Abstract
Even after a successful corneal transplant, patients experience severe refractive errors, impeding their rehabilitation and satisfaction. Refractive errors can be caused by recipient pathology and corneal thickness, as well as intraoperative factors such as donor–host discrepancy, recipient’s eccentric trephination, vitreous length, wound apposition, technique of suturing, and suture material. Also, wound healing and the interim between keratoplasty and suture removal contribute to astigmatism. Lamellar keratoplasty outperforms penetrating keratoplasty in terms of endothelial cell loss and endothelial graft rejection, yet the risk of developing refractive errors is comparable. Nonsurgical interventions such as spectacles and lenses fail to provide desirable vision in cases with high astigmatism and corneal irregularity. When these limitations are encountered, surgical interventions including incisional keratotomy, wedge resection, laser refractive surgeries, intracorneal segments, and intraocular lens implantation are employed. However, occasionally, none of these approaches deliver the desired effects, leading to the need for a repeat keratoplasty.
Keywords
Background
Corneal transplantation is a safe and effective option to restore vision. Yet, even after clear and successful corneal grafts, there is a significant risk of developing refractive errors, impeding the patients’ visual rehabilitation. 1 Astigmatism >5 D has a reported incidence rate of 10–31% post-penetrating keratoplasty (PK).2–6 According to previous studies, after deep anterior lamellar keratoplasty (DALK) for keratoconus, astigmatism ranges from 0.0 to 10.0 D, spherical ranging from −13.0 to +7.0 D. 7 Although DALK has some advantages over PK including the reduction of endothelial cell loss and elimination of endothelial graft rejection, it has a similar or even higher incidence rate of developing refractive errors compared to PK.8–10
Recipient pathology and corneal thickness, as well as intraoperative factors including donor–host disparity, recipient’s eccentric trephination, vitreous length, wound apposition, technique of suturing, and suture material, are all risk factors for developing refractive errors. Furthermore, wound healing and interval between keratoplasty and suture removal contribute to astigmatism.11–13
Surgical intervention is required for the management of post-keratoplasty astigmatism in 8–20% of patients. 14 In selected cases, spectacles provide favorable visual acuity; however, contact lenses are a more effective option when there is high or irregular astigmatism and anisometropia.15,16 Moreover, contact lens failure due to corneal irregularity, lens intolerance, dry eye, and problems with manual dexterity 17 creates a need for further intervention, including incisional keratotomy,18,19 wedge resection, 20 laser refractive surgeries,21,22 intracorneal segments,23–26 and intraocular lens (IOL) implantation.27–29 In eyes with prior keratoplasty, refractive surgeries should be performed once the corneal shape and refraction have stabilized. Several studies have recommended at least a 1-month gap between refractive surgery and complete suture removal, with 3–6 month interval after suture removal being optimum.30–32 Herein, we review the available surgical techniques addressing refractive errors following corneal transplantation.
Search strategy
A systematic search of online electronic databases, including PubMed, Scopus, and Web of Science, was conducted using combinations of the following terms: corneal graft, corneal transplant, keratoplasty, refractive surgery, intracorneal segment, keratorefractive surgery, photorefractive keratectomy (PRK), laser subepithelial keratomileusis, and laser in situ keratomileusis (LASIK), to identify all articles reporting refractive surgeries to address post-keratoplasty ametropia in the English language. These bibliographies were searched for English articles from their inception until August 2022. We further screened the reference list of each identified record.
Incisional surgery
Incisional keratotomy includes relaxing incision with or without compression sutures and corneal wedge resection. Relaxing incision corrects approximately 4–5 D of astigmatism 33 ; however, performing just incisional procedures may lead to unsatisfactory and unpredictable results. 34 Guell et al. have suggested to combine relaxing incision with laser refractive surgery in patients who have an astigmatism of greater than 6 D. 35
In recent years, astigmatic keratotomies have been the most widely used incisional surgeries. Arcuate and transverse keratotomies can be performed using mechanical techniques (arcitome) or femtosecond laser (FSL). 19 FSL-assisted keratotomy, especially with the guide of anterior segment optical coherence tomography, has shown advantages over mechanized keratotomy. 36 Post-keratoplasty FSL-arcuate keratotomies are usually performed at depth of 75–85% of thinnest corneal pachymetry on eyes with preoperative cylinder of more than 6 D.37–39 In addition, in a retrospective study on 56 post-keratoplasty keratoconus eyes, Mimouni et al. evaluated the factors associated with vision improvement after FSL-arcuate keratotomies. Multiple regression analysis showed that PK (versus DALK) [odds ratio (OR) = 8.52, p = 0.009], worse preoperative uncorrected distance visual acuity (UDVA) (OR = 9.08, p = 0.02), and higher preoperative cylinder (OR = 1.51, p = 0.04) were independently associated with better UDVA. 40
Intrastromal astigmatic keratotomy (ISAK) was first introduced to restore low astigmatism; yet, Wetterstrand et al. implemented this new approach on eyes with prior PK and a mean refractive cylinder of 6.8 ± 2.2 D and showed that cylinder reduced to 3.7 ± 1.7 within 3 months after the procedure. 41 Furthermore, Loriaunt et al. introduced a variant of this technique, performing deep ISAK under a LASIK flap, that can outperform the ISAK alone approach in correcting high astigmatism. 42 As described by Drouglazet-Moalic et al. in a prospective 2-year follow-up study, this novel technique, deep intrastromal arcuate keratotomy with in situ keratomileusis (DIAKIK), improved sphere from −5.0 to −1.5 and is a safe and promising procedure to treat post-keratoplasty astigmatism. 43
Laser refractive technique
PRK and LASIK are widely performed for the treatment of astigmatism and refractive errors after PK and DALK resulting in a significant improvement in UDVA, refractive, and keratometric improvement.21,22,44–46
Two types of customized laser ablation have been used to treat refractive errors post keratoplasty: wavefront-guided PRK, 47 that uses ocular wavefront and topography-guided photorefractive keratectomy (TG-PRK) 48 that uses corneal aberrations for customizing the ablation.
PRK and LASIK using topography-guided techniques have shown effective results with no difference in the final outcome.48–50 Customized laser ablation can be challenging in highly irregular corneas. Prediction of refractive outcome after TG-PRK procedure is also a challenge. 51
Postoperative course can be complicated by graft rejection due to inflammation induced by ablation. 52 Studies have reported regression of refractive cylinder 45 and corneal haze after PRK that limits its effectiveness53,54; however, in recent years, mitomycin-C application, and improvement in lasers and postoperative care have reduced corneal haze after PRK (Table 1).55,56
Results of post-keratoplasty refractive errors correction using laser refractive techniques in previous reports.
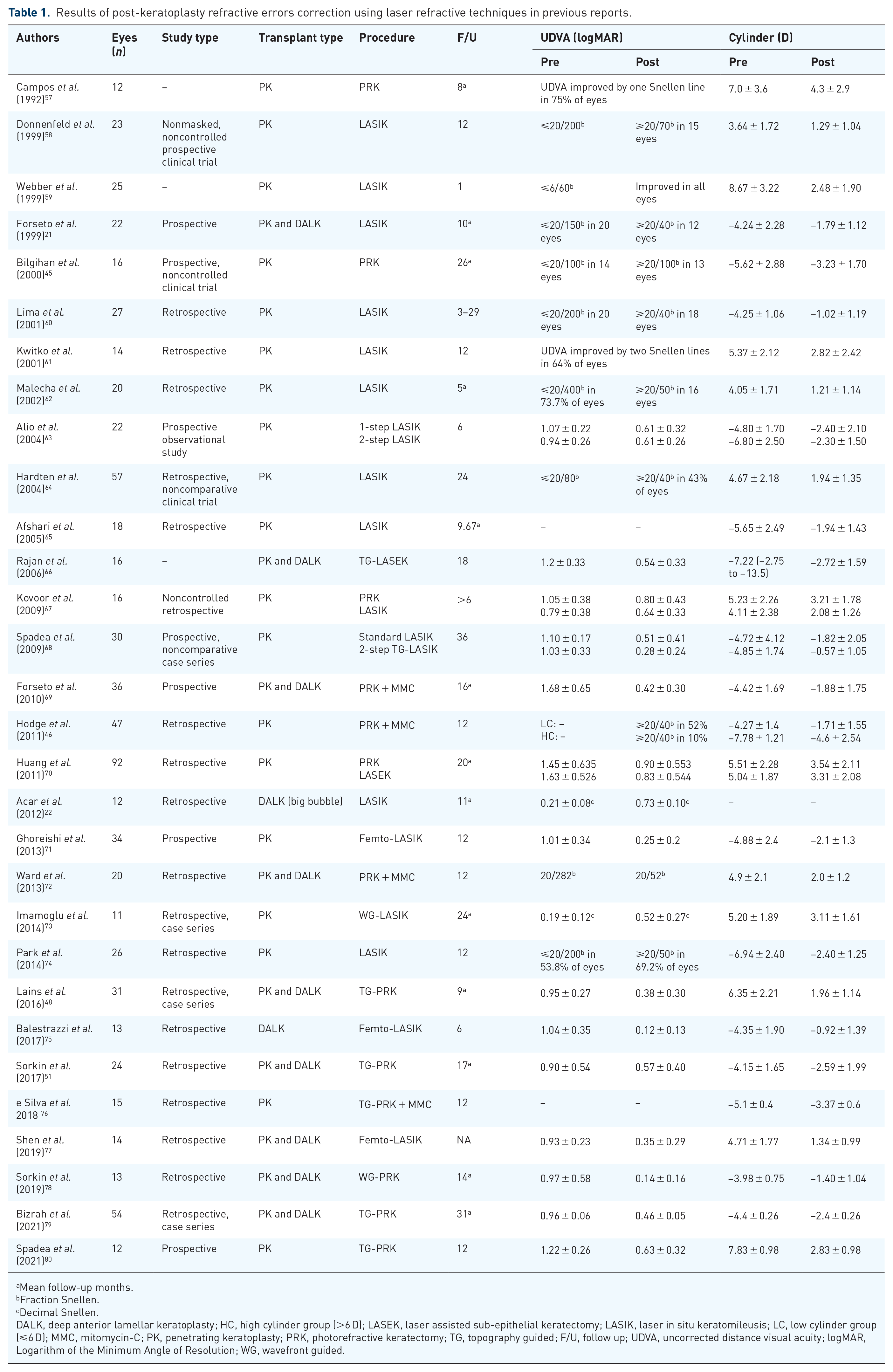
Mean follow-up months.
Fraction Snellen.
Decimal Snellen.
DALK, deep anterior lamellar keratoplasty; HC, high cylinder group (>6 D); LASEK, laser assisted sub-epithelial keratectomy; LASIK, laser in situ keratomileusis; LC, low cylinder group (⩽6 D); MMC, mitomycin-C; PK, penetrating keratoplasty; PRK, photorefractive keratectomy; TG, topography guided; F/U, follow up; UDVA, uncorrected distance visual acuity; logMAR, Logarithm of the Minimum Angle of Resolution; WG, wavefront guided.
Intracorneal segments
Intrastromal corneal ring segment (ICRS), first developed to restore refractive errors, is an efficient treatment for keratoconus. 81 Later some studies described the efficacy of implanting ICRS on correction of high astigmatism following PK, which can be implanted either manually or with femtosecond (FS) laser.23–25,82 Arantes et al. compared the efficacy of ICRS for post-DALK astigmatism implanted using manual technique versus FS laser. The postoperative cylinder, spherical equivalent, and K values were significantly reduced after two techniques. Furthermore, the results revealed lower postoperative complications using FS laser compared to the mechanical technique. Using a Ferrara ICRS with a 6- or 7-mm optical zone compared to 5-mm optical zone leads to less corneal flattening and also increases the probability of neovascularization and dehiscence of graft–host junction. 83 Also, a two-step approach using ICRS first, followed by PRK after a 3-month interval, has shown promising results and demonstrated more effectiveness than ICRS alone. 84
ICRS provides some benefits over laser refractive surgeries, such as lower regression rates, a decreased risk of graft failure or haze, correcting both refractive errors and graft irregularities, less dependence on corneal thickness, and the amount of refractive error. 82
Cataract surgery and IOL implantation
Patients with cataracts who are candidates of keratoplasty would benefit from the triple procedure, a combination of PK, cataract extraction, and implantation of IOL.85,86 Also, a variation of the triple procedure, which is performing DALK and phacoemulsification simultaneously, has been described and reported to be safe and effective in several case series.87–89 Contrastingly, some studies suggest sequential surgery, in which cataract surgery is performed after keratoplasty and complete suture removal when corneal curvatures become stable.90–92 Solaiman et al. introduced another variant of the two-step approach composed of simultaneous PK and lens extraction, ensued by IOL insertion. Following this alternative approach, among 29 patients, 89.7% achieved a mean spherical equivalent of −0.19 ± 0.93 D. 93 This two-step approach leads to a more accurate IOL power calculation; however, it causes trauma to the graft endothelium.93,94
A recent retrospective study adopted scleral incision to perform phacoemulsification on eyes that previously underwent DALK and showed encouraging results in terms of graft survival, and residual refractive errors. 95
Post-PK patients are prone to develop cataracts due to surgical manipulations and chronic use of high-dose steroids. 96 In cases of post-keratoplasty astigmatism with cataracts, the refractive correction can be achieved either by relaxing incision at the time of or preferably prior to cataract surgery. 97 However, toric IOLs offer a more reliable refractive outcome, simultaneously correcting cataracts and ametropia.98–101 On the other hand, toric IOLs are not suitable for irregular astigmatism. 28 Patients with high degrees of astigmatism could benefit from a stepwise approach encompassing both techniques, as demonstrated by Sorkin et al., performing femtosecond astigmatic keratotomy followed by phacoemulsification with toric IOL placement in post-keratoplasty patients with high astigmatism (⩾8 D) is an efficient and safe approach. After both procedures, the corneal astigmatism decreased from 13.56 ± 4.81 to 4.48 ± 2.83 D, and UDVA improved from 1.69 ± 0.45 to 0.23 ± 0.11 logMAR. 14
An important consideration with toric IOLs is rotational stability. In early generations of toric IOLs, the rotational stability was a significant concern with 16–50% rotation incidence; however, in recent studies the, reported mean IOL rotation has been lower than 4°.99,102–104 IOL power calculation after keratoplasty is entangled with errors and requires accurate measurements. 105 As reported in a recent study, all the assessed formulas have a trend to induce myopic refractive shift in post-DALK patients. Despite the lower accuracy of formulas in post-DALK eyes than eyes without prior surgery, SRK/T, Kane, Emmetropia Verifying Optical, Hoffer QST were more reliable than other formulas. 106
Phakic IOL
Compared to laser refractive surgery, phakic IOLs provide a broader range of refractive error correction, and apart from faster visual rehabilitation, maintaining accommodation, stable refraction, and reversibility, they are not limited by corneal thickness or topography. 107 In addition, Feizi et al. reported that phakic IOLs yield superior visual outcomes compared to LASIK in a retrospective study involving 33 post-PK eyes. The safety and efficacy indices favored phakic IOL (0.82 ± 0.34 and 1.13 ± 0.30, respectively) over LASIK (0.22 ± 0.17 and 0.85 ± 0.24, respectively). 108
IOLs are designed to address both spherical and cylindrical refractive errors. As reported, anterior chamber iris-fixated phakic IOLs such as artisan and artiflex correct myopia from −3.00 to −23.50 D and −2.00 to −14.50 D, respectively. Toric lenses can correct astigmatism from −1.00 to −5.00 D. Consistently, in a study on post-PK cases, Tahzib et al. reported an 88.8% decrease in astigmatism and a 103.6% decrease in sphere. 29 Also, following implantation of artisan/artiflex toric IOLs in eyes with previous DALK, the mean spherical and cylindrical errors were reported to decline 73.1% and 83.8%, respectively. 17
Visian implantable Collamer lens (ICL), a posterior chamber (PC) phakic IOL, corrects myopia from −3.00 to −23.00 and cylinder up to 6.00 D. 107 Correcting refractive errors with phakic ICL in post-keratoplasty eyes revealed promising results, gaining 1 or more lines of corrected distance visual acuity (CDVA) in 46–76.4% and achieving CDVA ⩾ 20/40 in 80% of cases.109,110
In a retrospective study on post-keratoplasty patients who underwent phacoemulsification, Moon et al. evaluated the differences of front-, back-, and bi-toric IOLs. All surgeries were performed at least 6 months after suture removal. While bi-toric IOLs demonstrated superior outcomes for individuals without prior transplant, post-keratoplasty patients exhibited comparable visual outcomes across different types of toric lenses. 111
Endothelial cell density (ECD) loss is a challenging complication following the implantation of IOLs, especially after PK which has a higher endothelial cell loss compared to DALK (27.7% versus 12.9% after 1 year, respectively). 112 ECD loss was 30.4% 3 years after IOL implantation in eyes with previous PK. 29 In a retrospective study on post-DALK patients who underwent subsequent toric phakic IOL implantation, Malheiro et al. reported a continuing ECD loss, reaching 14.0% after 5 years. 17 Thus, adequate endothelial cell count and anterior chamber depth are prerequisites for phakic IOL implantation; interestingly, PC IOLs are preferred due to less endothelium damage and small incision size. 107
Piggyback IOL
In post-keratoplasty pseudophakic eyes, the insertion of a piggyback IOL effectively corrects the residual ametropia after keratoplasty. This procedure is safer than exchanging the lens because it avoids capsular tears and subsequent vitreous loss, retinal tears and detachment, macular edema, cyclodialysis, and zonular damage. Also, it is reversible in the event of graft failure and need to repeat PK.113,114 Moreover, a retrospective study by Alfonso et al. evaluating the piggyback sulcus toric ICL for residual astigmatism in pseudophakic eyes with previous corneal surgery demonstrated good refractive outcomes and no complications. UDVA ⩾ 20/40 was achieved in 45.5% and 42.9% of post-PK (n = 11) and post-DALK (n = 7) eyes, respectively. 115
There are some concerns regarding piggyback IOL implantation, such as pigment dispersion, pigmentary glaucoma, interlenticular opacification, pupillary block, iridocyclitis, and hyphema. 116 To avoid interlenticular opacification, the supplementary IOL should be placed in the ciliary sulcus instead of capsular bag. 114
Like other toric lenses, a major drawback for piggyback toric lenses is rotational instability. A retrospective case series of 44 pseudophakic eyes (including 19 post-keratoplasty eyes) undergoing Sulcoflex toric lenses implantation reported a mean maximum rotation of 17.63°, with 62% of IOLs requiring repositioning. Fifty-six percent of post-keratoplasty eyes acquired UDVA of 20/40. 116
Repeat keratoplasty
When all previous procedures have failed to address post-keratoplasty refractive errors, re-keratoplasty can be considered as a final resort. By performing repeat PK with 193 nm with Zeiss-Meditec MEL-60 excimer laser using round metal masks (diameter, 7.5–8.0 mm) and implementing double running sutures, in a retrospective study, Szentmary et al. reported a considerable improvement in best corrected visual acuity and astigmatism with all sutures remained. 117 As a result, they suggested that final suture removal should be deferred as long as feasible to avoid a significant increase in astigmatism. Another retrospective study on 109 PKs (including 59 repeat PKs, with 30 due to high astigmatism) reported favorable visual outcomes using 8.5/8.6-mm excimer laser-assisted PKs for highly irregular astigmatism due to increased corneal regularity. Also, all the repeat grafts performed due to high irregular astigmatism remained clear at the last follow-up (with an average last follow-up of 19 months). 118
Furthermore, Gutfreund et al. described a modified version of microkeratome-assisted anterior lamellar keratoplasty (MALK) to correct high irregular astigmatism in post-PK eyes, achieving CDVA of 20/20 in the four cases included in the study. Additionally, unlike PK, MALK has the advantage of not destroying the healthy endothelium and not imposing the risk of a new immunologic response on patients. 119
Conclusion
Refractive errors hinder keratoplasty results, demanding further interventions. Additionally, only a limited number of patients can tolerate nonsurgical interventions, and the majority of the patients eventually require refractive surgeries. Each of these approaches is employable in specific situations. The optimal technique should be used, depending on the type of transplant, presence of cataracts, the type of refractive error, and the severity of ametropia. When all other interventions have proven ineffective, a repeat transplantation can be considered.
