Abstract
Keratoconus is a progressive ectatic corneal disease characterized by corneal thinning, steepening, and irregular astigmatism, presenting unique challenges for intraocular lens (IOL) calculations in cataract surgery. The use of traditional formulas for IOL calculations in eyes with keratoconus frequently leads to significant refractive prediction errors, often resulting in a hyperopic shift, which can lead to suboptimal outcomes and reduced patient satisfaction. This review examines the impact of keratoconus on IOL calculations and highlights recent advancements in formulas specifically designed for eyes with keratoconus, particularly the Barrett True-K and the Kane Keratoconus formulas. We assess the effectiveness of these formulas based on the current literature and provide clinical recommendations aimed at improving surgical outcomes in this complex patient group.
Keywords
Introduction
Keratoconus is a progressive ectatic corneal disease with an estimated prevalence ranging from 0.2 to 4790 per 100,000 people, 1 and is characterized by corneal thinning, steepening, and irregular astigmatism. These result in distorted vision and reduced visual acuity, which significantly compromise patients’ quality of life by affecting not only their vision but also their mental health and social interactions.1,2 When a cataract progresses in patients with keratoconus, surgeons have a unique opportunity to improve patient’s vision through cataract surgery while simultaneously correcting their refractive error. Nevertheless, traditional IOL calculation formulas become increasingly inaccurate in patients with keratoconus, raising the risk of postoperative refractive errors.3,4 This is because such formulas assume a regular corneal curvature and do not adequately account for anatomical differences such as altered anterior-to-posterior curvature ratios, posterior corneal astigmatism, and challenges in estimating effective lens position (ELP).4 –7
In recent years, dedicated formulas specifically adapted for keratoconus, such as Barrett True-K and Kane Keratoconus, have emerged and been evaluated in multiple clinical studies, showing promising results.3,4,6,8,9 However, their adoption in clinical practice varies, and clear guidance on their application remains limited. A surgeon’s understanding of the complexities of IOL calculations in patients with keratoconus,4,10,11 along with familiarity with the advanced formulas currently available for these cases, may significantly improve patients’ refractive outcomes and, as a result, their quality of vision and overall satisfaction.
In this pivotal moment in the evolution of AI-based and keratoconus-specific IOL formulas, this review examines recent studies on IOL power calculation in keratoconus, including ones that were not covered in earlier reviews, and provides an overview of the current state of the field. It aims to help cataract surgeons navigate the specific challenges of IOL calculation in this complex patient population by presenting the most up-to-date evidence and offering a structured, step-by-step clinical approach—from preoperative assessment to postoperative care—to facilitate better-informed decision-making and improve outcomes.
Methodology
In this review, we performed a comprehensive literature search to identify relevant studies on IOL power calculation in keratoconus. We searched PubMed, Web of Science, and Scopus databases using keywords such as “keratoconus,” “cataract surgery,” “IOL power calculation,” “IOL formula,” and “keratoconus IOL formula.” The search included articles published up to March 2025, with a focus on English-language peer-reviewed papers. We included clinical studies (retrospective or prospective), meta-analyses, and review articles addressing IOL calculation outcomes, as well as publications discussing best clinical practices. There were no strict exclusion criteria on study design given the narrative scope, but articles were selected based on relevance to keratoconus and cataract outcomes, recency, and scientific soundness (with preference for larger studies or those with modern biometric methods). Reference lists of pertinent papers were also hand-searched for any additional publications.
The impact of keratoconus on IOL calculations
The inherent corneal properties of keratoconus, characterized by thinning and protrusion, lead to a conical corneal shape that distorts vision. 12 The abnormal cornea in patients with keratoconus significantly disrupts the standard assumptions used in IOL calculations, particularly concerning corneal shape and uniform curvature. 13 Specifically, the altered ratio between anterior and posterior corneal curvatures—typically assumed constant in traditional IOL formulas—impacts the estimation of the total corneal power (TCP) and the ELP, resulting in a higher rate of refractive, mostly hyperopic, prediction error.4 –7 Additional anatomical considerations unique to keratoconic eyes that affect IOL calculation accuracy are outlined in Table 1, including the limitations of standard keratometry, which measures only the anterior corneal surface while assuming a fixed relationship with the posterior cornea. In keratoconus, the posterior curvature is steeper than normal, leading to an underestimation of TCP and introducing errors in IOL calculations. This discrepancy becomes more pronounced as keratoconus severity increases, resulting in significant refractive prediction errors that are less common in normal eyes. 14
Challenges of IOL calculations in eyes with keratoconus.

Furthermore, in keratoconus, the visual axis may not align with the corneal apex, while the decentered cone can cause asymmetric astigmatism and higher-order aberrations, complicating biometric measurements. Other challenges include the generally longer axial lengths (ALs) and deeper anterior chambers in keratoconic eyes, which impact ELP estimates and increase the risk of inaccurate refractive outcomes.15,16 Obtaining accurate preoperative corneal power measurements is also difficult, as keratometry readings are often unreliable due to steep curvature, multifocal and irregular surfaces, and a displaced visual axis, resulting in poor measurement repeatability.11,17 The heterogeneous severity of keratoconus further complicates IOL calculations, as grading standards vary across studies but are often based on the steepest K value.3,9,18,19 Accuracy in IOL prediction is generally diminished as keratoconus severity increases, largely due to increased variability in corneal power measurements and the fact that ELP models rely heavily on corneal power, which becomes less predictable in advanced stages.3,10,20
Recognizing the anatomical and refractive distinctions between keratoconic and normal corneas is essential for accurate IOL power predictions. This includes applying specific diagnostic criteria, identifying astigmatism patterns, and using advanced imaging modalities such as Scheimpflug tomography and swept-source optical coherence tomography (SS-OCT) to measure both anterior and posterior corneal surfaces, as well as performing preoperative pachymetry to ensure accurate preoperative assessments.1,15 These clinical and imaging inputs help identify the asymmetrical and irregular corneal shape characteristic of keratoconus, contrasting with the more regular curvature typical of normal corneas.14,21 Such patterns are critical during preoperative assessments, not only for diagnosing keratoconus but also for guiding IOL selection and determining the suitability of a toric lens.4,10 In addition, avoiding diagnostic confusion with pseudoectasia or corneal ectasia mimics—especially those arising from technical imaging errors—is essential to prevent inappropriate use of keratoconus-specific IOL formulas in non-keratoconic eyes, which could lead to refractive errors. 22 Thus, a meticulous preoperative evaluation is important for achieving precise IOL calculations and minimizing postoperative refractive surprises in keratoconic patients. This includes a thorough slit-lamp examination and tomography when indicated by corneal astigmatism measurements from biometry or keratometry. It is essential to perform all necessary preoperative assessments to confirm the diagnosis of keratoconus, obtain accurate measurements, and evaluate the severity of the disease to guide precise surgical planning.
Historical perspective of IOL calculations in patients with keratoconus
The aforementioned use of traditional Gaussian formulas often led to significant refractive errors and a significant hyperopic shift in particular, due to overestimation of the corneal power, especially in eyes with steeper keratometric values (⩾50.0 D).10,23 –25 Patients with keratoconus, who were accustomed to being highly myopic throughout their lives, were particularly intolerant to these hyperopic surprises. As a result, surgeons often aimed for a myopic refractive outcome, which led to inconsistent results. 10 Acknowledging this challenge, the SRK/T formula was previously considered more suitable for patients with keratoconus, as it tended to induce a myopic surprise in healthy eyes with steep corneas. However, it still was not performing very well in keratoconic eyes, with only 30%–59% of eyes achieving refraction within ±0.50 diopters (D) of intended correction and 64%–91% within ±1.00 D.3,4,9,11,16,26,27 The Haigis formula, designed for longer ALs, achieved even poorer results with 36%–78% within ±1.00 D of the intended refractive target in patients with keratoconus.4,9,10,16,26 Similarly, the Hoffer Q formula only reached 24%–69% accuracy within ±1.00 D from the refractive target in keratoconus patients, with a larger error in severe cases, exceeding ±4.00 D at times.4,9,10,16
Modern IOL formulas that perform well in normal eyes still underperform in keratoconus. The Barrett Universal II (BU2) formula, regarded as very accurate in normal eyes, 28 achieves only around 40%–78% accuracy within a ±1.00 D range for keratoconus.3,4,10,11,16,26,27 Similarly, the Kane formula only reaches 40%–81% accuracy within a ±1.00 D range in keratoconus.3,4,6,11,16,26,27 For longer ALs, Holladay 2 was introduced as an improved version of the Holladay 1 formula, yet only achieved an accuracy of 46%–55% within a ±1.00 D range from the refractive target in keratoconus.3,11,16,19 The Olsen formula exhibited similar results with only 50%–71% of the eyes within a ±1.00 D range.3,16
The EVO 2.0 formula also tends to result in hyperopic errors, and in a study by Heath et al., 26 when using either total keratometry or standard keratometry in its calculations, achieved a mean absolute error (MAE) of 0.799 D and 0.833 D, respectively, with 49.3%–55.2% of eyes within ±0.50 D and 65.7%–68.7% within ±1.00 D. The PEARL-DGS formula also resulted in a positive median numerical error (MedNE) of +0.32, 16 achieving 40.3%–51.5% of eyes within ±0.50 D and 65.7%–69.1% within ±1.00 D. The use of neural networks and artificial intelligence (AI) in IOL power calculations is promising 29 and may be beneficial in the future in complex cases like keratoconus. AI refers broadly to the ability of machines to perform tasks that typically require human intelligence, while machine learning, a subset of AI, focuses on systems that learn patterns from data to improve performance without explicit programming. 30 As more data on underrepresented eyes are collected and shared among institutions and countries, AI models may improve predictions by considering parameters that may not have been previously recognized as important. This collaborative data cultivation may allow AI to analyze unique eyes more effectively, potentially leading to better accuracy than formulas based on smaller datasets. However, like other formulas that are not specific to keratoconus, as the cornea becomes steeper, these formulas tend to result in hyperopic surprises. The RBF 3.0 formula, which uses machine learning, achieved 61.8% of eyes within ±0.50 D and 77.9% within ±1.00 D of target, showing a slight positive bias with a MedNE of +0.21 ± 1.09 D. 16 Therefore, although the modern nonkeratoconus specific formulas offer improved accuracy in normal eyes compared to their predecessors, they remain inadequate in addressing the anatomical irregularities of keratoconic eyes and are not recommended in these patients.
Modern IOL formulas designed for keratoconus
Because of the intricate challenges keratoconic eyes pose to IOL calculation (Table 1), and the underperformance of existing formulas, there was a need to develop new formulas that would specifically account for the eyes’ special characteristic anatomical features. Even with the best of efforts, evaluating the accuracy of keratoconus-specific formulas is challenging, and their performance, as well as the recommended refractive target when utilizing these formulas, may depend on the severity of the disease. Below, we review key formulas specifically designed or altered for eyes with keratoconus, focusing on their reported accuracy from recent studies and meta-analyses.
Barrett true-K for keratoconus
The Barrett True-K (BTK) formula (https://www.apacrs.org/disclaimer.asp?info=2) integrates both anterior and posterior corneal curvature into IOL calculations, enhancing the formula’s accuracy in eyes with keratoconus. Its strength lies in using measured posterior corneal astigmatism (MPC) or predicted posterior corneal astigmatism (PPC) to adapt calculations to individual corneal structures while also adjusting for the actual corneal thickness. This dual use of predictive modeling and measured data improves accuracy, especially compared to conventional formulas, as it accounts for irregular corneal power distribution and adjusts for the corneal thickness asymmetries found in keratoconus.6,26
Several studies have demonstrated the efficacy of the BTK formula in keratoconus patients. Kozhaya et al. 16 evaluated the results of 68 eyes (44 patients) with keratoconus undergoing cataract surgery. BTK achieved 61.8% of eyes within ±0.50 D and 76.5% within ±1.00 D, with a MedNE of −0.13 ± 1.15 D. Ton et al. 4 reported on 32 eyes of 23 patients with keratoconus and found that BTK with MPC achieved the highest precision, with 87.5% of eyes within ±0.50 D and 90.6% within ±1.00 D. This study highlighted the significance of incorporating measured posterior corneal astigmatism to improve accuracy in these patients. Vandevenne et al. 6 evaluated 57 eyes from keratoconus patients to compare the effectiveness of BTK’s predicted and measured forms. The study reported that when using either MPC or PPC, 71%–72% and 88%–90% were within ±0.50 D and ±1.00 D, respectively. Median absolute errors were 0.14 D and 0.10 D for PPC and MPC, respectively. In a meta-analysis by Reitblat et al., 3 which included 623 eyes, BTK with MPC was the most accurate in mild to moderate keratoconus. It achieved 62% of eyes within ±0.50 D and 81% within ±1.00 D, with an MAE of 0.62 D and a mean prediction error of +0.10 D. In moderate keratoconus cases, it achieved 88% within ±1.00 D. A Bayesian network meta-analysis by Tian et al. 8 analyzed 637 eyes across nine retrospective clinical trials comparing 18 IOL formulas. Based on Surface Under the Cumulative Ranking Curve (SUCRA), BTK with PPC and BTK with MPC ranked first (97.3%) and third (77.9%), respectively, for achieving outcomes within ±0.50 D, and first (95.1%) and second (90.1%) for ±1.00 D. The authors recommended BTK with PPC and MPC as preferred options for IOL power calculation in keratoconus patients. Yokogawa et al. 31 examined 50 eyes with keratoconus and found that when compared to Kane Keratoconus (Kane KCN), Kane, SRK/T, Haigis, and BU2, the BTK formula with PPC had the highest prediction accuracy in severe keratoconus (stage 3, K > 53 D), achieving an MAE of 0.55 D, with 65% of eyes within ±0.50 D and 88% within ±1.00 D of target refraction. In a recent study by Parra-Bernal et al., 27 BTK with PPC and MPC showed the highest overall accuracy among all formulas tested, with 52.83% and 60% of eyes within ±0.50 D, and 85.45% and 81.82% within ±1.00 D, respectively. The corresponding MAE were 0.66 D and 0.68 D, with mean prediction errors (RPE) of +0.12 D and −0.01 D, respectively.
Kane keratoconus formula
To account for irregular corneal surfaces and altered anterior-posterior ratios, the Kane KCN formula (https://www.iolformula.com/) modifies the anterior corneal radii to better represent the true anterior/posterior ratio. It incorporates advanced optical theories, regression analysis, and AI, which help the formula minimize errors in ELP calculation and adapt to different corneal shapes, ALs, and astigmatism patterns. 23 Kozhaya et al. reported that the Kane KCN formula achieved 58.8% of eyes within ±0.50 D and 73.5% within ±1.00 D, with a MedNE of +0.10 ± 1.09 D. 16 In a study by Kane et al. 11 on 147 eyes from patients with keratoconus across different disease severities, it achieved 50% of eyes within ±0.50 D and 90.5% within ±1.00 D. Notably, it minimized hyperopic errors to just +0.02 D in stage 3 keratoconus (steepest K > 53 D), outperforming traditional formulas that showed significant hyperopic shifts between +1.72 D and +3.02 D. 11 In the study by Ton et al., 4 this formula achieved 53.1% of eyes within ±0.50 D and 90.6% within ±1.00 D, although greater variability compared to the BTK was demonstrated. In the meta-analysis by Reitblat et al., 3 data for severe keratoconus cases were limited (n = 9); however, among the formulas evaluated, the Kane KCN formula showed the best results, reaching 48% accuracy within ±1.00 D. In the Bayesian network meta-analysis by Tian et al., 8 Kane KCN ranked third for outcomes within ±1.00 D (SUCRA 77.7%), following BTK with PPC (95.1%) and BTK with MPC (90.1%). For ±0.50 D, Kane KCN ranked fourth (SUCRA 72.5%), again behind BTK PPC (97.3%) and BTK MPC (77.9%). In the retrospective study by Parra-Bernal et al., 27 Kane KCN demonstrated the second-highest overall accuracy after BTK, achieving 45.45% of eyes within ±0.50 D and 72.73% within ±1.00 D, with an MAE of 0.75 D and a mean prediction error (RPE) of +0.20 D. According to Yokogawa et al., 31 Kane KCN achieved an MAE of 0.63 D in severe keratoconus, with 58% of eyes within ±0.50 D and 85% within ±1.00 D of the intended refraction. While slightly less accurate than BTK, the Kane formula still performed well in severe keratoconus cases, albeit with reduced predictability at very steep K values.
This review is limited by the heterogeneity of the included studies, which vary in keratoconus severity classification, biometry platforms, and outcome reporting. No formal meta-analysis was performed, and differences in formula versions or calculation tools across studies may have affected comparability. In addition, while the literature generally supports the use of keratoconus-specific formulas such as BTK and Kane KCN, a recent retrospective study by Kahuam-López et al. 32 found no statistically significant difference between these and standard formulas. It is of note, though, that the study spanned more than two decades, with variability in surgical technique, IOL models, and diagnostic equipment that may have obscured meaningful differences between formulas. Many cases relied on older-generation biometry and tomography that lacked measured posterior corneal data, limiting the evaluation of formulas such as BTK with MPC. These findings highlight the need for prospective studies using modern diagnostics, standardized IOL constants, and consistent methodology.
Clinical recommendations
IOL calculations and selection in keratoconus patients are challenging and require a personalized approach that addresses the corneal irregularities and unique refractive needs of each patient. Below are clinical recommendations to optimize refractive outcomes in these complex cases, with a summarized decision-making pathway illustrated in Figure 1.
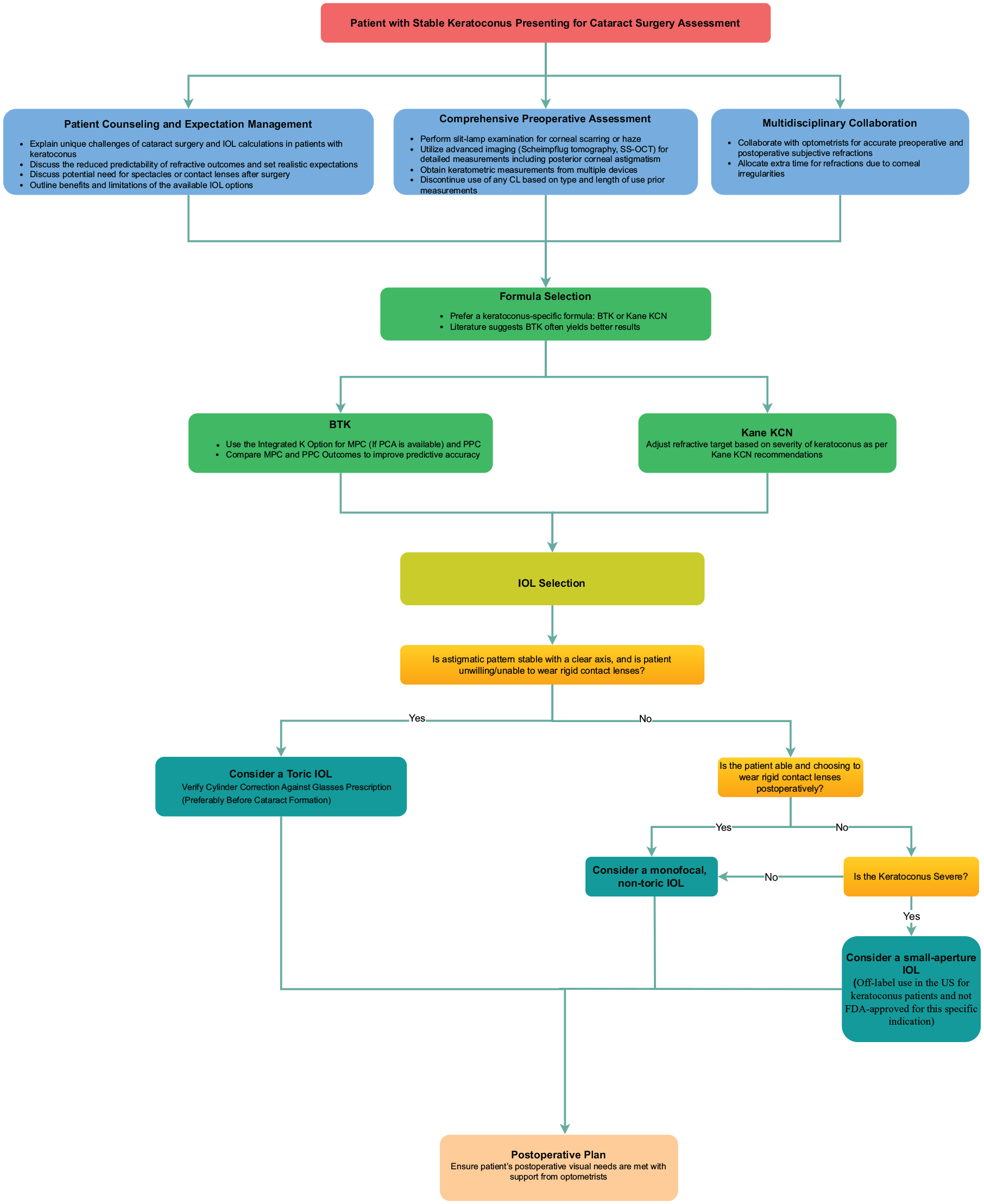
Clinical decision pathway for the perioperative management of cataract surgery in patients with stable keratoconus.
Patient counseling and expectation management
Adjusting patient expectations is immensely important in these cases. Often, patients with keratoconus (KCN) may have unrealistic expectations regarding their refractive outcomes, which should be addressed, and the specific challenges explained. Surgeons should explain how keratoconic eyes differ from normal ones and set realistic expectations about the reduced ability to predict the refractive outcome. The likely need for spectacles or contact lenses after surgery should also be thoroughly discussed, along with the benefits and limitations of different IOL options in the presence of KCN.
Comprehensive preoperative assessment
A thorough preoperative evaluation is essential. Careful slit-lamp examination can reveal corneal scarring or haze that may influence IOL selection as well as visual prognosis. Utilizing advanced imaging techniques such as Scheimpflug tomography and swept-source optical coherence tomography to obtain detailed measurements of both anterior and posterior corneal surfaces and pachymetry is also recommended to increase postoperative refractive accuracy.5,33 This aids in diagnosing keratoconus and accurately grading its severity. In addition, it can identify asymmetries or irregularities that may impact calculations and subsequent IOL selection. Because keratoconus presents along a wide clinical spectrum—from mild, nonprogressive forms to advanced ectasia with significant irregularity—surgical planning must be individualized. Disease severity and anatomical variation influence not only the choice of IOL formula, but also the refractive target, use of toric lenses, and counseling about visual expectations.
Assessing keratoconus stability is essential for guiding treatment and surgical planning. According to the Global consensus on keratoconus and ectatic diseases, disease progression is commonly identified by changes in at least two parameters: steepening of the anterior or posterior corneal surface, corneal thinning, or an increased rate of thickness variation from the periphery to the thinnest point. 34 The ABCD classification system is a widely used framework for grading keratoconus and following its progression, 35 evaluating anterior (A) and posterior (B) corneal curvature, corneal thickness at the thinnest point (C), and best-corrected distance visual acuity (D). Building upon this classification, recent studies have emphasized the importance of incorporating corneal biomechanical properties into keratoconus staging and surgical planning.36,37 An additional “E” parameter has been proposed to represent corneal stiffness.36,37 This ABCDE approach may offer a more complete evaluation of ectasia severity and may better guide surgical timing and the need for cross-linking. Although biomechanical data are not currently integrated into IOL power calculation formulas, they can support clinical decisions by identifying eyes at higher risk for ectatic progression or the potential for more unpredictable postoperative refractive outcomes.
Whenever possible, cataract surgery should be delayed until keratoconus is stable to ensure accurate preoperative measurements and optimize postoperative outcomes. For cases with documented progression, corneal cross-linking (CXL) may be considered to stabilize the cornea, while intrastromal corneal ring segments may be employed to improve the corneal shape and reduce irregular astigmatism.38,39 Advanced cases with significant thinning or scarring may require surgical interventions such as deep anterior lamellar keratoplasty or penetrating keratoplasty (PKP) to optimize visual outcomes. 40 Patients should also be advised to avoid eye rubbing, as it increases mechanical stress on the cornea, exacerbates ectasia, and accelerates disease progression. 1
Like keratoconus, other corneal ectasias, including pellucid marginal degeneration (PMD) and post-refractive surgery ectasia, exhibit features of progressive corneal thinning and bulging but require tailored management approaches.34,41 While keratoconus is characterized by paracentral thinning and conical protrusion, PMD involves inferior thinning near the limbus, often presenting with a “crab-claw” pattern on topography.34,41 PMD typically progresses more slowly but may continue later in life for some patients.34,41 Post-refractive surgery ectasia arises following prior surgical interventions, such as Laser-Assisted In Situ Keratomileusis (LASIK) or radial keratotomy, and commonly corresponds to zones of biomechanical weakening. 41 The clinical recommendations discussed in this review, such as the importance of achieving disease stability, the role of advanced diagnostic tools, and considerations for IOL formulas, also apply to patients with other corneal ectasias. However, post-refractive ectasia often manifests with less uniform corneal changes, 41 resulting in significant variability in topography and irregular astigmatism, imposing additional challenges in surgical planning. Similarly, the highly irregular astigmatism in PMD may further complicate IOL calculations and reduce predictability. Tailored management approaches, including the use of keratoconus-specific IOL formulas and detailed patient counseling, are important to optimize outcomes in these patients.
Acquiring keratometric measurements from multiple devices is valuable, as combining results, such as with the integrated K option in BTK, may improve accuracy, which has previously been shown in normal eyes. 42 Since corneal warpage from contact lens wear can affect tomographic measurements, it is important to discontinue contact lens use prior to preoperative imaging. For patients wearing soft contact lenses, studies suggest that ceasing lens wear for three weeks is generally sufficient to avoid corneal warpage and ensure accurate preoperative measurements, particularly in long-term lens users.43 –45 In contrast, for patients who wear rigid contact lenses, a commonly accepted guideline is to remove lenses for at least 1 month for every decade of wear. 46 However, the exact duration required to achieve stable measurements has not yet been fully elucidated, and no comprehensive comparative studies have been conducted to validate this specific timeline against shorter durations. Until more conclusive evidence is available, these recommendations are based on common clinical practice and expert consensus, ensuring the best possible preoperative measurements for surgical planning.
Incorporation of posterior corneal astigmatism
Whenever possible, it may be wise to incorporate measured posterior corneal astigmatism into IOL calculations. Studies have shown that formulas utilizing the measured posterior corneal data, such as BTK with MPC, achieve better precision.3,4,6,27 If direct measurements are not available, keratoconus-specific formulas using predicted posterior corneal astigmatism can still improve outcomes compared to traditional formulas. 3
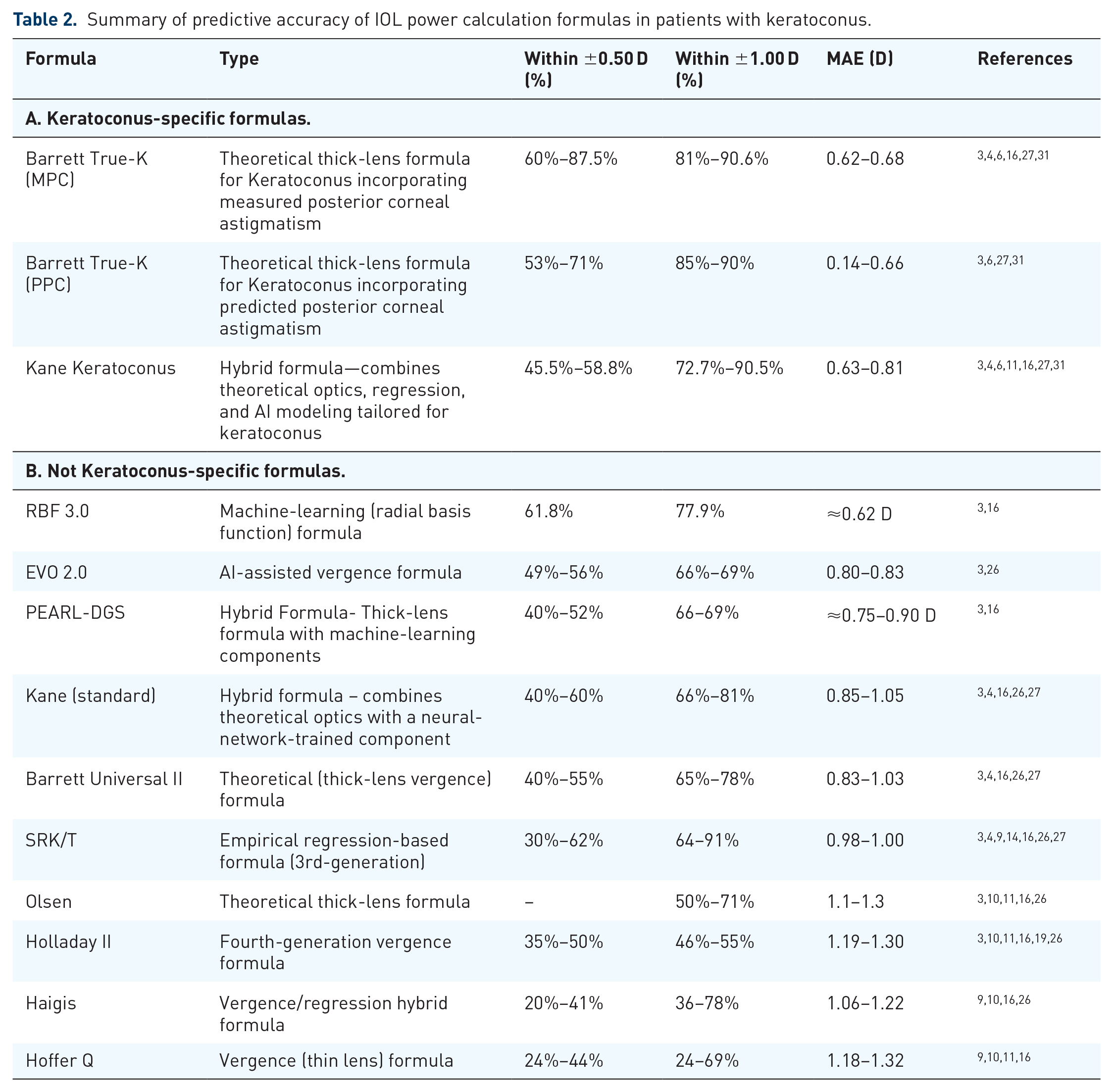
Formula selection
Table 2 presents a comparative overview of prediction accuracies from recent studies. The literature suggests using a keratoconus-specific formula such as BTK or Kane KCN.3,4,6,8,11,16,26,31 Either formula generally improves outcomes over standard formulas, but studies indicate3,4,6,8,16,26,27,31 that BTK often yields better results than Kane KCN. A notable difference between the formulas lies in how they address the challenges posed by steep keratoconic corneas. Kane KCN incorporates an adjustment to the refractive target based on the steepness of the K values, presumably to reduce hyperopic outcomes that are more likely with increasing steepness. For example, the Kane formula’s website recommends targeting between −1.5 and −2.5 D for K values above 59 D. 47 While the exact methodology is not publicly detailed, this adjustment suggests an empirical approach to addressing hyperopia, which may introduce variability. In contrast, the BTK utilizes a theoretical model to account for the disrupted relationship between anterior and posterior corneal radii in keratoconus. This model inherently manages steep K values within its predictive framework, eliminating the need for refractive target offsets. With BTK, we recommend using the integrated K option for both MPC and PPC and comparing the two outcomes to enhance predictive accuracy.
Summary of predictive accuracy of IOL power calculation formulas in patients with keratoconus.
IOL selection
Due to higher-order aberrations and irregular astigmatism in keratoconus, multifocal and extended-depth-of-focus IOLs are generally not recommended, with monofocal IOLs being preferable. Additionally, due to the negative spherical aberration more common in this disease, surgeons might consider IOLs with neutral or slightly positive spherical aberrations. Toric IOLs should be considered for patients with stable and repeatable astigmatic patterns and a clearly determined axis, particularly for those who are not planning to wear contact lenses postoperatively. In a retrospective multicentre study, Ton et al. 4 demonstrated that toric IOL implantation significantly improved visual acuity and decreased astigmatism in keratoconic eyes with a topographic central, relatively regular astigmatic component. Accurate keratometry and astigmatism axis determination from multiple devices are essential. If a toric IOL is considered, it is beneficial to evaluate the astigmatic correction against the patient’s former glasses prescription (ideally before cataract development).
For patients with severe keratoconus who are intolerant to contact lenses and unsuitable for a toric lens, small-aperture IOLs were recently explored. These lenses are off-label in the United States for keratoconus patients and were not FDA-approved for this specific indication. These lenses mitigate peripheral distortions through the pinhole effect, potentially improving visual acuity in eyes with irregular corneas. 48 In a recent study, van den Berg et al. 48 demonstrated that simulated small-aperture optics significantly reduced higher-order aberrations in keratoconus eyes—up to Amsler-Krumeich stage 4—to levels comparable with healthy eyes, with clinical improvement in visual acuity also reported. In a prospective study by Shajari et al., 49 it was found that implantation of a small-aperture IOL in 17 eyes with severe corneal irregularities led to statistically significant improvements in both corrected and uncorrected visual acuity, along with high patient satisfaction. Additional studies are underway, and further research is needed to establish their safety and efficacy in these eyes. Surgeons should weigh the potential benefits against regulatory restrictions and thoroughly discuss off-label use with the patient during informed consent.
Intraoperative visualization challenge
The highly irregular and ectatic corneal surface, combined with poor tear film distribution in keratoconus, often results in significant image distortion during surgery and can impair depth perception. 50 To improve visualization, frequent irrigation with a balanced salt solution or the application of viscoelastic agents to coat the cornea can be beneficial. Adjusting surgical lighting to reduce glare further aids in maintaining a clear operative field. In addition, staining the anterior capsule with VisionBlue® (trypan blue dye, D.O.R.C. Dutch Ophthalmic Research Center, Zuidland, The Netherlands) improves visibility and enhances contrast, and may help make capsulorhexis formation safer and more precise in selected cases.
Multidisciplinary collaboration
Collaboration with optometrists is highly recommended, given the potential postoperative refractive challenges in keratoconus patients. Performing accurate subjective refractions in these cases can be particularly difficult due to the irregular corneal surface, often requiring experienced optometrists to dedicate additional time to achieve precise measurements. Preoperative refractions play a critical role in refining the surgical plan, confirming the astigmatism axis, and assessing the patient’s suitability for contact lenses after surgery. Postoperative subjective refractions are an essential tool for evaluating the accuracy of IOL power predictions and addressing patients’ residual refractive errors, which significantly contribute to their overall satisfaction.
Rigid lenses remain a common option for keratoconus patients to achieve optimal vision correction. For patients unsuitable for toric intraocular lenses, it is crucial to inform them that they may still require rigid gas permeable (RGP) or scleral contact lenses postoperatively to correct residual refractive errors and irregular astigmatism. Including these lenses in the postoperative treatment plan is particularly important for patients accustomed to them prior to surgery and wishing to maintain using them postoperatively. A collaborative approach between surgeons and optometrists ensures that the patient’s visual needs are met, providing comprehensive care for this complex patient population.
Conclusion
Cataract surgery in patients with keratoconus presents unique challenges related to corneal irregularity, measurement variability, and IOL power calculation, as well as intraoperative and postoperative management. Based on recent literature, the Barrett True-K (BTK) formula with measured posterior cornea demonstrates the highest overall accuracy among currently available options for keratoconus. However, prediction error still increases with advancing disease for all formulas.
This review provides an overview of the most current evidence on IOL power calculation in keratoconus and translates it into actionable clinical guidance. We emphasize the importance of assessing disease severity and stability as part of individualized preoperative planning and provide structured recommendations for formula selection, refractive targeting, and patient counseling. Practical tools—including a decision-making flowchart and summary tables—are included to assist surgeons at each stage of care, from preoperative evaluation through postoperative follow-up.
A comprehensive preoperative evaluation, appropriate formula selection, incorporation of posterior corneal measurements, and careful management of patient expectations can significantly improve refractive outcomes and patient satisfaction. While outcomes in these complex patients have vastly improved, further studies are needed to refine keratoconus-specific calculations and validate approaches across different severity levels and technologies.
