Abstract
Background:
Micro-pulse transscleral cyclophotocoagulation (MP-TSCPC) has continued to gain popularity as a treatment in adult glaucoma patients. Thus far there is limited evidence reporting the efficaciousness and safety of retreatment.
Objective:
To evaluate safety and efficacy of primary and repeat MP-TSCPC procedures.
Methods:
Thirty-four of 67 eyes who failed to achieve target IOP from initial MP-TSCPC underwent repeat MP-TSCPC and followed for a minimum of 6 months. All treatments were performed using the laser power of 2000 or 2250 mW, duration of 100–200 s, and a velocity 16–20 s per hemisphere swipe. Success criteria were defined as intraocular pressure (IOP) reduction of greater than 20% from baseline or any medication reduction without additional glaucoma procedures at 6 months after repeat MP-TSCPC. The 6-month success rate after repeat MP-TSCPC was also compared to that of initial MP-TSCPC in the same group of eyes.
Results:
Mean baseline IOP before the repeat MP-TSCPC was 23.0 + /- 5.3 on 3.0 + /- 1.4 medications. At 6 months, mean post-op IOP was 18.2 + /- 5.4 (21.9% reduction, p < 0.002), with mean medication staying relatively the same (p = .976). Success rate was increased from 23.5% to 44.1% with the repeat procedure compared to that of initial procedure (p = 0.123). Mean IOP reduction was also greater after repeat MP-TSCPC (18.7%, p < 0.002) when compared to initial MP-TSCPC (10.4%). No adverse events occurred.
Conclusions:
MP-TSCPC is a safe and effective non-invasive means to lower IOP in a variety of glaucoma patients. While over 50% (34/67) of eyes required repeat MP-TSCPC, repeat treatment resulted in greater success rates and IOP reduction without any adverse events when using the total energy between 112 and 150 J.
Introduction
Glaucoma, a progressive optic neuropathy, is a leading cause of irreversible, yet preventable blindness worldwide. 1 Progression of optic nerve damage can be slowed by reducing IOP.2,3 General methods for reducing IOP focus on decreasing aqueous humor production or increasing the outflow through the conventional and alternative outflow pathways. Current methods consist of pharmaceutical medications, laser treatment, micro-invasive glaucoma surgery, incisional surgeries, glaucoma drainage implants, and cycloablative procedures.2–4
Traditional laser ablation techniques, such as diode cyclophotocoagulation (CPC), utilizes an 810 nm continuous diode laser that is transmitted over the sclera and absorbed by the pigmented cells of the ciliary body epithelium. This results in thermal coagulative necrosis and irreversible destruction of the ciliary body, thereby decreasing the rate of aqueous humor production.5–9 Traditional transscleral cyclophotocoagulation is not considered a first-line treatment of glaucoma due to the high rate of post-operative complications such as hypotony, chronic uveitis, cystoid macular edema, decreased vision, or even phthisis bulbi.10–13 MicroPulse® Transscleral Cyclophotocoagulation (MP-TSCPC) is a variation of the traditional in the application of CPC.2,6,14 In contrast to CPC, MP-TSCPC operates using short ‘on’ and ‘off’ cycles. During the ‘on’ cycle, the thermal energy from the laser is absorbed by the pigmented epithelium of the ciliary bodies. In an ‘off’ cycle the pigmented tissue temperature drops below the thermal threshold and the tissue is allowed to cool. 9 The cooling of the tissue between pulses prevents thermal damage and destruction of adjacent ciliary tissue, lowering the risk of post-operative complications associated with conventional continuous-wave laser photocoagulation.6,7,15
The subthreshold technology MP-TSCPC makes this laser a safe and effective treatment for patients with various types and severity of glaucoma with minimal risk of complications.2,6,10,14 Long-term follow-ups after MP-TSCPC treatment has been reported to be successful in numerous studies; however, data on the repeat rate and effect of multiple treatments with this laser is lacking.2,6,7,16,17
This study aims to determine the efficacy, safety, and survival of repeat rate of MP-TSCPC. We followed all patients who received initial MP-TSCPC for a minimum of 12 months, and additional 6 months for those who required a repeat treatment. Efficacy and safety outcomes of the repeat treatment were compared to initial treatment in the same eye.
Methods
We conducted a retrospective chart review on patients who received first-time MP-TSCPC (MP-TSCPC1) and repeat procedure (MP-TSCPC2) at the Mason Eye Institute at the University of Missouri between October 2016, and June 2019. This study was approved by the Institutional Review Board (approval code 2016702) from the University of Missouri and adhered to the tenets of the Declaration of Helsinki. The requirements for informed consent were waived due to the retrospective nature of this study. Only those with a minimum of 12-month postoperative follow-up for initial treatment and a minimum of 6 months follow-up after repeat treatments, if required, were included. A subgroup of eyes that did not achieve target IOP from the initial MP-TSCPC group, underwent repeat MP-TSCPC. Target IOP that deemed repeat MP-TSCPC was determined by the treating ophthalmologist based on the individual patient’s goal, adherence, the disease severity, and the rate of progression of glaucoma. The outcomes of the repeat MP-TSCPC at 6 months were compared to initial MP-TSCPC at 6 months. Best-corrected visual acuity (BCVA), Goldmann applanation IOP, glaucoma medications, complications, and subsequent IOP-lowering interventions were collected at the preoperative visit and postoperative follow-ups at months 1, 3, 6, and 12 for initial MP-TSCPC, and months 1, 3, and 6 for repeat MP-TSCPC. The primary outcome measure was postoperative success, defined as ⩾ 20% reduction in IOP or reduction of any medications without additional IOP lowering procedures at 6 months for the initial MP-TSCPC group. For the repeat MP-TSCPC group (MP-TSCPC2) primary outcome was postoperative success, defined as ⩾ 20% reduction in IOP or reduction of any medications without additional IOP lowering procedures at 6 months. For this repeat subgroup (MP-TSCPC2), we compared the postoperative success at 6 months after repeat MP-TSCPC to their outcomes at 6 months after initial MP-TSCPC (MP-TSCPC1). Secondary outcomes measures included % of eyes reaching IOP ⩽ 18 mmHg at 12 months after initial MP-TSCPC, postoperative success at 12 months for initial MP-TSCPC, mean IOP and medication reduction, and time to additional IOP-lowering procedure. Complications including loss of 2 or more lines in BCVA, hypotony, uveitis, cystoid macular edema, corneal edema, and phthisis bulbi were also monitored.
All patients were given a peri-bulbar block consisting of 2% lidocaine with epinephrine mixed with 0.5% bupivacaine. A generous amount of lidocaine gel was applied as a coupling agent and topical anesthesia. MicroPulse® P3 glaucoma device (IRIDEX IQ810 Laser Systems, Mountain View, CA, USA) with a laser diode of 810 nm infrared wavelength and a duty cycle of 31.33% (micropulse ‘on’ 0.5 ms, ‘off’ 1.1 ms) was used for all patients. Patients received a power of 2,000 or 2,250mW and a duration of 100 to 200 s for initial treatment based on surgeon discretion. All patients with repeat treatment received 2250 mW and 180-s duration. Dwell-time was 16-20 s per hemisphere swipe, avoiding the 3 and 9 o’clock positions, areas of scleral thinning, sites of failed filtering blebs, and glaucoma drainage devices. All eyes were performed or directly supervised by one surgeon who has several years of experience in this procedure. Postoperatively, patients received Prednisolone acetate 1% 4 times a day for 1 week. Patients were instructed to continue all preoperative glaucoma medications, and they were discontinued one class at a time only after having reached the target IOP at or after the 1-month postoperative visit.
Data analysis was performed with SPSS v24 (SPSS Inc., Chicago, IL, USA). Paired t-tests were used to compare preoperative and follow-up IOP values. Wilcoxon signed-rank tests were used to compare preoperative and follow-up medications. Chi-square was used to compare initial and repeat MP-TSCPC success rates. All tests were two-sided, α = 0.05. We report all values as mean ± SD for the data with normal distribution unless otherwise specified. We used the Kaplan-Meier survival curve to demonstrate time to additional glaucoma procedures. Patients with missing data were included as censored cases in survival analysis.
Results
A total of 67 eyes of 55 patients (Table 1) received initial MP-TSCPC treatment. The patient sample was diverse including patients of all ages (range: 27–96 years old; average 70.5), 51% female, and most were with severe primary open-angle glaucoma (68.7%) (Table 1). Initial treatment led to success in 41.8% of eyes at 3 months and 16.4% at 12 months (Figure 1) per our definition of success. Over half of all eyes (58.2%) reached an IOP ⩽ 18 mmHg after initial MP-TSCPC at 12 months. Mean IOP reduction at 12 months was 21.2% (from 24.1 ± 8.2 mmHg to 18.1 ± 6.1 mmHg, p < 0.012) (Figure 2), and there was no significant medication reduction at any time points (p = 0.487 at 12 month).
Baseline demographic data for initial MP-TSCPC.

MP-TSCPC, Micropulse® P3 Cyclophotocoagulation; SD, standard deviation.
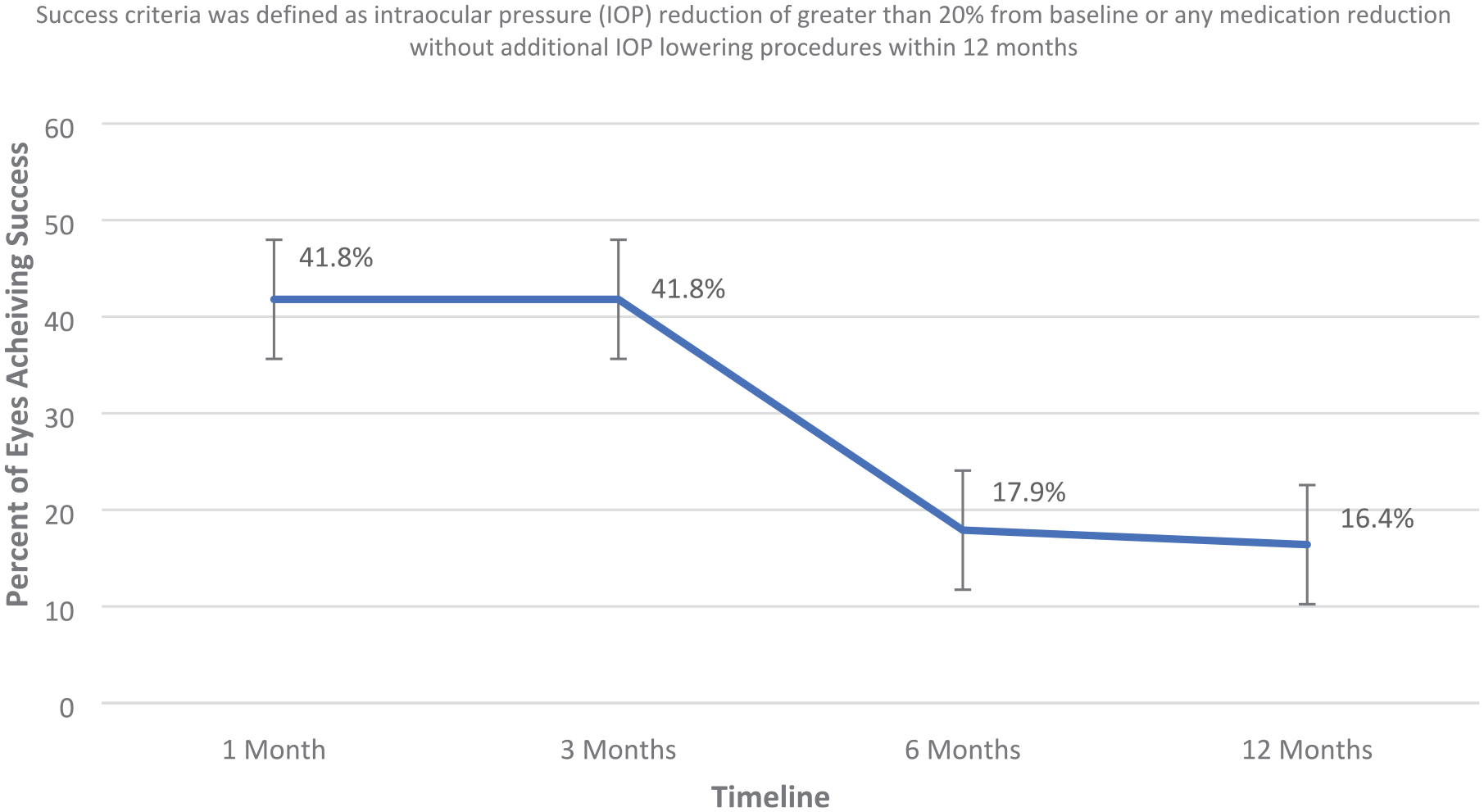
Percentage of eyes achieving success after initial micro-pulse transscleral cyclophotocoagulation (MP-TSCPC) throughout the 12-month postoperative period. Error bars reflect standard error the mean.

Mean intraocular pressure (IOP) from baseline (before treatment) throughout the 12-month postoperative period for initial micro-pulse transscleral cyclophotocoagulation (MP-TSCPC). Error bars reflect the standard error of the mean.
From the initial MP-TSCPC group, 34 eyes of 29 patients were retreated with MP-TSCPC due to the inability to maintain target IOP. Patient demographic data and distribution of glaucoma severity (Table 2) were similar to the initial treatment group (Table 1). Preoperative mean IOP for MP-TSCPC1 and MP-TSCPC2 was comparable at 23.0 ± 6.5 mmHg and 23.0 ± 5.3 mmHg, respectively (p = 0.717). Preoperative mean medication for MP-TSCPC1 and MP-TSCPC2 was also comparable at 3.1 ± 1.1 and 3.0 ± 1.4, respectively (p = 0.056). Success rate for repeat treatment at 6 months was 44.1%, compared to 23.5% after initial treatment (p = 0.123) (Table 3, Figures 1 and 3). Mean IOP significantly decreased from 23.0 ± 5.3 mmHg preoperatively to 18.2 ± 5.4 at 6-month after repeat treatment (p < 0.002) (Figure 4). Mean medication stayed relatively the same (p = 0.976).
Demographic data for repeat MP-TSCPC.

MP-TSCPC, Micropulse® P3 Cyclophotocoagulation; SD, standard deviation.
Comparison of post-operative IOP reduction, mean medication reduction, and success rates 6 months after initial and repeat micro-pulse transscleral cyclophotocoagulation (MP-TSCPC).

IOP, intraocular pressure; MP-TSCPC, Micropulse® P3 Cyclophotocoagulation; SD, standard deviation.
Success criteria was defined as intraocular pressure (IOP) reduction of greater than 20% from baseline or any medication reduction without additional IOP lowering procedures within 6 months of initial or repeat procedure.

Percentage of eyes achieving success after repeating micro-pulse transscleral cyclophotocoagulation (MP-TSCPC) throughout the 6- month postoperative period. Error bars reflect standard error the mean.

Mean intraocular pressure (IOP) from baseline (before repeat treatment) throughout the 6-month postoperative period for repeat micro-pulse transscleral cyclophotocoagulation (MP-TSCPC). Error bars reflect the standard error of the mean. p < 0.002.
No adverse events were noted, including any causes of 2 or more lines of BCVA, hypotony, or presence or sequelae of inflammation, such as cystoid macular edema.
Discussion
This study explores the long-term efficacy and safety of repeat rate MP-TSCPC in patients who have failed to maintain target after initial MP-TSCPC. While our study demonstrated initial MP-TSCPC has a moderate IOP lowering effect with a high repeat rate, we found that repeat MP-TSCPC is extremely safe and may lead to better outcomes compared to that of the initial treatment in the same eye. Our data demonstrated that patients who underwent repeat MP-TSCPC achieved a success rate of 44.1% at 6 months compared to a success rate of 23.5% in this same subgroup 6 months after initial MP-TSCPC.
There were difficulties in comparing our secondary outcome, 1-year success rates after initial MP-TSCPC, to other studies. This stemmed from varying definitions of success criteria, and varying laser parameters and techniques, including probe orientation, use of coupling agents, or dwell time.2,15–22 Our reported 1-year IOP reduction of 8.4 + 10.2 mmHg (21.2%) falls in the lower end of ranges reported in several other retrospective studies (range: 7.6–24.6 mmHg).2,6,16,21 Many of these studies involved patients with substantially higher baseline IOPs and lower medication needs, and the majority of our retreatment group comprised advanced glaucoma (76.5%) who may be more resistant to any treatment modalities. 6 However, when compared with studies with similarly reported mean baseline IOP and medications, our results fall in a similar range.16,21 Despite subtle differences in baseline IOP and definitions of success, all studies highlight the low side effect profile of MP-TSCPC, thereby supporting the safety and effectiveness of this novel non-invasive laser.2,6,14–18,20,21
Over 50% (34/67) eyes needed MP-TSCPC after initial MP-TSCPC alone. Despite the high attrition rate, effectivity and safety of repeat MP-TSCPC treatment have been rarely reported in the literature. Kuchar et al. reported a small sample (n = 3) that underwent retreatment and showed an increase in success rates (15.8%). In our study, eyes that underwent repeat MP-TSCPC achieved a significant IOP reduction of nearly 5 mmHg at 6 months (p < 0.002) without any adverse events. Furthermore, IOP reductions and success rates were greater when compared to that of initial treatment at 6 months despite their similar baseline characteristics. To understand if these effects are sustained beyond the 6-month period, a longer duration of follow-up time is needed. In addition, a prospective comparative study may be able to reveal the impact of timing of repeat MP-TSCPC on clinical outcomes and potential additive effects.
There are multiple variations of agreed-upon laser settings,2,13,22 which make it difficult to compare different studies. In our study, the range of total energy applied ranged from 112 J to 150 J. According to Sanchez et al., 19 this energy setting is within the range of having a good balance of efficacy and safety. With the recently revised probe design, it is estimated that energy delivery will improve significantly due to less technical variability. 22 Further studies are warranted to compare the long-term efficacy and safety results of those treated with a revised probe to our current study before making further recommendations with ideal energy settings.
Limitations of this study pertain to its retrospective design, variable power and duration settings used, and an overrepresentation of Caucasians (85.5%). The majority of the retreatment group was comprised of advanced glaucoma patients (76.5%) and may have contributed to the lower end of a successful outcome. Larger, prospective, randomized control studies with comparative controls, looking at the long-term effect of incremental repeat MP-TSCPC procedures using standardized energy settings are needed to better explore the efficacy and safety of repeat procedures, as well as possible predictive factors.
Nevertheless, our findings suggest that when compared to initial MP-TSCPC, repeat MP-TSCPC results in significant and greater IOP reduction with increasing success rates, without any vision-threatening complications associated with TSCPC. Due to the favorable safety profile, MP-TSCPC retreatment may be considered before proceeding with more invasive glaucoma procedures.
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Jella An has served as a consultant for Iridex. No involvement or research support was received from Iridex during this study. The authors report no other conflicts of interest in this work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
