Abstract
The current data on various aspects of Brown syndrome are limited and sporadic. This review provides a coherent and comprehensive review of basic features, etiology, classification, differential diagnosis, and different management strategies of patients with Brown syndrome. In this topical review, PubMed, Scopus, and Google Scholar search engines were searched for papers, published between 1950 and January 2023 based on the keywords of this article. The related articles were collected, summarized, categorized, assessed, concluded, and presented. Brown syndrome is identified by restricted passive and active elevation of the eye in adduction. The condition is divided into congenital and acquired causes. The clinical features result from a restricted motion of the superior oblique tendon sheath through the trochlea while trying to look up in adduction. The newest explanation of the underlying pathophysiology has been explained as the presence of a fibrotic strand in the superior oblique muscle tendon with variable insertion sites which creates various elevation deficits seen in Brown syndrome. The most common clinical features include the presence of an abnormal head posture, V-pattern strabismus, and hypotropia in the primary position. Management of Brown syndrome includes watchful observation, surgical, and non-surgical procedures. Some cases might resolve spontaneously without any intervention; however, some acquired cases might require systemic and/or intra-trochlear steroid administration to treat the underlying causes. Surgical procedures such as superior oblique tenectomy and using a silicon tendon expander are indicated in the presence of hypotropia and significant abnormal head posture in the primary position.
Introduction
Brown syndrome is identified as an active and passive restricted elevation of the eye in adduction. 1 It was first introduced by Dr Harold Whaley Brown. In 1950, he characterized a syndrome with a restricted elevation of the influenced eye in adduction, drift downward in adduction, a ‘V’ pattern, and a positive forced duction test (FDT) for the passive elevation in adduction which he called Brown syndrome. 1 Primarily, it was thought that it occurred because of anomalies within the tissue surrounding the anterior superior oblique tendon, restricting the free motion of the tendon through the trochlea. Accordingly, it was named ‘superior oblique tendon sheath syndrome’. 2 Moreover, it was initially thought that the short tendon sheath occurred because of a congenital paresis of the ipsilateral inferior oblique muscle. 3 However, the clinical manifestation of the syndrome was not consistent with an inferior oblique paresis which usually shows an associated A pattern and superior oblique overaction. 4 Therefore, the hypothesis of inferior oblique paresis in Brown syndrome was discarded, as patients usually show a V pattern accompanied by minimal or no superior oblique over action. 5 Brown syndrome was first classified as true or simulated in the previous literature. True Brown syndrome refers to a congenital form with constant limitation in elevation in adduction caused by a tight superior oblique tendon. Simulated Brown syndrome, however, was used in cases with acquired history, intermittent cases, and cases with no evidence of superior oblique tendon pathology. 5 The definition of the ‘true’ or ‘simulated’ sheath syndromes was also disregarded when Parks and Brown did not find any tight or short anterior superior oblique tendon sheath in patients they assessed. 6 Therefore, the new definition of ‘Brown syndrome’ refers to an elevation deficit in adduction accompanied by a pathology in the superior oblique tendon/trochlea complex. Brown syndrome is a restrictive strabismus that can impact patients’ ocular alignment and binocularity. While it was first described over 70 years ago, the literature on various aspects of this condition remains limited and scattered. This review aims to consolidate the current knowledge of Brown syndrome into a single coherent source to aid clinicians. Specifically, this review provides a comprehensive overview of the basic features, etiology, classification, differential diagnosis, and management strategies for Brown syndrome based on the latest evidence. It discusses important topics such as the debated pathophysiology, inheritance patterns, prevalence data, associated clinical findings, as well as evidence for spontaneous recovery and surgical outcomes. By synthesizing knowledge from over 70 years of literature across multiple specialties, this review addresses current gaps and inconsistencies and aims to enhance practitioners’ understanding of this syndrome and help optimize patient evaluation and care.
Methods: In this review, PubMed, Scopus, and Google Scholar search engines were searched for papers published between 1950 and January 2023 based on the keywords of this article. The related articles were collected, summarized, categorized, assessed, concluded, and presented in this review. The figures presented were obtained from Farabi Eye Hospital, Iran and the subjects’ legally authorized representative consented to their photos to be shared in this article for academic publications.
Basic features
Clinical features of Brown syndrome have not changed significantly from what Brown initially described. The consistent features include limited or no elevation in adduction, mild elevation deficit in up gaze, normal or minimal deficit of elevation in abduction, positive FDT in elevation and adduction, and minimal or no superior oblique overaction (Figure 1). Various features may or may not be present in every patient. For instance, V pattern in up gaze, eyelid widening in adduction, abnormal head posture (AHP; chin up and contralateral face turn), the presence of hypotropia in the primary position, and down shoot in adduction.1,7 Vertical double vision is a less common clinical manifestation of the disorder which could happen temporarily and is frequently accompanied by a click feeling.8,9

A patient with left Brown syndrome with no hypotropia in primary position. There is no elevation in adduction in the left eye when eyes are looking up and right, with no significant overaction of the left superior oblique muscle when eyes are looking down to right. There is a mild deficit in looking at up gaze and while abduction.
Pathophysiology, inheritance, prevalence, and demographic information
Pathophysiology
There has been an ongoing controversy on the various underlying reasons for this syndrome.
Previous evidence suggests an anomaly within the trochlea and superior oblique tendon–muscle complex, whereas more recent evidence proposes anomalies in the development of the extraocular muscles nervous system such as congenital cranial dysinnervation disorders (CCDDs) as a possible reason in some cases. 10 The newest explanation of the pathophysiology, however, is the presence of a fibrotic strand at the superior oblique tendon.
As first explained by Brown, a tight or short superior oblique tendon sheath was discovered in many patients with Brown syndrome which may lead to mechanical restriction in the tendon, preventing the eye from moving up and inside.8,9 When the tight or immobile tendon was cut during the surgical intervention, FDT improved during surgery; although postoperative ductions did not improve. 2 According to Sevel’s extensive research on extraocular muscles, tendons and insertion, trochlea, superior oblique tendon, and muscle originate from a common mesenchymal tissue. As the eye develops, the superior oblique muscle, tendon, and trochlea gradually become identifiable. This may explain the gradual improvement seen in some congenital Brown cases. 11 It has been suggested that abnormal development of the fourth nerve and its axons may result in the poor differentiation of the superior oblique muscle–tendon–trochlea complex and may result in clinical features of Brown syndrome. 10 However, abnormal neurologic development arising from CCDDs cannot cover the full spectrum of Brown syndrome, especially with evidence of no limitation in depression while adduction. 12
The newest explanation emphasizes the fibrotic strand found in some forms of Brown syndrome during surgical intervention. The strand is located at the posterior part of the superior oblique tendon, originating from the trochlear region with the same insertion as the superior oblique tendon. 12 It is in accordance with what was reported by Fink in various animals who do not have a trochlea but whose superior oblique muscle originates from the anterior superior nasal orbit. This may explain the various forms of Brown syndrome, not only the typical forms but also atypical cases with different elevation anomalies because the fibrotic strand might have variable insertion sites which creates various elevation deficits seen in atypical Brown syndrome. 12
Inheritance
There is evidence of heredity’s influence on Brown syndrome.13,14 Monozygotic twins might be equally influenced14,15 or in some cases, just one of the twins is involved. 16 There also have been reports on dizygotic twins with Brown syndrome. 13 There have been instances of brothers and/or sisters in one family pedigree, with bilateral Brown syndrome. 17 In a case report study, all three siblings in one family had bilateral Brown syndrome, whereas the other family was uninfluenced. 18 Another study showed the occurrence of unilateral late-onset Brown syndrome in three siblings on the left side while no other family member was involved and parents were non-relatives. No abnormalities of the tendon–muscle–trochlea complex were observed. The inheritance pattern was reported to be either autosomal recessive or autosomal dominant with reduced penetrance. 19 Kenawy et al. 20 reported two siblings and their mother with left Brown syndrome with an autosomal dominant inheritance. The autosomal recessive and autosomal dominant patterns with reduced penetrance have been reported as transmission patterns in cases with a family history of the disease.13,17–20 As inheritance has a significant role in the transmission of Brown syndrome, genetic counseling of these patients to identify the responsible genes and investigating the risk of occurrence in posterity are recommended.
Prevalence
There is not much information on the prevalence of Brown syndrome and the data are sporadic. In one of the earliest reports, Brown syndrome was estimated to occur 1 in every 450 strabismus cases. 1 Another study that investigated the prevalence of various types of strabismus among 2217 strabismic patients between 2 and 56 years during 2014–2018, reported 18.03% of cases with Brown syndrome. 21 In another report with 226 strabismic cases between 4 months and 36 years, 6.2% of patients had restrictive strabismus including Brown syndrome. 22 Urist 23 showed that among vertical strabismus cases, 10.17% of cases had Brown syndrome with a positive FDT test. For familial Brown syndrome, the incidence is 1 in 20,000 live births. 1 Since many cases with Brown syndrome have no symptoms to visit a doctor, it is estimated that the real statistics might be higher than reported.
Demographic information
Gender
Several reports show a slight predominance of females with Brown syndrome.4,5,24,25 Brown reported 59% occurrence in females, whereas, in the Wright study it was 52%.4,5 In a recent article, demographic data from 190 cases with Brown syndrome were gathered. The authors reported 111 (59%) female cases. 25 Interestingly, in case report studies with the familial Brown syndrome, women with Brown syndrome are slightly more.14,19 The possible underlying reason for this predominance might be the predisposition of females for inflammatory diseases such as rheumatoid arthritis, Sjogren syndrome, scleritis, and lupus erythematosus.
Laterality
Studies have demonstrated that Brown syndrome is bilateral in about 10% of cases (Figure 2).1,5,25 In unilateral cases, Wright 4 found a right eye bias (55%) in 96 Brown syndrome patients evaluated. Khorrami-Nejad et al. 26 analyzed 100 Brown syndrome patients and found 60.4% of patients had right eye involvement which was significantly higher than the left eye. A familial syndrome case report by Iannaccone et al., 19 however, described three siblings all manifesting unilateral left-sided Brown syndrome, suggesting possible genetic factors influence laterality in some cases. Therefore, according to the evidence, there is a slight predominance of right-eye involvement in unilateral Brown syndrome. However, there are also reports of specific families exhibiting left-sided or bilateral presentation. Further research is still needed to better understand possible underlying factors on laterality.

Patient with bilateral Brown syndrome. The limitation in adduction while elevation is evident in both right and left gazes.
Brown syndrome classification
Brown syndrome is classified as congenital versus acquired based on the onset. However, in some cases, there is an uncertainty on the age of onset or the underlying pathology. These types of cases have been classified as idiopathic in studies. 27
Congenital Brown syndrome
Congenital Brown syndrome is present at birth and constant restriction in movement is not associated with any inflammation or pain. In most cases with congenital Brown syndrome, there is a tight superior oblique muscle–tendon complex that limits elevation of the eye in adduction. The presence of an inelastic muscle–tendon complex matches the congenital Brown syndrome. 4 However, recent evidence suggests that some congenital cases might be associated with CCDD as discussed earlier in the text. Studies have demonstrated evidence of spontaneous recovery in congenital cases.28–30 Therefore, conservative management in such cases is recommended.
Acquired Brown syndrome
In the acquired form, limitation in elevation while adduction occurs after infancy. The acquired form can be intermittent, with pain and a clicking sensation while moving the eye in the superior nasal position.4,5 Some acquired forms might resolve spontaneously without any treatment. According to Wright, 4 the underlying reasons for acquired cases can be classified into peritrochlear scarring, tendon–trochlear inflammation, superior nasal orbital mass, tight or inelastic superior oblique muscle, short superior oblique tendon, and idiopathic acquired form which are further explained as follows.
Peritrochlear scarring
Surgical or accidental trauma to the trochlea can cause acquired Brown disease. Even chronic sinusitis can lead to chronic scarring in the trochlea appearing as Brown syndrome.31,32 Peritrochlear scarring can also occur following upper lid blepharoplasty because of the removal of periorbital fat during the procedure. 4
Tendon–trochlear inflammation and edema
Inflammation in the area of the superior oblique tendon and trochlea can cause acquired Brown syndrome. Inflammatory diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, and systemic lupus erythematous are in association with acquired Brown syndrome.33–35 Possibly, the local inflammation due to the inflammatory disease or even blunt trauma would lead to swelling in the vascular tissue surrounding the superior oblique tendon–trochlea complex which results in limitation in free movement of the superior oblique around the trochlea. 36
Superior nasal orbital mass
One of the complications following adjusting the glaucoma drainage implant in the superior nasal quadrant is the occurrence of acquired Brown syndrome, which can be prevented.37,38 Another reason for this type of Brown syndrome is the presence of neoplasm in the superior nasal region. In a study, tumor removal surgery returned eye movements to normal. 39 The reason for this type of Brown syndrome is the pushing of the implant or tumor on the superior oblique tendon–trochlea complex and deflecting its function.
Tight or inelastic superior oblique muscle
Acquired loss of elasticity following thyroid eye disease and peribulbar anesthesia in ocular surgery can present signs like an acquired Brown syndrome.4,40,41 This might have occurred due to superior oblique muscle fibrosis secondarily to swelling of the ipsilateral superior oblique muscle or inattention in the intramuscular injection of anesthetics.
Short superior oblique muscle tendon
A superior nasal mass or a superior oblique tuck may shorten the superior oblique tendon. A short tendon is associated with significant hypotropia with intorsion in the primary position. Brown syndrome due to short superior oblique muscle tendon is not as common as inelastic superior oblique muscle. 4
Idiopathic acquired Brown syndrome
It has been shown that most cases with acquired Brown syndrome are classified as idiopathic, as no significant cause of restriction can be found. 4 Idiopathic forms might occur due to low-grade local edema or inflammation in the area of the superior oblique muscle tendon complex, usually not discovered while imaging. It should be noted that normal imaging results would not rule out an inflammatory cause for an idiopathic Brown syndrome. Sometimes remote trauma or hormonal changes might play a role in the manifestation of an idiopathic Brown syndrome. Wright 4 reported a case of having a car accident with a fracture of both legs and reported diplopia soon after the accident. In the examination, left Brown syndrome was diagnosed. The author hypothesized that emboli from a leg fracture obstructed tiny vessels surrounding the superior oblique muscle tendon, leading to local ischemia, inflammation, and edema. Interestingly, many idiopathic cases resolve without any treatment after some time.1,7,42
Diagnosis and differential diagnosis
The diagnosis is usually made following a detailed history taking and during a routine ophthalmologic examination. Evaluating the extraocular muscle motilities (ductions and versions) is a vital step. The cover test and prism alternate cover test are also conducted to reveal any deviation in the primary position and side gazes. The presence of AHP and V pattern strabismus is also common and should be noted. Sometimes, imaging is requested to discover the underlying reasons. Imaging may be indicated to aid in diagnosis and investigate potential underlying causes, especially in acquired or atypical cases. Tests commonly requested include computed tomography scans to detect bony abnormalities or changes from prior trauma/surgery, and magnetic resonance imaging to provide soft tissue contrasts and detect compressive lesions/masses, inflammation, or vascular issues near the trochlear region.4,43,44 Common normal findings on imaging do not exclude inflammatory causes. Rare abnormal findings include trochlear fibrosis or fibrotic bands in the superior oblique tendon region in congenital cases. While imaging may identify underlying pathology, inflammation cannot be ruled out as a cause for idiopathic Brown syndrome based on normal imaging results alone. Together, clinical examination and imaging aim to elucidate the etiology, especially in atypical presentations.
Genetic or family history should be attained from the patient to rule out the familial Brown syndrome.13,17,19,20 As Brown syndrome results from mechanical muscular restriction rather than a paretic cyclovertical muscle, the three-step test might be negative and instead, FDT should be employed in cases with enough cooperation and in suspicious cases to confirm the diagnosis. Conditions with nonrestrictive causes of limited elevation in adduction including inferior oblique muscle palsy, primary superior oblique muscle overaction, double elevator palsy, orbital fracture, and congenital fibrosis syndrome should be differentiated from Brown syndrome.
Inferior oblique palsy and primary superior oblique overaction
It is a rare condition recognized as hypotropia in the primary position with an AHP, superior oblique muscle overaction, and a negative FDT. However, in Brown syndrome, FDT is positive and limitation in elevation, while adduction is not associated with superior oblique muscle overaction.1,7
Double elevator palsy
In double elevator palsy, the limitation of elevation occurs in both adduction and abduction. 7
Orbital fracture
It is sometimes associated with extraocular muscle entrapment and limitations of elevation both in adduction and abduction. Moreover, there is usually a history of trauma, infraorbital tingling, or enophthalmos. 45
Congenital fibrosis syndrome
This rare congenital syndrome affects multiple extraocular muscles. The elevator muscles involved cause limitations in abduction and adduction. 45
Clinical findings
Amblyopia and sensory findings
The evidence obtained so far indicates that amblyopia can occur in patients with congenital unilateral Brown syndrome; however, the incidence is usually low.4,46 Sekeroglu et al. 46 showed that among 44 patients between 4 and 21 years old with unilateral congenital Brown syndrome, 15.9% had amblyopia at initial examination. Amblyopia was shown to have no significant relationship with the age of the patient, ocular alignment at the primary position, anisometropia, or stereoacuity; however, loss of sensory fusion was significantly related to amblyopia. In another study, 8 out of 39 (20.5%) cases with Brown syndrome had amblyopia, although the underlying reasons were horizontal deviation in primary position, anisometropia, and nystagmus, not Brown syndrome. 27 Accordingly, Khorrami-Nejad et al., 26 observed amblyopia in 21.7% of Brown syndrome patients with combined strabismic and anisometropic in seven (53.85%) and strabismic in six patients (46.15%). Other studies have reported less frequency of amblyopia in Brown syndrome. For instance, Wright 4 showed that only 0.05% of patients in his study had amblyopia. Therefore, amblyopia due to Brown syndrome itself is less likely to occur because patients with Brown syndrome may develop an AHP which prevents suppression of one image in cases with vertical strabismus in the primary position. The discrepancy in the prevalence of amblyopia suggests that every patient with Brown syndrome should be assessed carefully for any evidence of amblyopia, either due to the amblyogenic factors or the Brown syndrome itself, especially before any surgical approach.
Spontaneous recovery
There is a possibility of spontaneous recovery in both congenital and acquired cases of Brown syndrome, although the actual incidence is variable in different reports (Table 1).4,29,30,42,47 Considering congenital cases, variable amounts of spontaneous resolution have been reported. In a study with 60 cases with presumed congenital Brown syndrome who continued to follow-up to 46 months, six (10%) patients showed a complete spontaneous resolution in eye movement restriction, that is, elevating the eye while adduction at 4, 6, 8, 10, 12, and 15 years of age. Only 2 out of 38 (5.3%) cases who were orthotropic in the primary position developed a small hypotropia, while most of them tended to remain stable or improve at follow-ups. 29 In another study with 32 congenital Brown cases with an age range of 1 and 14 years, 75% of cases showed spontaneous improvement. 30 Spontaneous resolutions might also occur in bilateral cases. For instance, Capasso et al. 47 reported a 4-year-old girl with bilateral Brown syndrome who recovered in the right eye after 7 months, whereas her eye movement limitation in the left eye did not show any significant change over a 36 months of follow-up.
Evidence of spontaneous recovery in Brown syndrome in different studies.
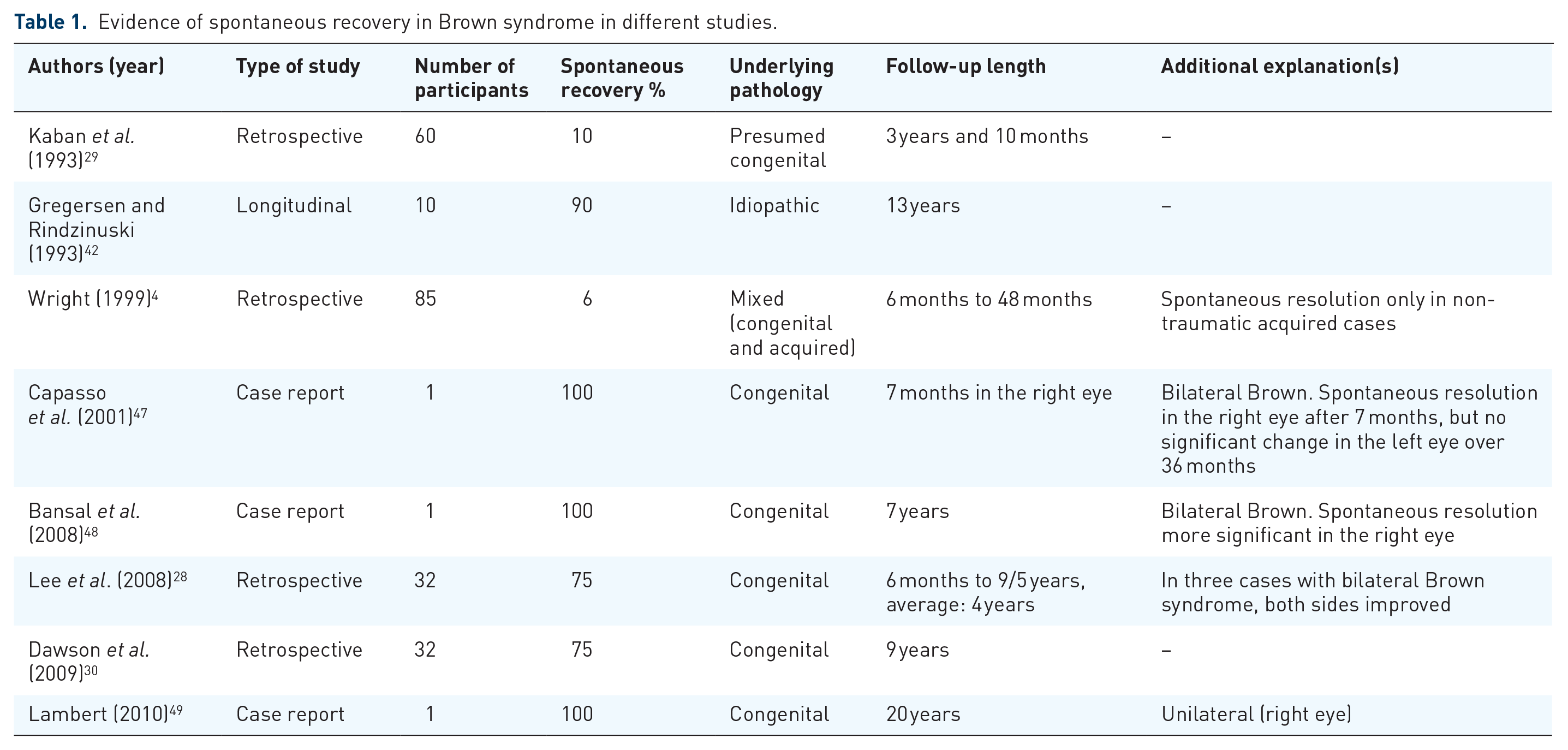
Among acquired cases, in a longer follow-up study with a mean follow-up of 13 years, 3 out of 10 (30%) patients with idiopathic acquired Brown syndrome showed a complete resolution. 42 Wright 4 showed that 5 out of 85 (6%) patients with Brown syndrome recovered spontaneously from 6 to 48 months. All recovered cases had a history of non-traumatic acquired Brown syndrome. Given the possibility of spontaneous recovery regardless of the underlying etiology, therapeutic approaches should be conducted conservatively and after careful observation.
Abnormal head posture
AHP might occur in Brown syndrome cases with vertical strabismus in the primary position to avoid hypotropia and diplopia. The most common type of AHP is head or chin up4,50; however, in some cases, other types of head posture including head tilt to the affected side and head turn to the normal side might be observed. 25 Previous studies have reported that 45.5% of idiopathic cases and 59% of patients with congenital Brown syndrome had AHP.27,30 Figure 3 represents a case of unilateral left Brown syndrome with ipsilateral head tilt. A patch test was conducted in this case and after 45 min, the AHP disappeared.

The left picture shows a 5-year-old girl with left Brown syndrome and ipsilateral head tilt. The right picture shows the same patient after the patch test for 45 min.
Severity
Staging the severity of Brown syndrome is important for management strategies and selecting suitable surgical techniques in cases that require surgery. It is divided into mild, moderate, and severe.
Mild: There is no hypotropia in the primary position. However, there is restricted elevation in adduction.
Moderate: There is no hypotropia in the primary position. However, there is restricted elevation and downshoot in adduction.
Severe: There is hypotropia in the primary position and restricted elevation and marked downshoot in adduction. 45
Management
The management protocol for Brown syndrome depends on the underlying reasons and anatomical features and may contain non-surgical or surgical procedures that should be customized to each case’s pathophysiology. 51
Non-surgical methods
Due to the possibility of spontaneous recovery in all types of Brown syndrome, even in congenital forms, conservative observation is usually recommended.7,52 Dawson et al. 30 showed that 75% of congenital Brown syndrome cases improved in their ocular movement limitations without any intervention when followed up from 6 months to 9.5 years. In a 46-month follow-up study, patients with congenital Brown syndrome and orthotropic in the primary position had a higher chance of staying constant or recovering over time without surgery. 29
In acquired cases, if there is a history of local or systemic diseases, it should be treated first. Local steroid injections in the trochlea, oral corticosteroids and non-steroidal anti-inflammatory drugs are usually prescribed. A case report study showed improvement in signs and symptoms of a bilateral Brown syndrome after weekly injection of intramuscular gold for the treatment of rheumatoid arthritis. 53 In another study with 13 patients with acquired Brown syndrome secondary to trochleitis, early administration of intra-trochlear steroids showed remarkable improvement in clinical features. Patients received 1 ml of triamcinolone 7.84 ± 5.40 days after the diagnosis. Among them, 11 patients (84.6%) showed complete improvement in signs and symptoms after 22.45 ± 13.85 days of the first infusion. Authors advised the surgery in cases who failed to respond to multiple steroid administrations. 54
Surgical methods
According to von Noorden and Campos, 7 surgery is indicated in cases with hypotropia in the primary position and a significant AHP and traumatic cases with no spontaneous recovery to restore binocular vision in the primary position. Another group of patients who might need surgery are those without any binocular vision who habitually fixate on the involved eye and develop an AHP. Due to the possibility of spontaneous resolution in this syndrome, surgery is mostly indicated in patients with moderate to severe Brown syndrome. 62 Careful long-term monitoring is important before recommending surgery, especially in congenital forms. While there is no definitive ideal age, earlier intervention is reasonable if amblyopia risk factors like large deviations are present, or if AHP is fully established and could hinder development.
Brown syndrome has been treated using a variety of operative procedures since it was first described in 1950. Studies have demonstrated that the tendon sheath removal is not effective.55,56 However, tenotomy or tenectomy has been proven to be more successful in improving ductions than another procedure like sheatectomy. 5 Sprunger et al. 57 investigated surgical results of the superior oblique tenectomy in 38 cases with Brown syndrome and showed that the technique was the most effective primary procedure. Patients with AHP were successfully treated with one surgery, with no significant improvement or deterioration in sensory fusion. Another technique that Wright 58 introduced is a silicon tendon expander via elongating the superior oblique tendon in Brown cases with the tight superior oblique tendon. In this technique, elongation was made by a nasal superior oblique tenotomy, and then a piece of a medicinal-grade silicone 240 retinal band was inserted across the severed ends of the tendon. This procedure was carried out on four cases (one eye in each case) having Brown syndrome. Three out of four cases improved significantly in elevation in adduction, with no evidence of sequential superior oblique palsy. One patient, however, did not show improvement even after one more surgery (superior oblique tenectomy), possibly due to misdiagnosis (i.e. a pseudo-Brown syndrome). This method permits for a preplanned measure of tendon disconnection and a graded weakening of the superior oblique muscle. Moreover, the technique is reversible in cases with residual overaction, as the severed ends of the tendon are in control and prompt access. Moghadam et al. 61 investigated the surgical outcome of superior oblique tendon split elongation (10 mm) in 14 patients with severe Brown syndrome (limitation of elevation in adduction > −4). The results showed a significant reduction in hypotropia in the primary position, as well as improved elevation in adduction. The technique was suggested to be used in severe cases of Brown syndrome. In a recent study, the outcome and complications of superior oblique split lengthening were investigated retrospectively in 20 patients. The mean limitation of elevation on adduction and the mean vertical deviation decreased postoperatively and there were few complications following surgery including overcorrection in two patients, hematoma in one patient, and one patient required reoperation. The authors concluded that the technique was safe for people with Brown syndrome. 63 Another technique that has been suggested in cases with moderate to severe Brown syndrome is superior oblique tendon thinning. This technique improved elevation in adduction and hypotropia in the primary position in 21 eyes with Brown syndrome with thickened superior oblique tendon. 62 Table 2 reviews different surgery techniques used in studies.
Different surgery techniques are used in studies.

AHP, abnormal head posture; IO, inferior oblique; SO, superior oblique.
Conclusion
As a restrictive anomaly of eye movements, Brown syndrome can occur in both congenital and acquired forms. Acquired forms can be due to trauma, inflammation, or idiopathic. Spontaneous resolution has been observed in congenital and non-traumatic acquired forms; therefore, careful observation has been recommended. In presumed inflammatory cases, steroid local injection might help. In cases with a need for surgery, superior oblique tendon expansion or superior oblique tenectomy is the preferred method of surgery.
