentially meeting the metabolic demands of retinal cells while allowing for the natural re-cannulation of the central retinal artery, thereby preserving retinal tissue. However, this hypothesis lacks supporting evidence. Nevertheless, hyperbaric oxygen has been shown to reduce tissue edema and ischemia-reperfusion injury during recanalization. For the inner retinal layers to function effectively, oxygen levels in choroidal circulation must be sufficiently high. If this is achieved, enough oxygen may diffuse downstream to maintain some degree of viability. Studies using CRAO animal models have shown that preserving normal inner retinal layer architecture is possible when choroidal oxygen levels are high enough to diffuse into the inner retinal layers.
Hyperbaric therapy at 203 kPa provides an inspired oxygen partial pressure nearly 10 times higher than that of normal atmospheric air. This increased pressure allows oxygen to diffuse from the choroidal circulation to the ischemic retina, reactivating cellular metabolism and preserving retinal tissue while giving time for emboli to dissolve or migrate. Most patients in this cohort reported visual improvement after their first HBOT session, likely due to reduced retinal edema and improved visual acuity. However, HBOT can only extend the time for restoring retinal circulation by a few hours and is ineffective if the retina has already infarcted. Therefore, it is most beneficial for patients who receive treatment within 10 h of artery blockage, as these patients generally show better visual recovery. 1
The primary rationale for using intraocular hyperbaric oxygen therapy in ischemic eye disorders is that the vitreous body, despite being a liquid medium with around 80% dissolved nitrogen, becomes saturated with oxygen during treatment. Once the nitrogen is replaced with oxygen, the vitreous can act as an oxygen reservoir. Oxygen desaturation from the vitreous to the retina occurs more slowly than saturation due to the lower partial pressure gradient outside the chamber. This allows the vitreous to serve as an oxygen depot during the initial days of therapy. In the choroidal circulation, rapid blood flow and low arteriovenous oxygen difference limit the impact of oxygen tension changes on blood flow compared to the retina. While high levels of free dissolved oxygen can be harmful, this method facilitates a higher diffusion gradient from the choroidal circulation to the inner retinal layers. 16
HBOT’s molecular mechanisms
The influx of calcium during ischemia and reperfusion leads to excitotoxicity, mitochondrial dysfunction, and ultimately cell death. This calcium overload is often triggered by glutamate receptor activation and impaired ATP-dependent ion transport, resulting in elevated intracellular concentrations of sodium, chloride, and water, which exacerbate cytotoxic edema.1,17,18 Upon reperfusion, enzymes such as xanthine oxidase generate reactive oxygen species (ROS), including superoxide and hydrogen peroxide, which contribute to oxidative stress and lipid peroxidation. These free radicals damage cellular membranes, proteins, and DNA, amplifying tissue injury.1,17,18 Acidosis, a hallmark of ischemic tissue, further exacerbates injury by promoting lysosomal rupture, enzyme activation, and inflammatory signaling. The accumulation of lactic acid and failure of pH regulation mechanisms can lead to irreversible cellular damage.1,17,18 HBOT, by delivering 100% oxygen at elevated atmospheric pressures, enhances oxygen diffusion through choroidal circulation to ischemic retinal tissues. This oxygenation helps stabilize mitochondrial function, reduce ROS production, and support ATP synthesis. Moreover, HBOT has been shown to correct local acidosis by improving aerobic metabolism and facilitating the removal of acidic byproducts, thereby mitigating the cascade of ischemia-reperfusion injury. This approach may preserve retinal tissue within the ischemic penumbra, reducing the extent of irreversible damage and improving visual outcomes when administered promptly after the onset of CRAO.1,17
HBOT contraindications
HBOT has both absolute and relative contraindications. Absolute contraindications include untreated tension pneumothorax and certain chemotherapy treatments.1,17 Relative contraindications are pregnancy, epilepsy, severe claustrophobia, acute infections (especially of the upper respiratory tract), chronic sinusitis, or uncontrolled high fever. According to the guidelines from the Gesellschaft für Tauch- und Überdruckmedizin (GTUM), European Underwater and Baromedical Society (EUBS), and Underwater and Hyperbaric Medical Society (UHMS), there are no known contraindications for pressure chamber treatment in emergency situations. Extremely severe or long-lasting side effects are very rare. Inadequate pressure equalization can result in barotrauma by damaging closed, air-filled body cavities and their adjacent anatomical structures. Patients with chronic bronchitis, asthma, or pulmonary emphysema are at higher risk for lung complications. 1 According to current scientific literature, there is no conclusive evidence that HBOT is contraindicated in patients with lung cancer. In fact, multiple systematic reviews have concluded that HBOT does not promote tumor growth or recurrence. On the contrary, in certain cancer subtypes, HBOT may even exert tumor-inhibitory effects.19,20 A temporary increase in oxygen sensitivity in the brain, known as oxygen cramp or sensory disturbances, may occur but is always reversible and has no lasting negative effects. 1 It is important to note that the use of HBOT should always be based on individual clinical assessment, particularly in cases of active inflammation or malignancy. The treating physician must consider the patient’s ocular and overall condition, and any potential risks, before initiating the therapy.
Case report
A 70-year-old male patient arrived at our emergency department due to painless vision loss in his right eye. He noticed the symptoms in the morning after waking up, and his blood pressure was measured at 190 mmHg.
The patient had a history of smoking, which he quit 40 years ago. He consumes alcohol moderately (1–2 units/day). Case history includes type 2 diabetes mellitus and hypertension, with recorded maximum blood pressure values reaching 280/140 mmHg. In 2009, the patient underwent coronary artery bypass surgery. In 2012, he experienced an acute angle closure in the right eye, with an Intraocular Pressure (IOP) of 38 mmHg, treated with Yttrium-Aluminum-Garnet (YAG) iridotomy and medical therapy (mannitol, acetazolamide, and timolol), resulting in stabilization. The patient has no known drug or other allergies or current infection (Supplemental Material).
The initial examination (Table 1) during the emergency hours at the Ophthalmology Clinic of the Clinical Center of the University of Pécs (UP MS CC) revealed that his best-corrected visual acuity in the right eye was light perception from 1 m at the upper temporal region, without object vision, while in the left eye, it was 0.6 (20/33) with correction. His last visit to our clinic was in 2012, when his visual acuity was 0.8–1.0 (20/25-20/20) in both eyes. After taking his medical history, we measured his blood pressure again, which was 200 mmHg systolic value. Slit-lamp examination showed a clear anterior segment with fine nuclear opacity and YAG iridotomy openings. The IOP measured with a Goldman applanation tonometer was 11/11 mmHg. Biomicroscopic ophthalmoscopy with pupil dilation revealed a CRAO in the right eye, with a blurred optic disc margin, a cherry-red spot in the macula surrounded by edematous retina, dilated veins, fragmented blood columns in the arteries, and a lack of circulation. Signs of hypertension were also visible in the left fundus, with a sharp-edged optic disc and pigment disturbance in the macula. A macular optical coherence tomography (OCT) examination confirmed the right-sided circulatory disorder, showing a disorganized and edematous retinal structure. In the right macula, drusen and fine pigment disturbance were observed (exact measurement in micrometers needed) (Figure 1).
Short summary of the timeline of clinical events.
CRAO, central retinal artery occlusion; FA, fluorescein angiography; HBOT, hyperbaric oxygen therapy; OCT, optical coherence tomography.

Visual field test before the first HBOT treatment (a); after the first treatment: MS: 1.9 dB, MD: 24.7 dB, sLV: 5.1 dB (b); on the second day of treatment: MS: 1.4 dB, MD: 25.2 dB, sLV: 4.1 dB (c); on the third day of treatment: MS: 2.4 dB, MD: 24.2 dB, sLV: 5.2 dB (d); on the fifth day of treatment: MS: 4.3 dB, MD: 22.3 dB, sLV: 5.9 dB (e); on the sixth day of treatment: MS: 3.8 dB, MD: 22.8 dB, sLV: 6.3 dB (f).
Fluorescein angiography (FA) and OCT angiography were not performed during the initial emergency care due to time constraints and the patient’s unstable blood pressure. These imaging modalities were later conducted during follow-up visits. The clinical presentation was consistent with CRAO. However, differential diagnoses considered included cilioretinal artery occlusion, which typically spares central vision, and ocular ischemic syndrome, which often presents with bilateral findings and systemic vascular symptoms. These were ruled out based on unilateral presentation, fundoscopic findings, and imaging results. The following day, hyperbaric therapy was decided on as the patient arrived beyond the time window for thrombolysis according to the protocol. The patient was informed about the hyperbaric procedure, and he consented to the treatment, which was initiated within 48 h. His vision improved after the first treatment, with light perception appearing in a larger area, not just at the periphery.
HBOT treatment
During HBOT, the patient is in a pressure-controlled environment, known as a “hyperbaric chamber.” This chamber can be a single-person “monochamber” or a multi-person “multichamber.” The advantage of the multi-person chamber is that accompanying staff can be present with the patient and provide immediate assistance if needed. Inside the chamber, the pressure is higher than atmospheric pressure, typically ranging from 1.5 to 3 absolute atmospheres, which is 0.5 to 2 atmospheres above normal atmospheric pressure. The patient breathes 100% oxygen with intermittent oxygen breaks depending on the need to prevent oxygen toxicity versus the need for optimal oxygenation. Oxygen is delivered “per pulmo” via a mask. Treatments are usually repeated daily, and in severe cases, multiple times a day. The pressure and duration of the treatment depend on the specific condition being treated. The high oxygen content generally causes vasoconstriction, which, along with the increased environmental pressure, helps reduce edema. The significance of dissolved oxygen content is crucial. Oxygen does not dissolve well in blood and tissue fluids; its solubility depends on environmental pressure (Henry’s law). The solubility value is 0.003 mL/mmHg/100 mL. In atmospheric air, the amount of dissolved oxygen is minimal, approximately 0.2–0.3 mL/100 mL, with oxygen transport mainly dependent on the amount of hemoglobin (1.34 mL per 15 g/100 mL hemoglobin = 20 mL/100 mL blood). When administering 100% oxygen at sea level pressure, the dissolved oxygen increases to about 2 mL/100 mL. If the pressure is increased to, for example, three atmospheres (bars), the dissolved oxygen can exceed 6 mL/100 mL of blood, which at a 6-L minute volume equates to 360 mL/min. This amount can cover the body’s resting oxygen needs (250–300 mL/min, 3 mL/kg/min). Since a large quantity of dissolved oxygen can diffuse further from the capillaries into tissue fluids, it can reach areas that are otherwise deprived of blood flow. Typically, one treatment is administered daily, with the main exceptions being emergency cases (e.g., gas gangrene, air embolism, and carbon monoxide poisoning). There are various recommendations regarding the applied pressure, its incremental increase, and the duration of treatment. In our case, to achieve rapid oxygenation, we initially followed a modified Boerema protocol, starting at an absolute pressure of 3 bars for 50 min, and then continued at 2.4 bars. The total duration of isobaric oxygen administration was 90 min. Both the pressure and the induced vasoconstriction can reduce retinal edema, potentially improving oxygenation further. Since we started the treatment in the late afternoon, we administered only one session that day, but the patient reported a return of light perception immediately. From the following day, we conducted two treatments daily for 3 days, with gradual improvement. Starting from the third treatment, we used a problematic regimen involving an absolute pressure of 2.4 bars with 90 min of isobaric oxygen inhalation. Treatments were performed in a HAUX Starmed 2500 chamber (10 + 2-person capacity). Due to a delay of one and a half days before starting the treatment, we opted for higher pressure initially. Even partial recovery of central and peripheral vision was a significant benefit for the patient. Central retinal artery recanalization typically begins around 72 h, which influences the duration of treatment. Overall, we administered 10 treatments over 6 days. During this period, the patient’s peripheral vision returned, he subjectively felt well, and there were no complications caused by the treatment. The blood pressure (systolic: 171.87 ± 6.71 mmHg; diastolic: 92.53 ± 5.04 mmHg; heart rate: 101.20/min ± 32.07/min) has been controlled throughout the entire HBO therapy and follow-up period by a multidisciplinary team, including cardiologist, general internist, sports medicine physician, oxyologist, and ophthalmologist.
During the HBOT, multiple visual field tests were performed to monitor the progression of the patient’s visual field (EyeSuite Static perimetry, V3.6.1 OCTOPUS 600). Based on the results of the visual fields, central vision did not return, but an improvement was observed in the peripheral visual field, which the patient also experienced (Figure 1).
Follow-up examination after the HBOT treatment
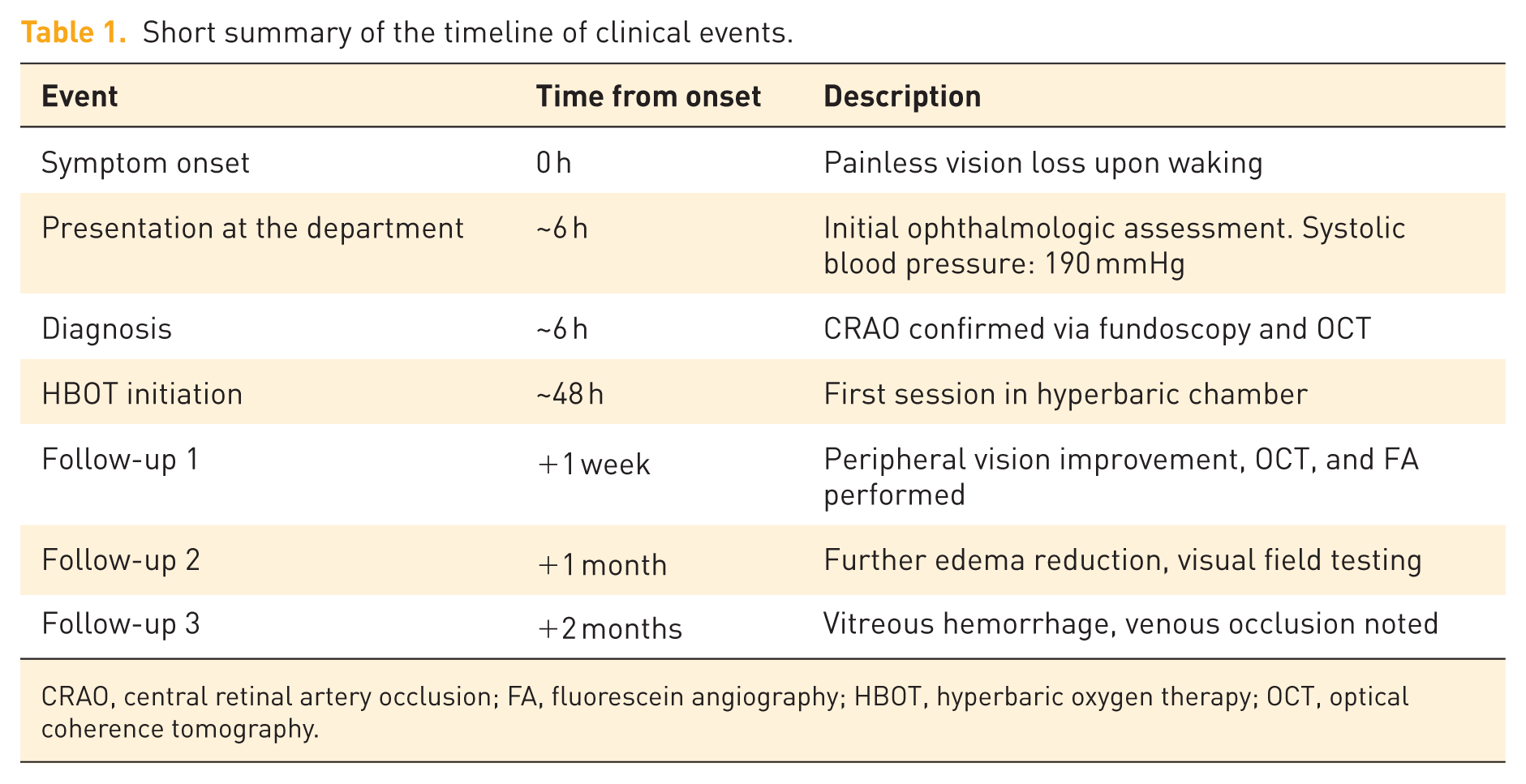
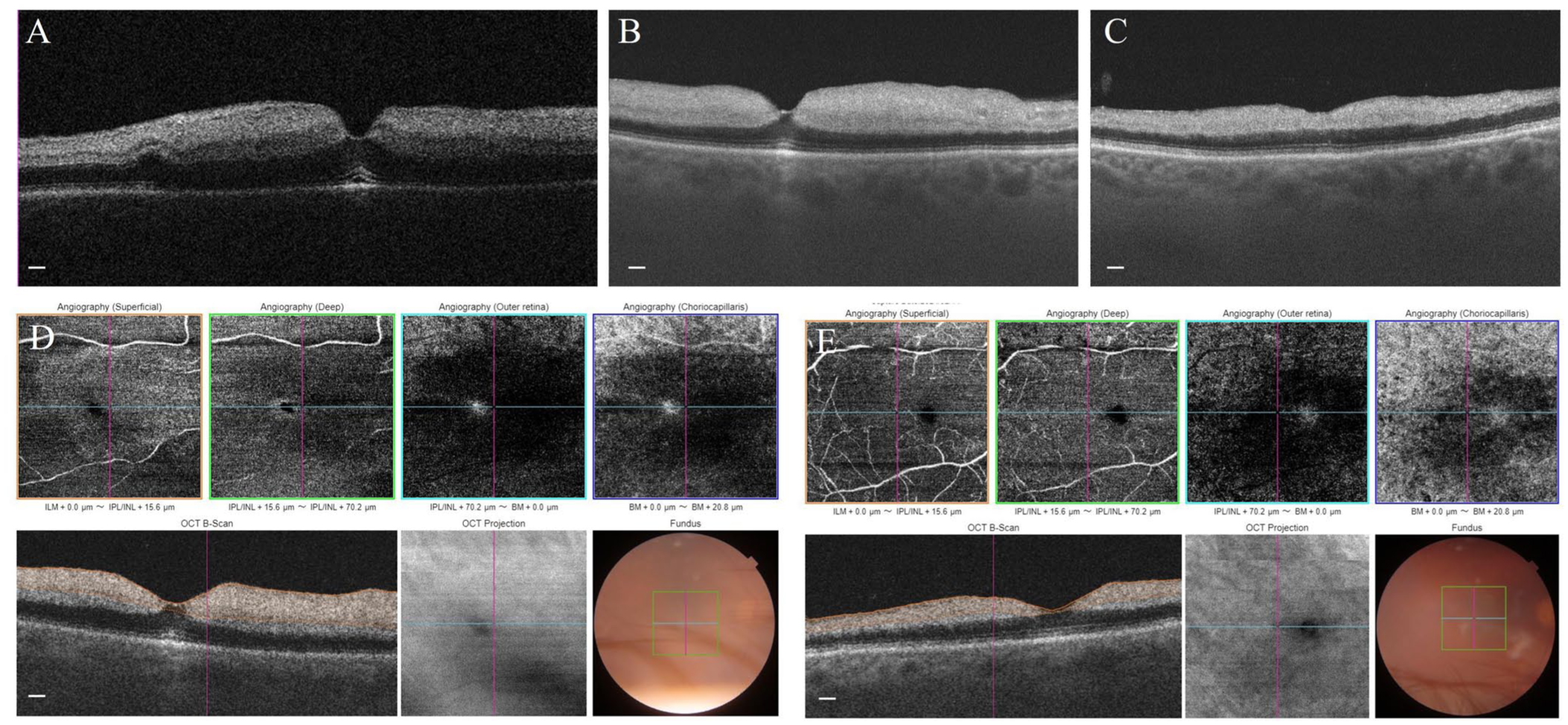
The patient returned to our clinic for a follow-up examination 1 week after the treatment (Table 1). This time, his visual acuity in the affected eye was light perception from 1 m, and he could detect hand movements in front of the eye. Slit-lamp examination showed no changes compared to the initial assessment. During the dilated fundus examination, the macula in the right eye was still edematous, the optic disc had slightly blurred edges, but circulation in the retinal vessels was less fragmented. No changes were found in the other eye’s status compared to the previous examination. An OCT examination showed that the retina in the right macula area was still disorganized, but the edema had decreased (Figure 2). No new findings were detected in the OCT of the opposite eye. Optical coherence tomography angiography (OCTA) images were also taken of the macular and optic disc regions (Figures 2 and 3). An edematous optic disc is visible on the papilla OCT image (Figure 3). A fundus photo was taken, showing an edematous macula and fragmented blood columns (Figure 4). FA examination was performed, revealing slower and more fragmented circulation (Figure 5).
Macular edema and disorganized retina on macula OCT in the first examination (a). Reduction of edema 1 week after the HBOT on macula OCT (CMT O.D./O.S.: 221 µm/167 µm, PMT O.D./O.S.: 408 µm/301 µm) (b). Reduction of edema and atrophy 1 month after the CRAO on macula OCT (CMT O.D./O.S.: 170 µm/164 µm, PMT O.D./O.S.: 318 µm/293 µm) (c). OCT angiography one month after the CRAO (d); and OCT angiography 1 week after the HBOT (e). Scale bar: 50 µm.
Edematous optic disc on OCT reports 1 week after the HBOT (O.D. RNFL-S/N/I/T: 176/108/169/139 µm and O.S. RNFL-S/N/I/T: 94/70/107/62 µm) (a). Diffuse nerve fiber loss 1 month after the CRAO (O.D. RNFL-S/N/I/T: 18/21/72/1 µm and O.S. RNFL-S/N/I/T: 98/74/117/65 µm) (b). OCT angiography 1 week after HBOT (c). OCT angiography 1 month after the CRAO (d). Scale bar: 50 µm.

Color fundus photo on the right (a) and the left eye; (b) 1 week after HBOT.

FA examination phases: baseline photo (a); at 25 s (b); photo at 33 s (c); photo at 40 s (d).
At the one-month follow-up (Table 2), the visual acuity in the right eye was counting fingers in front of the eye and light perception from 1 m. The patient reported significant improvement in peripheral vision and stated that he could see shapes. Slit-lamp examination showed no changes compared to previous results. The status of the other eye remained unchanged. The OCT scan showed further reduction in edema in the right eye, but the retinal structure remained disorganized (Figure 2). The OCT of the optic disc in the right eye revealed diffuse nerve fiber damage, while the nerve fiber thickness in the left eye was within the normal range (Figure 3). OCTA images were also taken of the macular and optic disc regions (Figures 2 and 3). Goldmann kinetic perimetry of the right eye showed a visual field boundary between 30° and 50° with III/4 and V/4 stimuli, while the left eye did not exhibit significant abnormalities (ZEISS).
Ophthalmological measurements obtained at 1 week after the initiation of HBOT, and at 1-month follow-up.

These parameters were used to monitor structural and functional retinal changes associated with HBOT.
BCVA, Best-Corrected Visual Acuity; CMT, Central Macular Thickness; IOP, Intraocular Pressure; MD, Mean Deviation; MS, Mean Sensitivity; O.D., Oculus Dexter (Right Eye); O.S., Oculus Sinister (Left Eye); PMT, Peripheral Macular Thickness; RNFL-I, Retinal Nerve Fiber Layer—Inferior; RNFL-N, Retinal Nerve Fiber Layer—Nasal; RNFL-S, Retinal Nerve Fiber Layer—Superior; RNFL-T, Retinal Nerve Fiber Layer—Temporal; sLV, Standard Loss Variance.
At the 2-month follow-up, the visual acuity in the right eye remained unchanged at light perception from 1 m and counting fingers in front of the eye. Slit-lamp examination revealed vitreous hemorrhage. No changes were found in the status of the left eye. Fundus examination showed a pale optic disc and macula in the right eye, with suspected blotchy hemorrhages in the periphery, though not fully assessable due to the vitreous hemorrhage. The left eye’s fundus condition was unchanged. OCT examination showed atrophy in the right macula, and the diffuse nerve fiber damage at the optic disc was similar to the previous examination. Goldmann kinetic perimetry of the right eye showed slight constriction in the superior and temporal visual fields, partly due to the hemorrhage. No abnormalities were found in the OCT and visual field examinations of the left eye. The patient reported fluctuating blood pressure, often exceeding 200 mmHg. A venous occlusion had also developed in the right eye, leading to additional treatments at our clinic. The results of further follow-ups were influenced by the venous occlusion, preventing us from continuing to record the outcomes of hyperbaric treatment.
Following the completion of HBOT, the patient expressed a high level of subjective satisfaction with the outcome. Notably, he reported being able to orient himself using peripheral vision in the affected eye, even when the contralateral, unaffected eye was covered. He further stated that, if he would lose vision in his better-seeing eye in the future, he would still be able to manage daily life independently, despite the absence of central visual acuity. This subjective feedback highlights the potential functional relevance of partial visual recovery, even in cases where full restoration of vision is not achieved.
Discussion
CRAO is an ocular emergency similar to an ischemic stroke. Although various reports propose different treatment approaches for CRAO, there is limited evidence supporting a definitive management strategy. According to the European Committee of Hyperbaric Medicine (ECHM) at the Tenth European Consensus Conference on Hyperbaric Medicine (2016), Type 2 therapy is recommended for HBOT due to acceptable evidence levels, while Level C therapy is recommended when randomized controlled trials (RCTs) are not feasible but there is substantial international evidence. In addition, clinical practice guidelines of the American College of Cardiology/American Heart Association (ACC/AHA) classify HBOT as IIb (Usefulness/effectiveness is unknown/unclear/uncertain or not well-established), indicating that it is not unreasonable to proceed with the procedure or administer the treatment in such cases.1,21,22 It is widely recognized that the time between diagnosis and therapy is crucial for a patient’s visual prognosis. In CRAO, early HBOT is vital as it delivers oxygen to the retinal layers through the choroid during the disease’s early stages. However, the optimal timing for treatment is debated. Generally, the sooner treatment is administered after injury, the better the chances of recovering the ischemic, yet still functional, retina. Inner retinal cells have a limited window for recovery from hypoxia, regardless of reperfusion. 1 In Wu et al.’s meta-analysis, which included seven RCT publications, the timing of treatment from onset could not be statistically analyzed because all RCTs were conducted within the same specific time window. 13 Butler et al. 23 suggest that the strongest evidence indicates success when HBOT is initiated within 12 h of a CRAO diagnosis. Hertzog et al., as well as Beiran et al., found that HBOT is most effective when started within 8 h of the onset of vision loss.24,25 Another group of researchers concluded that individuals with CRAO who receive HBO therapy within 6–8 h of symptom onset achieve the best outcomes. 13 Based on the studies mentioned above, there is no definitive time frame for starting HBOT, but all studies agree on the importance of initiating it as soon as possible after a CRAO diagnosis. Retinal blood flow can be restored within 72 h after recanalization surgery. However, if ischemia and hypoxia have caused cell death in the inner retinal layers, vision may not be restored even with recanalization. CRAO often leads to vision loss because the inner retinal layers, normally supplied by the retinal artery, are occluded. If these layers receive enough oxygen via diffusion from the choroidal circulation, they may continue to function. HBOT’s effectiveness in oxygenating these ischemic layers depends on choroidal perfusion rates. Without blood flow to the posterior ciliary vessels, collateral circulation to the inner retina is limited, especially if the ophthalmic artery is fully obstructed. Maintaining adequate blood oxygen levels is crucial to keep the retina viable until natural recanalization typically occurs within 72 h. 1 In our case, HBOT therapy began after 24 h but within 48 h and lasted for 5 days. The patient’s visual acuity improved, with greater improvement observed in peripheral vision than in central visual acuity. The improvement in peripheral vision was trackable through visual field testing, and the patient also noticed this improvement. Our case supports the notion that the extent of central vision improvement decreases with a delayed time window. However, it is still beneficial to start HBOT within 48 h of CRAO, as significant changes in peripheral vision can still occur, positively impacting the patient’s daily life.
HBOT was initiated in our case within 48 h of symptom onset, applying a modified Boerema protocol with an initial pressure of 3.0 ATA for 50 min, followed by 2.4 ATA for 40 min, totaling 90 min per session. The patient received two sessions daily for 3 days, then one session daily, completing 10 treatments over 6 days in a multiplace chamber (HAUX Starmed 2500). Despite the delayed initiation, peripheral visual improvement and subjective functional benefit were observed. When compared to Hadanny et al., our protocol used higher initial pressure (3.0 ATA vs. 2.0–2.4 ATA) and more intensive early treatment (two sessions daily vs three in the first 24 h and then once daily). Both studies utilized multiplace chambers and 90-min sessions with 100% oxygen. However, Hadanny et al. discontinued treatment upon plateauing BCVA improvement, 26 while our protocol continued based on peripheral recovery and patient-reported outcomes. Lee et al. implemented HBOT as an emergency rescue therapy, using 2.8 ATA for 90 min twice daily for 3 days, followed by 120-min sessions once daily, aiming for continuous treatment over 14 days. 27 Their approach was more prolonged and standardized, with HBOT prioritized over conventional therapies unless contraindicated. Unlike our case, Lee et al. reported improvements in BCVA and preservation of outer retinal layers on enhanced depth imaging I-OCT, while our patient showed peripheral recovery without central acuity improvement and progressive retinal disorganization. Yip et al. also began with 2.8 ATA for 90 min, followed by 2.4 ATA sessions, aligning with US Navy protocols. 28 Their staged decompression and emergency-first approach resemble our initial intensity, though their protocol was more standardized and less tailored to individual response. Thoongsuwan et al. applied a unique regimen with 2.0 ATA for 180 min in the first session, followed by 2.4 ATA for 90 min daily, incorporating air breaks and a fixed 5-day duration regardless of visual acuity changes. Their protocol emphasized oxygen saturation and safety, differing from our more flexible and response-driven approach. 29 While all protocols share core elements: 100% oxygen, elevated pressure, and early initiation, our approach stands out for its higher initial pressure, adaptive scheduling, and focus on peripheral functional recovery. The variability in outcomes across studies underscores the need for individualized HBOT strategies, especially when treatment initiation is delayed or complicated by comorbidities such as venous occlusion.
The characteristic fundoscopic appearance—a cherry-red spot at the fovea accompanied by surrounding inner retinal edema—typically develops within approximately 100 min. 2 Hadanny et al. recently found that patients with a cherry-red spot (CRS) on their macula during initial fundoscopy showed significantly better visual improvement and higher treatment success rates compared to those without CRS. Specifically, 86.0% of patients with CRS experienced meaningful visual improvement, versus 49.4% of those without CRS. The authors suggest that CRS is a key marker of irreversible retinal damage in candidates for high-dose oxygen therapy and should be considered the most critical indicator of treatment success, instead of the time elapsed since symptom onset. 26 Although HBOT is described to be most effective when initiated within the first 12–24 h after symptom onset, our case demonstrates that even delayed treatment can result in measurable visual improvement. This observation aligns with the findings of Yang et al., who recently reported that visual recovery is still possible in CRAO patients treated outside the conventional therapeutic window, particularly when some degree of retinal viability is preserved. 30 Their study emphasizes that the presence of residual retinal function and structural integrity, as assessed by multimodal imaging, may be more predictive of treatment response than time alone. In our case, the presence of a CRS and the partial preservation of the Retinal Nerve Fiber Layer (RNFL) thickness may have contributed to the favorable outcome, despite the delayed initiation of HBOT. 31 Our findings also support what Hadanny et al. previously reported. 26
In contrast to the findings of Lee et al., who reported significant improvements in BCVA and preservation of outer retinal layer thickness using enhanced depth imaging-OCT over a 6-month period, 27 our case demonstrated partial peripheral visual recovery without central acuity improvement. OCT imaging of our patients revealed persistent disorganization of the retinal structure and progressive macular atrophy, despite reduced edema. While Lee et al. 27 observed increased central choroidal thickness and maintained outer retinal integrity, our OCT and OCT-angiography findings showed diffuse nerve fiber damage and fragmentation of retinal circulation. Furthermore, our patient developed a secondary venous occlusion, which likely confounded the long-term OCT outcomes and limited the ability to assess sustained structural recovery. Nonetheless, the subjective improvement in peripheral vision and functional independence highlights the potential benefit of HBOT even in delayed or complex cases.
The literature describes several ocular side effects related to HBOT, such as oxygen toxicity, increased IOP, cataracts, reversible myopia, and refractive errors, among others. 1 In our case, no side effects attributable to HBOT were observed. The patient had a preexisting nuclear cataract, which did not worsen. The venous occlusion that developed after treatment did not occur immediately following the therapy and is attributed to the persistently high and fluctuating systemic blood pressure. This case is unique due to the initiation of HBOT beyond the conventional 24-h window, yet resulting in measurable peripheral visual improvement. The presence of a CRS and preserved RNFL thickness may have contributed to the favorable outcome.
Conclusion
Conducting large-scale RCTs for CRAO is challenging due to its low incidence compared to other ischemic events, making it difficult to find effective treatments. Published trials have shown that no intervention is conclusively more effective than doing nothing. 1 Well-designed prospective trials are needed to identify the most effective treatment for CRAO. Previous studies suggest that HBOT, as a safe alternative, can improve visual acuity associated with CRAO before therapy. While waiting for definitive outcomes, ophthalmologists will continue their efforts to increase oxygenation of the ischemic retina. HBOT may be a relevant and approved treatment option for individuals presenting CRAO symptoms within 24 h, offering a low-risk profile and potential benefit in improving visual prognosis. This case report has several limitations that should be acknowledged. First, the initiation of hyperbaric oxygen therapy (HBOT) was delayed, which may have influenced the clinical outcome and limited the ability to assess the full therapeutic potential of early intervention. Second, the occurrence of venous occlusion introduced a confounding factor that complicates the interpretation of the patient’s response to treatment. Finally, as a single case report, the findings are inherently limited in their generalizability and should be interpreted with caution. Further studies involving larger patient cohorts are necessary to validate these observations.
Supplemental Material
sj-pdf-1-oed-10.1177_25158414251405382 – Supplemental material for Exploring hyperbaric oxygen therapy for central retinal artery occlusion beyond 24 h: case report
Supplemental material, sj-pdf-1-oed-10.1177_25158414251405382 for Exploring hyperbaric oxygen therapy for central retinal artery occlusion beyond 24 h: case report by Dóra Werling, Tibor Rák, Gábor Kanász, Anna Göbl, Gábor Borbély and Adrienne Csutak in Therapeutic Advances in Ophthalmology
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to the staff of Kolos Vaszary Hospital (Esztergom, Hungary) for their direct and indirect contributions to providing hyperbaric oxygen therapy for patients of the Department of Ophthalmology, Clinical Centre, University of Pécs Medical School. The authors also thank the dedicated professionals of the Hungarian National Ambulance Service for their essential role in ensuring the timely transportation of patients.
Declarations
Generative AI statement
The authors declare that no Generative AI was used in the creation of this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
