Abstract
Background:
Diabetic macular edema (DME) is the most common cause of visual deterioration in patients with diabetes mellitus. Various treatment options have been used for DME, including intravitreal injection of steroids and anti-vascular endothelial growth factors.
Objectives:
To evaluate and compare the functional and anatomical outcomes of intravitreal ranibizumab (IVR) and intravitreal aflibercept (IVA) treatments in patients with DME.
Design:
Retrospective study.
Methods:
Four hundred three eyes of 235 naïve patients who underwent IVR or IVA treatment for DME followed up to 36 months included in the study. Best corrected visual acuity (BCVA) and central macular thickness (CMT) were measured at baseline, year 1, 2 and 3. Primary endpoint of the study was the change in BCVA and CMT each year from baseline and requirement of additional treatment (laser/steroid injection).
Results:
There were 198 eyes in IVR group and 205 eyes in IVA group. The changes in mean BCVA were 0.09 ± 0.32 versus 0.17 ± 0.41 Logarithm of the minimum angle of resolution (logMAR) (p = 0.042) at year 1, 0.09 ± 0.37 versus 0.12 ± 0.45 logMAR (p = 0.512) at year 2 and 0.13 ± 0.36 versus 0.15 ± 0.48 logMAR (p = 0.824) at year 3 in IVA and IVR groups, respectively. The baseline mean BCVA were lower (p = 0.004) in IVA group. The mean total number of injections was 7.93 ± 3.38 versus 7.42 ± 3.05 (p = 0.112).
Conclusion:
At year 1, change in mean BCVA was statistically significantly higher in IVA group; however this difference did not persist at years 2 and 3. Although the mean total number of injections was similar between groups, the requirement for adjuvant steroid treatment was significantly higher in ranibizumab group, which may affect the number of visits and treatment costs. Both ranibizumab and aflibercept treatments achieved a good long-term visual and anatomical response in DME patients.
Introduction
The most common cause of visual deterioration in patients with diabetes mellitus is diabetic macular edema (DME) 1 with an estimated prevalence of 7.5% in population with diabetes. 2 In the treatment of DME, various options have been used. Intravitreal injection of steroids and anti-vascular endothelial growth factors (VEGF) are the most favored treatment methods nowadays. 3
The VEGF has a primary role in the pathogenesis of DME. 4 Increase in VEGF causes breakdown of the inner blood-retinal barrier and leakage which leads to DME and visual impairment.5,6 The anatomical and functional success of intravitreal anti-VEGF treatment has been shown in the major randomized clinical trials (RCT).4,7 Ranibizumab is a monoclonal antibody fragment that suppresses angiogenesis by inhibiting VEGF-A). 8 Intravitreal ranibizumab (IVR) improves vision depending on varying treatment protocols.9,10 On the other hand, Aflibercept is a recombinant fusion protein that binds to VEGFs and acts like a ‘VEGF trap’. Hence it inhibits the activity of VEGF-A, VEGF-B and placental growth factor. 11 It has been shown in previous studies that patients with DME treated with aflibercept gain a remarkable improvement in functional and anatomical outcomes.12,13
Injections of corticosteroids are other alternative pharmacological treatment method available for DME. In the pathogenesis of DME, there are many proinflammatory cytokines besides VEGF. 14 Blocking the inflammatory response, corticosteroids may indirectly reduce VEGF production. 15 Corticosteroids could be chosen as first-line treatment in patients with recent history of cardiovascular event, in pregnancy and DME in eyes undergoing cataract surgery. 16
It may not be easy to follow-up the tight schedule and treatment protocols recommended by prospective studies in real life practice. Studies around the world have shown that the real-life conditions in regard to the injection and visit numbers was away from the landmark prospective studies.17,18 Still, we need to find an agreement that yields this effective therapy in an appropriate and pragmatic way for the patients. In this study, we aimed to compare the long-term efficacy and outcomes of IVR and aflibercept treatment for naive DME patients in a real life setting in a tertiary referral center.
Patients and methods
This retrospective, single-center study which included up to 36-month observation of treatment-naive DME patients without proliferative diabetic retinopathy was conducted at a tertiary eye care center in Istanbul, Turkey.
The clinical records of the patients who had DME and underwent IVR or intravitreal aflibercept (IVA) treatment between January 2016 and 2019 were analyzed. The patients ⩾18 years old with type 1 or 2 diabetes, who had naive centrally involved DME with at least 12 months of clinical follow-up were included in the study. Pseudophakic patients were included if cataract surgery was performed at least 3 months ago. Patients who were treated with previous intravitreal or subtenon injections, panretinal or focal/grid laser or pars plana vitrectomy were excluded. Any eyes switched from ranibizumab to aflibercept or vice versa were also excluded. Other exclusion criteria contain proliferative diabetic retinopathy, ischemic maculopathy, macular edema due to other causes, another vitreous or retinal disease.
At each visit, all patients had a standard ophthalmologic examination including best corrected visual acuity (BCVA) with Snellen chart, measurement of intraocular pressure (IOP) by Goldmann applanation tonometer, evaluation of anterior segment and non-contact dilated fundus examination with slit-lamp biomicroscopy. The measurement of central macular thickness (CMT) was taken with spectral-domain optical coherence tomography (SD-OCT) (Spectralis, Heidelberg Engineering, Heidelberg, Germany) at each visit. CMT which is described as the average thickness of the retina 1 mm around the center of the fovea, was measured by mapping software of the SD-OCT. Baseline fundus fluorescein angiography (FFA) (Spectralis, Heidelberg Engineering, Heidelberg, Germany) was performed to exclude proliferative diabetic retinopathy and ischemia of macula for all patients before the intravitreal injections and repeated according to clinicians’ decision. DME was defined by a CMT⩾ 250 μm.
Every injection was performed under sterile conditions following administration of topical anesthesia, disinfection with 10% povidone-iodine on periorbital skin, eyelids and eyelashes, and 5% povidone-iodine on the conjunctival surface. IVA 2 mg/0.05 ml (Eylea; Bayer, Basel, Switzerland) and IVR 0.5 mg/0.05 ml (Lucentis; Novartis, Basel, Switzerland) were injected through the pars plana at 3.5 mm posterior to the limbus in pseudophakic patients and 4 mm posterior to the limbus in phakic patients.
In our routine clinical practice, each treatment was started with three intravitreal injections as loading dose, followed by the pro re nata regimen and monthly follow-up. The clinician decided the type of anti-VEGF drug. In refractory DME in addition to monthly treatment, macular laser and/or subtenon steroid injection (triamcinolone acetate) were added as ancillary to IVA/IVR treatment. After three loading doses, if the CMT did not decrease from the baseline and leaky microaneurysms in central 1000 μm area were detected in FFA, macular laser treatment was added. After three doses of anti-VEGF drugs, if the CMT increases or decreases less than 50 μm from the baseline and serious retinal detachment develops, subtenon steroid injection was added. The number of injections of the drugs per year and whether the ancillary macular laser applied or not was recorded.
Primary endpoint of the study was the change in BCVA and CMT each year from baseline and requirement of additional treatment (laser/steroid injection). Secondary endpoints were the number of intravitreal injections delivered per year, BCVA and CMT values each year, and visual gain and loss percentages.
Statistical analysis was performed using the software SPSS for Windows (version 22.0, IBM Corp.). BCVA measurement were transformed to Early Treatment Diabetic Retinopathy Study letters and logarithm of minimum angle of resolution (LogMAR) for statistical analysis. Continuous variables are reported as the mean and standard deviation. The Kolmogorov-Smirnov test was used to assess the normality of the data. The Student-T test was used to compare groups for the statistical analysis, chi-square test was used for categorical data. Bonferroni correction was used for confidence interval adjustment, and a repeated-measures analysis of variance (ANOVA) was used for repeated measurements. Pearson correlation coefficient was used for BCVA and CMT changes. Differences were considered statistically significant when the p value was less than 0.05.
Results
A total of 235 patients’ 403 treatment naive eyes were enrolled in this study. There were 198 eyes in IVR group and 205 eyes in IVA group. The baseline characteristics of each group are shown in Table 1. No significant differences were appreciated in terms of sex and laterality. The mean age of patients treated with IVA (57.53 ± 9.31) were significantly younger than those treated with IVR (60.34 ± 8.93) (p = 0.002). Baseline mean BCVA of IVA group (0.66 ± 0.45 logMAR, 52.2 letters) was significantly lower than IVR group (0.54 ± 0.40 logMAR, 58.3 letters, p = 0.004). There was no significant difference in baseline CMT measurements (435.57 ± 146.26 versus 440.58 ± 131.11 µm, p = 0.717) between groups. The mean BCVA and CMT values of eyes each year are shown in Figure 1. The mean follow-up time was significantly longer in IVR group than IVA group (29.02 ± 8.93 versus 26.26 ± 9.57 months, p = 0.030). The mean number of injections was 4.98 ± 1.29 versus 5.32 ± 1.70 (p = 0.023) in first year, 2.14 ± 1.67 versus 2.24 ± 1.75 (p = 0.621) in second year, and 2.30 ± 1.74 versus 1.70 ± 1.42 (p = 0.020) in third year in IVR and IVA groups, respectively. The mean total number of injections was 7.93 ± 3.38 versus 7.42 ± 3.05 (p = 0.112) and the mean ratio of the number of injections to follow-up period was 0.29 ± 0.11 versus 0.31 ± 0.12 (p = 0.087) per month, during whole follow-up period in IVR and IVA groups, respectively.
The baseline demographic and clinical data of the patients.

Student T test.
Chi-square test.
BCVA, Best corrected visual acuity; CMT, Centreal macular thickness; LogMAR, Logarithm of the minimum angle of resolution.
Bold values indicate p < 0.05 significant.

The mean BCVA and CMT in IVR and IVA groups at baseline, years 1, 2 and 3.
In terms of BCVA changes, both groups resulted in significant improvement in each year of treatment compared to the baseline (Table 2). There was no significant difference in the improvement between groups except the first year. The mean BCVA difference in first year in IVA group (0.17 ± 0.41 logMAR, 8.2 letters) was significantly higher than IVR group (0.09 ± 0.32 logMAR, 4.5 letters, p = 0.042). At 2nd and 3rd years, changes in BCVA were not significant (p = 0.512 for 2nd year and p = 0.824 for 3rd year) between groups. At last visit, the change in mean BCVA was 0.12 ± 0.35 LogMAR (5.9 letters) in IVR group and 0.13 ± 0.43 LogMAR (6.6 letters) in IVA group (p = 0.717). In eyes with baseline BCVA equal to or higher than 20/50 Snellen, there was no statistically significant difference in baseline (0.26 ± 0.11 versus 0.24 ± 0.14, p = 0.427), year 1 (0.27 ± 0.13 versus 0.22 ± 0.11, p = 0.298), year 2 (0.25 ± 0.10 versus 0.24 ± 0.12, p = 0.306) and year 3 (0.24 ± 0.14 versus 0.25 ± 0.09, p = 0.222) BCVA in IVR and IVA groups, respectively.
Changes in mean BCVA (LogMAR) values of patients each year.

Student T test.
BCVA, Best corrected visual acuity; IVA, Intravitreal aflibercept; IVR, Intravitreal ranibizumab; LogMAR, Logarithm of the minimum angle of resolution; N, Number of eyes.
Bold values indicate p < 0.05 significant.
In terms of CMT changes, both groups resulted in significant improvement in 1st, 2nd and 3rd years compared with the baseline values (p < 0.001 for each). There was no statistically significant difference between groups (p = 0.270 for 1st year, p = 0.841 for 2nd year, p = 0.883 for 3rd year) (Table 3). At last visit, the change in mean CMT was −89.28 ± 157.27 µm in IVR group and −95.58 ± 163.90 µm in IVA group (p = 0.694). In correlation analysis, changes in BCVA and CMT were significantly correlated in IVR and IVA groups at year 1 (p < 0.01 versus p < 0.01), 2 (p < 0.01 versus p < 0.01) and 3 (p = 0.06 versus p < 0.01), respectively.
Changes in mean CMT values of patients each year.

Student T test.
CMT, Centreal macular thickness; IVA, Intravitreal aflibercept; IVR, Intravitreal ranibizumab; N, Number of eyes.
At last visit, 32.8% of patients in IVR group and 38.5% of patients in IVA gained 10 letters or more in BCVA (p = 0.232), 25.3% of patients in IVR group and 31.2% of patients in IVA group gained 15 letters or more in BCVA (p = 0.184), versus baseline (Figure 2). Also, 12.6% of patients in IVR group and 19.5% of patients in IVA lost 10 letters or more in BCVA, 6.6% of patients in IVR group and 8.8% of patients in IVA group lost 15 letters or more in BCVA. At last visit, there was no significant difference between groups in terms of letter gain (p = 0.741).

Percentages of BCVA gain and loss of treatment groups.
During the follow up time, 37.4% of eyes in IVR group and 36.2% of eyes in IVA group underwent focal laser therapy (p = 0.812). In addition, 47.5% of eyes in IVR group and 34.6% of eyes in IVA group were delivered steroid injections, hence the eyes in IVR group were statistically significantly received more steroid injections (p = 0.009). The mean number of steroid injections needed during the 3 years follow-up was 0.88 ± 1.12 and 0.56 ± 0.97 (p = 0.002) in IVR and IVA groups, respectively. Moreover, 20.7% of eyes in IVR group and 14.6% of eyes in IVA group were administered both laser and steroid therapy (p = 0.082). IOP was 14.6 ± 2.4, 15.3 ± 2.7, 14.9 ± 3.1, 15.8 ± 3.5 mmHg (p = 0.157) at baseline, year 1, 2 and 3, respectively. No serious ocular complications were reported like endophthalmitis or retinal detachment.
In multivariate regression analysis (Table 4), while variables in the study were not significantly associated with final BCVA, steroid injections and focal laser therapy were significantly associated with final CMT in IVR (p = 0.04) and IVA (p = 0.02) groups, respectively.
Multivariate regression for the final BCVA and CMT of both groups.
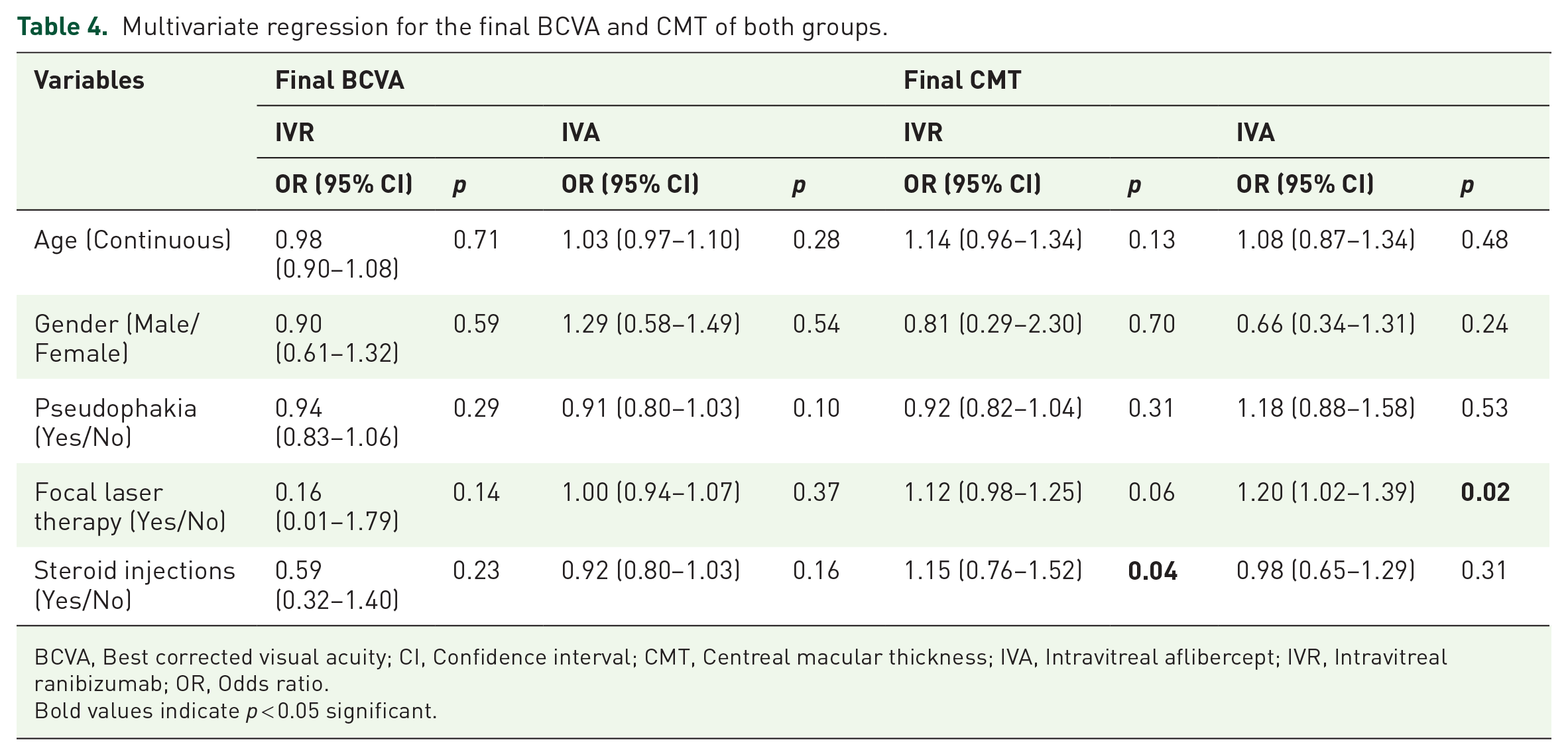
BCVA, Best corrected visual acuity; CI, Confidence interval; CMT, Centreal macular thickness; IVA, Intravitreal aflibercept; IVR, Intravitreal ranibizumab; OR, Odds ratio.
Bold values indicate p < 0.05 significant.
Discussion
The increase in retinal vascular permeability which drives the accumulation of liquid within the layers of retina and increases its thickness results in DME. It is associated with the disruption of the blood-retinal barrier and increase in the levels of VEGF. 19 The DME treatment has been revolutionized by the progress in anti-VEGF injections, with a well-documented recovery in BCVA and a reduction of CMT, was confirmed by several RCTs.20,21 Nevertheless, there are only a few studies in real-life setting comparing IVR and IVA in DME patients, while most of them have included low number of patients.22–24
This retrospective study shows long-term visual and anatomical outcomes in patients with sight-impairing, center-involving DME treated with IVR or IVA up to 3 years in routine clinical practice in a tertiary eye center. The visual and anatomical functions improved at year 1 and maintained stability at years 2 and 3 in both groups, compared to the baseline. The amount of BCVA improvement between the groups was similar except at first year. At year 1, BCVA recovery was significantly superior (p = 0.042) in IVA group. The change in CMT was also similar between the groups.
Protocol T is a RCT which compares IVA and IVR in DME patients. Based on the results of protocol T, IVA seemed to be more efficient in BCVA improvement, especially in eyes with BCVA between 20/50 and 20/320 at first year. 25 However, there was no difference in BCVA improvement between two drugs at second and fifth years.4,26 In our study, we also measured BCVA differences between IVR and IVA groups at 1st and 2nd years, similar to the Protocol T. However, there were some major differences. First of all, in our study, the baseline mean BCVA of IVA group was significantly lower than IVR group (52.2 letters versus 58.3 letters, p = 0.04), which is probably the most important factor to explain the difference. In Protocol T, the median baseline BCVA were 68 and 69 letters in IVR and IVA groups, respectively. The mean number of injections in IVA group was significantly higher than IVR group (5.32 versus 4.98, p = 0.023) which may also be associated with the BCVA difference. In Protocol T, the median number of injections were 10 and 9 in a monthly period in IVR and IVA groups, respectively. Furthermore, over 2 years, 52% and 41% of eyes treated with IVR and IVA, respectively, received focal/grid laser (p < 0.01). Unlike our study, the macular laser treatment rates were higher and there was a statistically significant difference between two groups.
In general, the number of injections in real-life studies remains to be lower than that of RCTs. In our study, the mean number of injections received in first year was 5.2 similar to other real-life studies in DME patients (between 3.1 and 7.2 in first year).17,27,28 However, in RCTs, injection numbers were higher (mostly 8–10 in first year).20,29,30 After the 1st year, the number of injections was reduced which may be related to disease modifying properties of anti-VEGF treatment in DME patients. 31
In protocol T, the mean BCVA changes was 11.2 versus 13.3 letters at year 1, 12.3 versus 12.8 letters at year 2 in IVR and IVA groups, respectively. 4 In RESTORE-RESOLVE studies and Protocol I, visual improvement after IVR at year 1 were 6.1, 10.3 and 9 letters, respectively.9,32,33 In RISE and RIDE studies, mean visual improvement after IVR at year 2 were 12.5 and 12 letters, respectively. 7 In VISTA and VIVID studies, improvement after IVA at year 1 were 12.5 and 10.7 letters, respectively. 21 In our real-life population, improvement in BCVA was less pronounced than in Protocol T and other prospective studies. The most probable reason for the difference is the higher frequency of injections reported by these studies, being up to 12 injections per year. Additionally, patient selection, strict adherence to follow-up periods, early intervention to complications may contribute to the difference.
Plaza-Ramos et al. 22 conducted a real-life study with 213 eyes (122 IVR versus 91 IVA) and there was no difference between groups at 12 months. Conversely to our study, the mean baseline BCVA values were lower in IVR group (0.55 versus 0.48 logMAR) but not statistically significant (p = 0.109). Change in mean BCVA at 1st year was 0.15 logMAR in IVR group and 0.08 logMAR in IVA group which are similar to our year 1 results, and naïve patient ratio were higher in IVR group (70% versus 26%) which may contribute to final BCVA because chronic DME patients who are previously treated with anti-VEGF therapy have a lower response to treatment. 34 Eleven eyes (9%) in IVR group and three eyes (3.3%) in IVA group were administered focal laser therapy (p = 0.096) which were not significantly different, but the frequency of laser therapy was much lower than our study (37.4% in IVR and 36.2% in IVA groups). In the real-life study of Shimizu et al. 23 with a similar concept, 49 eyes in IVR group and 46 eyes in IVA group are included and followed up for 6 months. The mean baseline BCVA was 0.48 logMAR in IVR group and 0.39 logMAR in IVA group. Change in mean BCVA at month 6 was 0.03 versus 0.09 logMAR in IVR and IVA groups, respectively and they reported that the effectiveness of IVA in improving the BCVA might be better than IVR. In the study of Bhandari et al. 24 which includes 303 treatment naïve eyes (136 IVR versus 167 IVA) of 228 patients with DME who completed 12 months follow-up period, authors found that both IVR and IVA were effective for DME over 12 months, with aflibercept having somewhat better anatomical outcomes (change in CMT was −126 µm versus −89 µm, p < 0.01). Larger BCVA gains were observed in IVA group when the initial BCVA was ⩽ 0.3 logMAR (change in BCVA was 10.6 letters versus 7.6 letters, p = 0.01). Also five eyes (3.7%) in IVR group and two eyes (1.2%) in IVA group underwent macular laser treatment (p = 0.24), furthermore seven eyes (5.1%) in IVR group and four eyes (2.4%) in IVA group received steroid injection. Frequency of these additional treatments was much lower than our study. In a recently published retrospective study 35 which includes 534 treatment naïve eyes (267 IVR versus 267 IVA), authors reported that both IVR and IVA were safe and effective over 3 years follow-up period similar to our study, but IVA group was better in anatomical outcomes. Also, visual gain at year 1 was higher in IVA group. Twenty-four (9%) eyes in IVR group and 12 (4.5%) eyes in IVA group (p = 0.058) received additional macular laser treatment which were less often than our study. And lastly like our study, the median number of injections at year 3 was 7 and 8 in IVR and IVA groups (p = 0.30), respectively.
The main limitation of our study is its retrospective design which are inherent in real world studies. The treated eyes were not divided to stages of non-proliferative diabetic retinopathy. The real-world population was heterogeneous. However, we believe that publishing real-world data is of benefit to the clinicians who treat DME patients in real-life because clinical trial settings are difficult to apply in routine clinical practice. Still, further prospective studies with a larger cohort and longer follow-up time may be required to better understand and compare the output of IVR and IVA for DME. To the best knowledge of the authors, the current study which compares ranibizumab and aflibercept efficiency in DME patients, includes one of the largest real-world populations in literature and have a satisfactory potential to reflect realistic routine clinical practice.
In conclusion, both ranibizumab and aflibercept treatments achieved a good long-term visual and anatomical response in DME patients except for the first year. Although the mean total number of injections was similar between groups, the requirement for adjuvant steroid treatment was significantly higher in ranibizumab group, which may affect the number of visits and treatment costs. In real-life setting, it may not be simple to comply with the strict follow-up and treatment protocols that were used in RCTs while treating DME. However, the clinical management of the patients should be optimized to have better outcomes in real-life.
