Abstract
Background:
Intravitreal injection (IVI) is a common practice in today’s ophthalmology clinics. The pain that patients will experience after the application may be important in compliance with the treatment.
Objectives:
This study aimed to investigate the correlation between various clinical characteristics of patients receiving IVI and corresponding visual analogue scale (VAS) scores (0: no pain to 10: severe pain).
Design:
Single-centre, Prospective study.
Methods:
A total of 313 participants (168 females, 145 males) with a mean age of 66.91 ± 9.67 years underwent IVI for diabetic retinopathy (DRP), retinal vein occlusion (RVO), or age-related macular degeneration (AMD). Eye examinations, including visual acuity and intraocular pressure measurements, were also conducted, and injection indications were determined based on dilated fundus examinations and spectral domain optical coherence tomography images. Following the injections, the researchers solicited VAS scores ranging from 0 to 10 (no pain to severe pain). The study explored the relationships between clinical characteristics, headache frequency, joint and muscle pain, analgesic use, surgical history, antidepressant use, vasovagal syncope, previous injections, and VAS score.
Results:
The mean VAS score was 4.77 ± 2.90. While DRP and RVO had similar VAS scores (4.95 ± 2.98 and 5.22 ± 2.70, respectively), the AMD group had significantly lower scores (4.09 ± 2.64). Compared with nonusers, antidepressant users had significantly greater VAS scores (5.79 ± 3.43) (4.52 ± 2.70) (p < 0.05). Patients with a history of syncope had significantly greater VAS scores (p < 0.05). In patients reporting monthly headaches, a positive correlation was found between headache frequency and VAS score (r = 0.23, p < 0.01).
Conclusion:
For individuals experiencing daily headaches, inquiries about vasovagal syncope and antidepressant use may be beneficial, considering the potential association of these symptoms with higher VAS scores after IVIs.
Introduction
Intravitreal injections (IVIs) are among the frequently utilized interventional procedures in contemporary ophthalmic practices. 1 They are commonly employed, particularly in the treatment of diabetic retinopathy (DRP), macular oedema due to retinal vein occlusion (RVO), and oedema associated with age-related macular degeneration (AMD). The main agents used in this context are anti-vascular endothelial growth factor (anti-VEGF) agents.2,3 In addition, intravitreal treatments have been used for diseases such as retinopathy of prematurity, posterior segment infections and inflammatory conditions. 4 However, due to the short half-life of agents used in chronic diseases such as DRP, RVO, and neovascular AMD, repetitive IVIs are necessary for continuous treatment. The frequency of IVIs varies by country but averages between 3.5 and 8 injections per year for conditions such as DRP.
These injections can be safely performed under local anaesthesia, typically from 3.5 to 4 mm away from the limbus. However, complications such as endophthalmitis, vitreous haemorrhage, retinal tears, detachments, and traumatic cataracts related to trauma to the posterior surface of the lens may occur, albeit rarely.5,6 Consequently, patients often experience fear and anxiety about undergoing such a procedure. 7 Various techniques, anaesthetic agents, and preoperative analgesics have been employed to enhance patient comfort during this procedure. Furthermore, investigations into the effects of different needle gauges have been conducted to assess patients’ pain. 8
Although parameters such as the type of needle, type of anaesthesia, or active ingredients have been compared with patients’ pain scores during injections, the evaluation of pain-related systemic diseases and the analgesic drugs used by the patient have not been assessed until now. Considering the average age of the patients, joint pains or rheumatic pains, the frequency of headaches, and the frequency of analgesic drug use related to these causes of systemic pain were recorded. Additionally, the use of antidepressants related to patients’ anxiety status was investigated. Finally, we tried to clarify whether those who had previously experienced vasovagal syncope during any interventional procedure experienced pain and whether they experienced pain more due to this acquired anxiety. We believe that querying the clinical details of patients is crucial for preinjection preparation.
We believe this study is valuable because of how patients assess pain before IVI and how, based on this experience, clinicians can anticipate the pain patients may experience by inquiring about it in their medical history.
Methods
In this single-centre, prospective study, we evaluated the pain scores of 383 patients who underwent IVI for DRP, RVO or AMD in our clinic between 1 April and 30 June 2023. Prior to participation, each individual in the study read and comprehended the informed consent form, providing their consent voluntarily. All patients were 18 years and older. All patients underwent detailed eye examinations, including visual acuity and intraocular pressure (IOP) measurements. Injection indications were determined by our clinic’s specialist (TS) after dilated fundus examinations and spectral domain optical coupler tomography (Heidelberg Engineering, Heidelberg, Germany) imaging. Patient’s pain was evaluated with a visual analogue scale (VAS). 9 Written informed consent was obtained from each participant before the study. Patients were categorized into three groups based on their diagnosis: DRP, RVO and AMD. Bevacizumab, ranibizumab and aflibercept were applied as anti-VEGF agents. To ensure consistency in pain assessments among patients, all injections were administered by the same doctor (KE) using a 27-gauge needle from the superior temporal quadrant. The researcher conducting the study was blinded to all study information except for the injected drug. Before the injection, all patients received 0.5% proparacaine hydrochloride (Alcaine; Alcon, Fort Worth, TX, USA) as a topical anaesthetic. Eye and periocular cleansing were performed with povidone-iodine, and a sterile drape was applied before the injection. Ciprofloxacin 0.3% ophthalmic solution (Ciloxan; Alcon) was instilled before and after the injection. Immediately after the injection, three researchers (BA, KC and EA) asked patients to rate their pain on a VAS from 0 (no pain) to 10 (severe pain). Patients were questioned about the presence of joint rheumatism or joint pain that could cause pain. Additionally, complaints of headaches were investigated, and the monthly amount of oral analgesics used for these pains was recorded. Patients were asked if they visited psychiatric clinics and about the psychiatric medications (antidepressants) they used. Surgical history, previous cataract surgery history, and history of vasovagal syncope during previous interventional procedures were also documented in the patients’ files.
Inclusion criteria were patients with intravitreal anti-VEGF indications (diabetic retinopathy, RVO, AMD). Exclusion criteria were patients who received anti-VEGF treatment for an indication other than the above-mentioned diagnostic criteria, previous vitreoretinal and glaucoma surgery, ocular trauma history and previous argon laser treatment for diabetic retinopathy.
Statistical analysis
The data analysis was conducted using Statistical Package for the Social Sciences (SPSS) software (v23.0 Windows; SPSS Inc., Chicago, IL, USA). For normally distributed qualitative data, the independent sample t-test was used, while non-normally distributed data were compared using the Mann–Whitney U test. Spearman’s rho correlation analysis was performed to explore the relationships between different parameters. Furthermore, regression analyses were carried out to evaluate the correlation between VAS score and variables such as the average number of headaches per month, average number of analgesic drugs used per month, previous injection number, and age. The threshold for statistical significance was set at p < 0.05.
When the studies were examined, a power analysis was performed by considering the VAS score, which is one of the main results of the study. Taking into account a similar study, the sample size was calculated taking into account the Type I error (0.05) and effect size d (0.50) targeted power (0.95) and it was concluded that the total sample size should be at least 210 people.
Results
The mean age of the 313 participants in the study (168 F, 145 M) was 66.91 ± 9.67 years. The average ages of the female and male patients were 66.64 ± 9.16 and 65.90 ± 10.13 years, respectively, with no significant difference between the groups (p > 0.05). Among them, 212 patients received injections due to DRP, 20 due to RVO, and 81 due to AMD. The distribution of injections included 261 patients treated with bevacizumab, 14 with ranibizumab, and 38 with aflibercept. The mean ages of the groups according to the diagnosis and injections administered are given in Table 1.
Table of mean age and standard deviation according to diagnosis and injections.

AMD, age-related macula degeneration; DRP, diabetic retinopathy; RVO, retinal vein occlusion.
The mean VAS score was 4.77 ± 2.90. The VAS scores for females and males were 4.69 ± 2.76 and 4.87 ± 3.07, respectively, with no significant difference between the groups (p > 0.05). When VAS scores were evaluated according to diagnosis, the VAS scores for DRP and RVO were 4.95 ± 2.98 and 5.22 ± 2.70, respectively, with no significant difference between the two groups. However, the average VAS score for the AMD group was 4.09 ± 2.64, which was significantly lower than that for the other two groups (Figure 1). There was no significant correlation between age and VAS score (r = −0.13, p = 0.74). Some clinical characteristics of the patients are shown in Table 2 and mean VAS scores according to clinical characteristics are shown in Table 3.
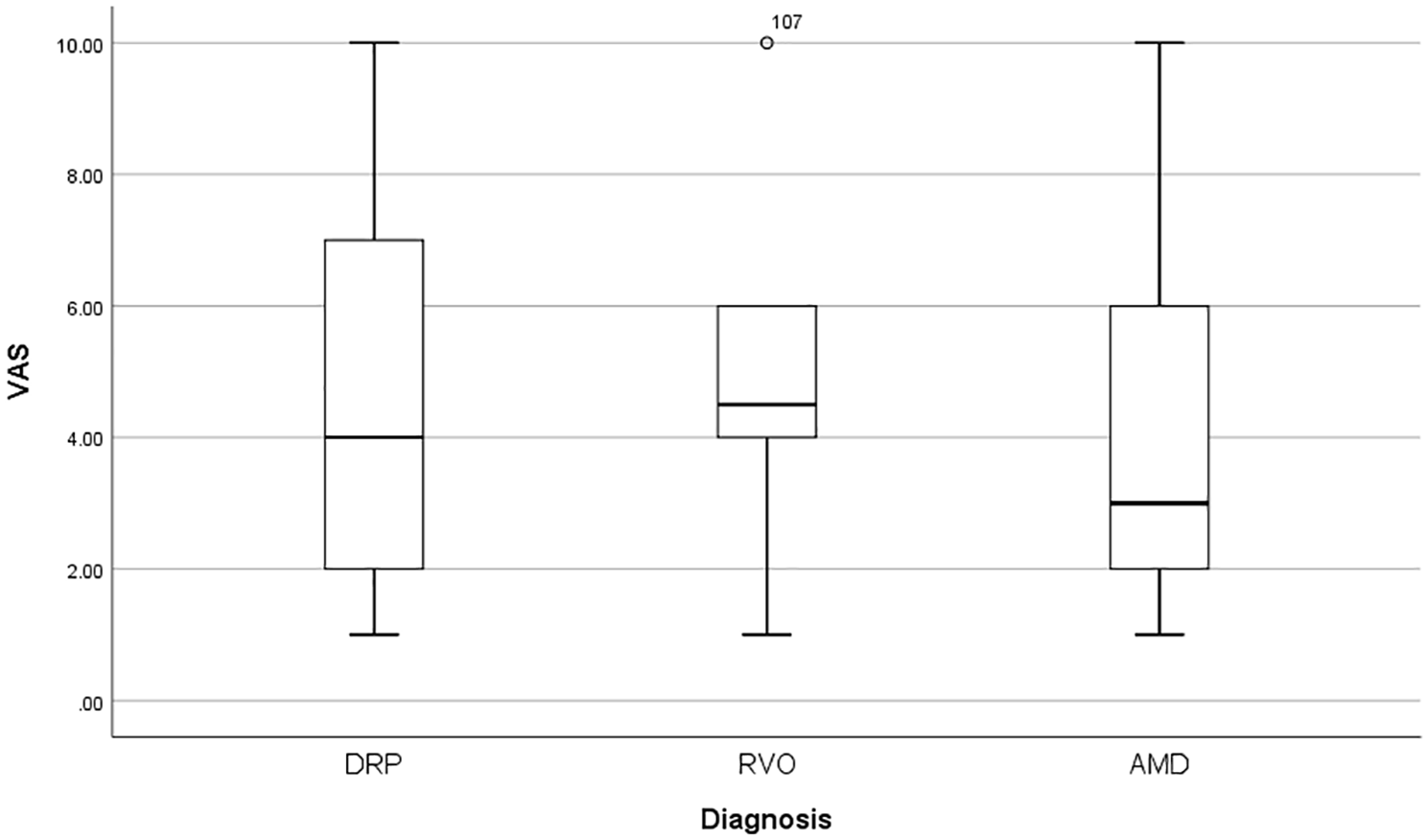
Distribution of VAS scores according to diagnosis groups.
Clinical characteristics of the patients who participated in the study.

VAS, visual analogue score.
Mean VAS score according to clinical features.
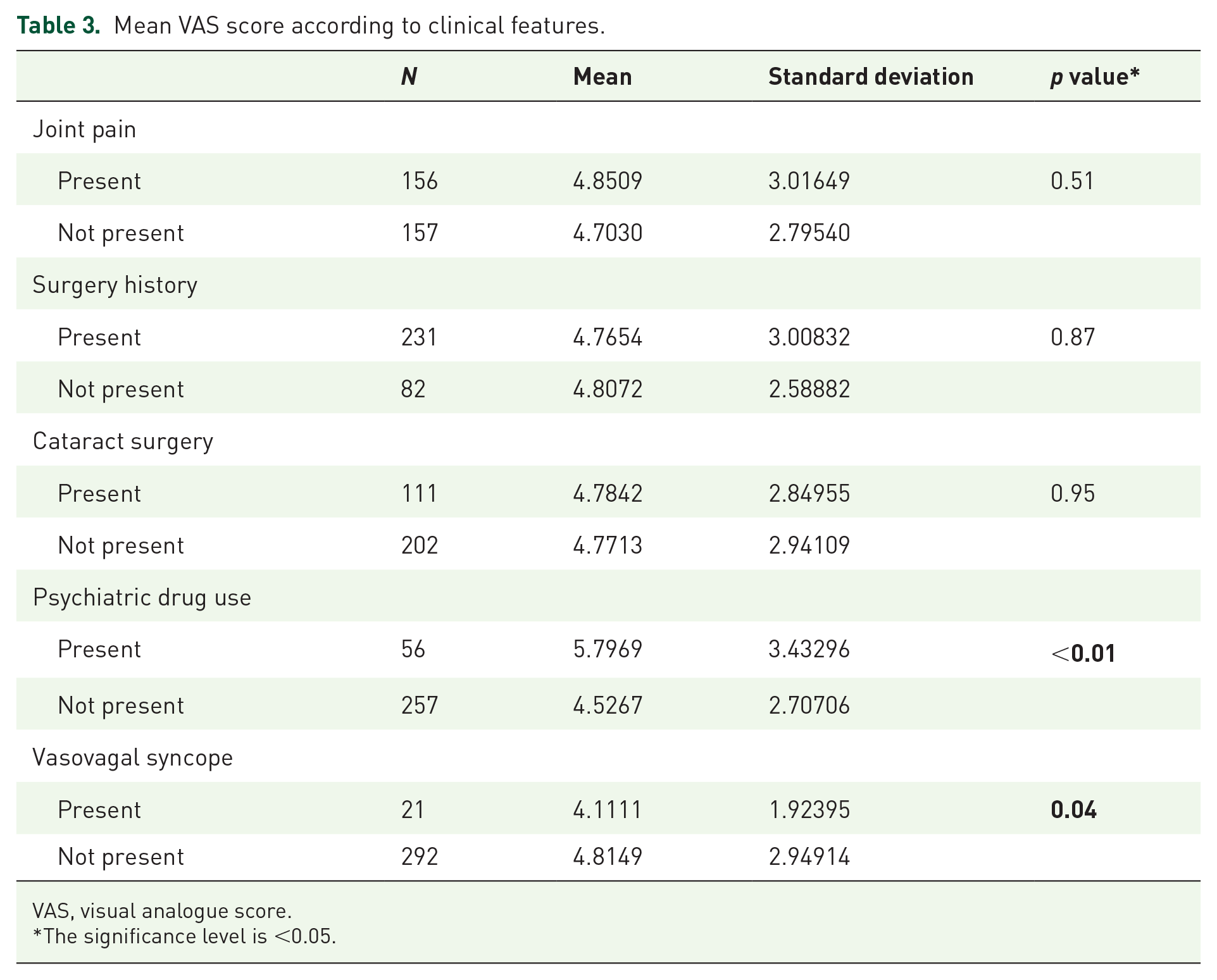
VAS, visual analogue score.
The significance level is <0.05.
In the group that experienced monthly average headache complaints, a positive correlation was found between headache frequency and VAS score (r = 0.23, p < 0.01) (Figure 2). Additionally, there was no correlation between the frequency of monthly analgesic use or injection count and the VAS score (r = 0.23, p > 0.05; r = 0.46, p > 0.05, respectively).

Correlation graph between monthly average headache count and VAS.
Discussion
Interventional procedures are widely employed for IVIs in contemporary eye clinics. Notably, these agents are used to treat macular oedema due to diabetes and RVOs, as well as in AMDs, employing anti-VEGF therapies. Furthermore, the administration of intravitreal agents is a common route for the treatment of other posterior segment diseases. In the United States alone, 7 million patients receive IVIs annually. 7 This number is expected to increase further due to the increasing prevalence of diseases such as diabetes and hypertension and the growing geriatric population. Patients receiving IVIs often require numerous injections, making the management of their fear and anxiety crucial for treatment adherence. The fear and anxiety experienced by patients, especially concerning adherence, are more challenging when conveyed to other patients. Additionally, patient anxiety during the procedure can lead to complications such as endophthalmitis, retinal detachment, or traumatic cataracts. 5
To ensure a comfortable and reliable IVI procedure for the patient, many clinics use topical local anaesthetic agents preoperatively. Numerous studies have been conducted to determine the effectiveness of various local anaesthetic agents. In a study by Blaha et al. involving 24 patients, no significant differences were observed in pain scores among patients treated with four different topical anaesthetic agents. 10 Another group of patients in the study received subconjunctival 2% lidocaine in addition to topical 0.5% proparacaine, but no significant difference was detected in terms of pain scores. In contrast, Andrade et al. reported that pain was more effectively controlled in patients receiving subconjunctival 2% lidocaine than in patients in the other groups. 11 Yau et al. did not observe significant differences in pain scores among patients treated with three different topical anaesthetic agents. 12 In this study, a group of patients receiving 4% cocaine and 1/100,000 epinephrine drops did not show any additional advantages compared to the other groups. Örnek et al. reported that 0.5% proparacaine was associated with lower pain scores than was 0.75% levobupivacaine. 13 Kumar et al. did not find statistically significant differences between 0.5% proxymetacaine and subconjunctival 2% lidocaine. 14 Davis et al. did not find a significant difference between topical proparacaine and 3.5% lidocaine gel. 15 However, Shiroma et al. observed a correlation between increasing lidocaine gel concentrations (ranging from 2% to 12%) and increasing anaesthetic efficacy. 16 There are also studies showing that combined preparations containing tetracaine and proparacaine do not provide an additional advantage in terms of pain. The varying results of numerous studies evaluating pain during intravitreal procedures may be indicative of differences in pain assessment and pain threshold parameters among patients.
Even slight variations during the procedure can lead to patients experiencing pain of varying intensity. Therefore, previous studies have evaluated whether there is a relationship between needle thickness and pain. Among the initial studies on this subject, no significant difference was found in VAS scores between patients treated with 22 gauge dexamethasone implants (Ozurdex) and patients treated with 29 gauge intravitreal bevacizumab injections. 17 Similarly, Ertan et al. did not detect a difference in pain scores between 30 gauge aflibercept and ranibizumab injections or 22 gauge dexamethasone implants. 18 However, Güler et al. observed a statistically significant difference in favour of 30 gauge in terms of the VAS pain score between patients receiving intravitreal ranibizumab injections performed with 27 and 30 gauge needles. 19 In a similar study, Haas and colleagues reported that there was no significant difference in VAS pain scores between 27- and 30-gauge needles. 8 Loureiro et al., while comparing 27 and 30 gauge injections, also evaluated IOP before and after injection. This study revealed that as the needle diameter decreased, the increase in postinjection IOP significantly increased. 20 However, unlike in previous studies, no significant difference in VAS score was detected between the two groups. This finding suggested that pain might be due to reasons other than an increase in IOP. Additionally, it is believed that a larger needle diameter is associated with less postinjection reflux, leading to a lower increase in IOP.
Another parameter evaluated for IVI and pain is the quadrant and location of the injection. Moisseiev et al. reported that injections via the lower left quadrant of the right or left eye resulted in lower VAS scores. 21 However, it is important to note that in this study, injections were specifically administered in this region. Proximity to the nasal area might be considered somewhat challenging for practical application. Karimi et al., in another study with 1004 patients, observed that injections from the superior nasal region caused a lower VAS score than injections from other areas. 22 Additionally, in this study, factors such as sex, number of injections, indication for injection, history of cataract surgery, and vitreous reflux were not found to be associated with pain. These results are consistent with our study, where a history of extraocular surgery, cataract surgery or previous injections were not associated with the VAS score. Additionally, individuals with a history of vasovagal syncope during previous procedures were questioned for the first time, and the relationship of these findings with the VAS score was investigated. Significantly higher VAS scores were detected in patients who had previously experienced vasovagal syncope. It should be noted that in this context, the pain that could lead to vasovagal syncope, especially during intramuscular or intravenous procedures, was relieved without an additional local anaesthetic procedure. However, despite the application of topical anaesthetic agents in intravitreal procedures, patients with a history of vasovagal syncope may be better prepared for preprocedural pain.
One of the reasons for patients experiencing pain and anxiety during the procedure is considered to be the use of eyelid specula and retractors. When comparing the pain scores of patients receiving injections with those receiving speculum-free or cotton-tipped applicators, it was observed that patients receiving procedures without specula or with applicators experienced less pain.23,24 However, in such cases, it might be challenging to apply the procedure to all patients due to the possibility of involuntary movement of the eyelids and increased susceptibility to potential complications. Additionally, the experience and skill of the person performing the procedure in this regard can be crucial. In our study, all the factors, including the anaesthetic used before the injection, the quadrant where the procedure was performed, and the location of the speculum, were planned to be the same under standardized conditions. Therefore, all patients underwent a standard procedure administered by the same doctor, with a 26-gauge needle applied from the superior temporal quadrant. Topical 0.5% proparacaine was applied before the procedure.
Numerous studies have been conducted on patients’ pain experiences, the way they evaluate pain, and its severity. Many parameters, such as a patient’s previous experience with pain and invasive procedures and surgeries, play a crucial role in pain assessment. This study particularly aimed to focus on other factors influencing patients’ perceptions of pain. Two significant parameters that were not evaluated in previous studies were identified in our study: the relationship between other pain-causing pathologies in the injection-receiving patient group and how patients assess these pathologies with the VAS score. Another aspect is how the VAS scores during IVIs compare patients who use antidepressants and those who do not. Considering the average age of the patients in our study, it was hypothesized that the two pathologies causing the most pain could be headaches and joint and muscle pains. Therefore, parameters such as the monthly frequency of headache, the presence of joint and muscle rheumatism and pains, the average number of analgesics used monthly due to experienced pains, previous history of nonocular surgeries, the number of previous injections, cataract surgery under local anaesthesia, history of vasovagal syncope during an invasive procedure, and the use of antidepressants prescribed by psychiatrists due to anxiety were examined. First, a positive correlation was found between the monthly frequency of headaches in patients with headache complaints and the VAS score. However, there was no significant difference in the VAS score between the group with joint or muscle pain and the group without this pain. There was no correlation between the average number of analgesics used by the patients due to their pain and the VAS score. The higher VAS scores observed in individuals who had previously experienced vasovagal syncope may be attributed to anxiety related to prior invasive procedures. Additionally, studies have shown that the use of topical analgesics, even oral analgesics, in addition to topical anaesthetics before injection contributes to lower VAS scores. A study by Makri et al. showed that patients who used oral and 0.1% topical diclofenac 4 hours before the injection and 45 min before the injection had lower VAS scores. 25 This effect has also been demonstrated with other active ingredient analgesics, such as nepafenac and ketorolac.26,27 However, the use of analgesics before the procedure rather than the frequency of use may be more effective. Furthermore, our study showed that patients who used antidepressants due to anxiety had higher VAS scores. These data could be an essential question during preinjection preparation to assess the pain a patient may experience in indicated cases. However, additional extensive studies with larger patient groups exploring the subcategories of antidepressant drugs are needed. This situation could be associated with individuals using antidepressants being more psychologically sensitive to pain and having higher levels of anxiety.
In our study, there was no statistically significant difference in VAS score according to sex. There are studies with different results on this matter. While Rifkin et al. mentioned that males had higher VAS scores after IVI, Karimi et al. and Ertan et al. reported in two separate studies that the VAS score was greater in the female patient group.18,22,28 In line with our study, Moisseiev et al. reported that sex was not associated with the VAS score. 21 Moreover, in our study, when the correlation between age and VAS score was examined, no statistically significant correlation was found. However, Makri et al. reported lower VAS scores in elderly patients. 26 Additionally, Ertan et al. reported a negative correlation between VAS score and age in their study, although they did not find differences in injection indications or agents used. 18 Despite the lack of correlation in our study, it should be noted that relatively younger patients with certain indications may experience more anxiety and, consequently, more pain.
Our study has several limitations. First, studies with a much larger number of patients are needed to assess the VAS score for IVIs, which are commonly used in daily practice. Additionally, our study lacked an evaluation of patients who used antidepressant medication from psychiatric clinics in terms of which type of medication they used in the subgroups. Therefore, if there is such an effect, the effectiveness of each class needs to be evaluated with a larger number of patients. In addition, double-blind VAS score evaluation with a larger number of patients to reduce possible biases in self-reported pain measurements would be more effective for future studies. Another limitation of our study is the lack of evaluation of vitreous reflux and IOP increase. However, although previous studies have detected an increase in IOP with thinner needles, no significant effect of vitreous reflux or an increase in IOP on the VAS score has been observed. Another limitation of the study is the lack of any validation of the survey questions. In the present study, despite the lack of correlation between the frequency of oral analgesic use and VAS score among patients receiving oral analgesics, subgroup analysis was not performed to determine which class of analgesic could be effective in long-term use. Additionally, to fully understand the effectiveness of the analgesic, it is necessary to inquire about when the patient last took oral analgesics. In the future, more extensive studies examining subgroups will be useful.
In conclusion, since intravitreal procedures are frequently performed in daily practice, the experience of pain and anxiety by patients during the procedure can affect the comfort of the procedure and, furthermore, treatment adherence. It is believed that both the factors affecting the procedure and the patient’s ability to assess pain, along with the related precautions, are crucial for treatment continuity. Additionally, there might be a relationship between a patient’s history of antidepressant drug use and VAS score, but further studies are needed to elucidate this relationship.
