Abstract
Purpose:
The purpose of this study was to evaluate the correlation between corneal astigmatism and the morphology of pterygium with anterior segment optic coherence tomography (AS-OCT).
Material and Methods:
The size of pterygium (horizontal length, vertical width) was measured manually; pterygium area and percentage extension of the pterygium onto the cornea were calculated. Anterior and posterior corneal astigmatism, Sim K, K1, K2 were measured using a dual Scheimpflug analyzer. Morphological patterns of the pterygium analyzed with AS-OCT were determined according to the extension of the pterygium apex below the corneal epithelium. Two tomographic patterns were identified: continuous and nodular. Correlation between anterior corneal astigmatism and pterygium size, percentage extension of the pterygium, and morphological pattern of the pterygium was analyzed.
Results:
The mean ages of the 47 patients were 49.4 ± 16.6 (22–80) years. Mean horizontal pterygium length, vertical width, pterygium area, and percentage extension of the pterygium were 2.8 ± 1.2 mm, 4.8 ± 1.6 mm, 7.42 ± 5.6 mm2 and 24.5 ± 10.4%, respectively. Mean anterior corneal astigmatism was 2.3 ± 2.3 D and simulated keratometry was 43.4 ± 2.02 D. In terms of the morphological pattern of the pterygium, 24 eyes had continuous, 23 eyes had a nodular pattern and the median (interquartile range) anterior corneal astigmatism was 1.87 (1.01–3.80) and 1.22 D (0.58–2.35), respectively (p = 0.102). Other topographic and pterygium size parameters were similar between groups. Analyzing the correlations in groups separately, a positive moderate statistically significant correlation was present between vertical width, percentage extension, pterygium area, and anterior corneal astigmatism in both continuous and nodular groups.
Conclusions:
Although not statistically significant, anterior corneal astigmatism was higher in continuous group. Using AS-OCT to standardize the morphology of pterygium could provide additional clinical information.
Introduction
Pterygium is a wing-shaped fibrovascular connective tissue that originates from the bulbar conjunctiva and extends toward the center of the cornea. The prevalence of pterygium has been reported to be 1.1–23.4%, depending on the population studied. 1 In addition to cosmetic problems and recurrent inflammation, it can also cause a significant visual disturbance.1,2 Pterygium can lead to considerable changes in corneal refractive status before it reaches the optical zone. It often induces with the rule corneal astigmatism due to the localized flattening of the area between the central cornea and the advancing end of the pterygium. 3 Numerous studies have investigated the effect of different sizes of pterygium on corneal refractive status measured by refraction, keratometry, and corneal topography.4–7 These studies revealed that larger sizes of pterygium have stronger correlations with corneal astigmatism.
There are a limited number of histological and clinical studies about the morphological patterns of pterygium.8–11 Anterior segment optic coherence tomography (AS-OCT) provides detailed information about the ocular surface and can be used to understand the anatomical relationship between pterygium and the cornea.9,10 In a study that used AS-OCT, the researchers described a thick hyperreflective layer between the corneal epithelium and Bowman’s layer in pterygium. 9 Gasser and colleagues used AS-OCT to identify the morphological parameters of pterygium according to the corneoscleral transition zone and anterior stromal scarring. They also evaluated the relationship between the AS-OCT features of the pterygium and visual acuity and astigmatism. 11 To our knowledge, a limited number of publications have investigated the correlation between corneal astigmatism and the morphology of pterygium. The present study aimed to use AS-OCT to evaluate how pterygium size and morphology affect corneal astigmatism.
Material and methods
This study was performed with the prior approval of the Sakarya University Institutional Review Board (71522473/050.01.04/81) and followed the tenets of the Declaration of Helsinki. Written informed consent was obtained from each subject.
This study was conducted at the Sakarya University Training and Research Hospital (Serdivan, Turkey) between April 2019 and February 2020. Data of patients diagnosed with pterygium and followed up in our cornea department were evaluated retrospectively. Forty-seven patients with primary pterygium were enrolled. Eyes that had recurrent pterygium, history of ocular surgery or trauma, corneal scarring, or irregularity influencing the corneal topography were excluded.
Comprehensive ocular examination, including demographic data, subjective refraction including spherical equivalent [spherical error plus (cylindrical error/2)], best-corrected visual acuity measurement with a Snellen chart, anterior and posterior segment examination with slit-lamp microscopy, keratometric measurements by corneal topography, and pterygium morphology analyzed with AS-OCT were performed in all subjects.
To assess the pterygium sizes the vertical width and horizontal length (millimeters) of the pterygium were measured manually with a millimetric scale of biomicroscope by one examiner (ED). The length between the apex of the pterygium and nasal limbus is recorded as horizontal length, and the line between the two limbal points where the pterygium intersects the limbus is recorded as vertical width (Figure 1). The percentage extension of the pterygium to the cornea was calculated by dividing the horizontal length of the pterygium by the diameter of the horizontal cornea.

Anterior segment image of the pterygium depicting pterygium sizes. Horizontal length was defined as the line between the apex of the pterygium and nasal limbus. Vertical width was defined as the dashed line between the two limbal points where pterygium intersects the corneal limbus.
For topographic measurements, a dual Scheimpflug analyzer (Galillei Ziemer Ophthalmic Systems AG, Port, Switzerland) was used and measurements obtained by a single experienced examiner. Images were confirmed under a quality-specification window and good-quality scans were included. Simulated keratometry (Sim K), flat (K1), and steep (K2) keratometry, anterior and posterior corneal astigmatism were obtained.
Cirrus HD-OCT (Carl Zeiss Meditec, Dublin, CA, USA) was used to obtain high-resolution anterior segment scans. AS-OCT images were taken under the same room conditions by the same experienced examiner. The high-resolution images of AS-OCT allow the examiner to evaluate the anatomical relationship between pterygium and cornea in detail. Two independent observers who were masked to participants’ clinical data evaluated images separately and analyzed all images similarly. Images were obtained with five-line raster horizontal OCT scans at three locations: limbus, central cornea, and apex of the pterygium tissue. Scans with the signal strength of ⩾8 were used for analysis. Scans with misalignment and poor illumination or those out of focus were excluded.
On AS-OCT, pterygium images are seen dense, hyperreflective subepithelial lesion located between the corneal epithelium and Bowman layer, covered with normal epithelium with various amounts of hyperreflectivity. Morphology of pterygium was evaluated according to the extension of the pterygium apex below the corneal epithelium. Two tomographic patterns were identified. The presence of extension and creeping of the end of the pterygium apex below the corneal epithelium was assessed as continuous. The presence of well-delineated masses of pterygium tissue and less creeping and less advanced extension of the apex below the corneal epithelium was assessed as nodular (Figure 2).

(a) Anterior segment photograph of pterygium with nodular pattern. (b) Anterior segment optical coherence tomography horizontal scans of nodular pattern with well delineated masses of pterygium tissue bulging the ocular surface contour and with less advanced and creeping extension of the apex below the corneal epithelium. (c) Anterior segment photograph of pterygium with continuous morphology. (d) Anterior segment optical coherence tomography horizontal scans of continuous morphology pattern with more extension and creeping of the end of the pterygium apex below the corneal epithelium.
Statistical analyses were performed using SPSS software version 21.0 (IBM® SPSS Statistics). The variables were investigated using analytical methods (Kolmogorov–Smirnov test) to determine whether they were normally distributed or not. Descriptive analyses were presented using medians and interquartile range (IQR) for the non-normally distributed variables, using means and standard deviations (SD) for the normally distributed variables. The Mann–Whitney U tests were utilized for comparing two non-normally distributed variables and the independent sample t test for normally distributed variables. Spearman correlation analysis was performed for evaluations of the horizontal length, vertical width, percentage extension (%), and corneal astigmatism values. A p value of less than 0.05 was considered statistically significant.
Results
The study comprised 47 patients (24 males and 23 females) and the mean ages of the patients were 49.4 ± 16.6 (22–80) years. The mean best-corrected visual acuity was 0.7 ± 0.3. The mean horizontal pterygium length ranged from 1.1 to 6.9 mm with a mean of 2.8 ± 1.2 mm and the vertical width ranged from 2.4 to 8.6 mm with a mean of 4.8 ± 1.6 mm. The pterygium area was 7.42 ± 5.6 mm2 and the percentage extension of the pterygium was 24.1 ± 10.6% (Table 1).
Clinical and topographic parameters of patients.
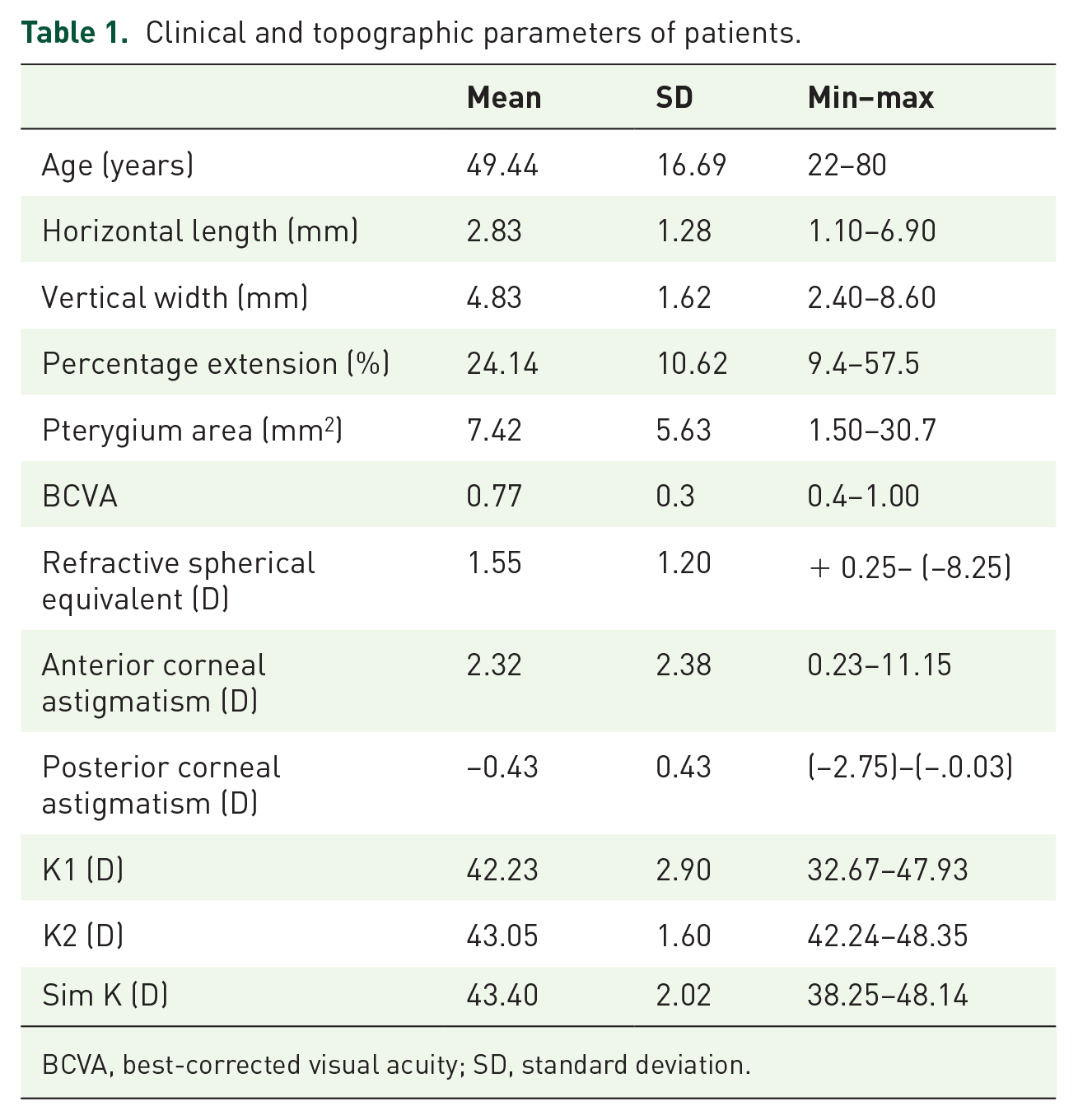
BCVA, best-corrected visual acuity; SD, standard deviation.
Mean anterior corneal astigmatism was 2.3 ± 2.3 D (0.2–11.1). With the rule astigmatism (axis from 0–30, 150–180) was the main type of astigmatism (29 eyes, 61.7%), followed by against the rule (axis from 60–120) (14 eyes, 29.7%) and oblique astigmatism (axis from 30–60 or 120–150) (4 eyes, 8.5%). Table 1 shows the mean values of the other topographic parameters.
In terms of the morphological pattern of the pterygium; 24 eyes had continuous, 23 eyes had the nodular pattern. The median (IQR) anterior corneal astigmatism was 1.87 D (1.01–3.80) and 1.22 D (0.58–2.35), respectively (p = 0.102) (Figure 3). The median (IQR) posterior corneal astigmatism was −0.34 D (−0.53–(−0.14)) and −0.30 D (−0.42–(−0.24)), respectively (p = 0.925) Demographic, clinic, other topographic, and pterygium size parameters were similar between groups (Table 2).

Distribution of corneal astigmatism according to pterygium morphology.
Comparison of parameters according to pterygium morphology.
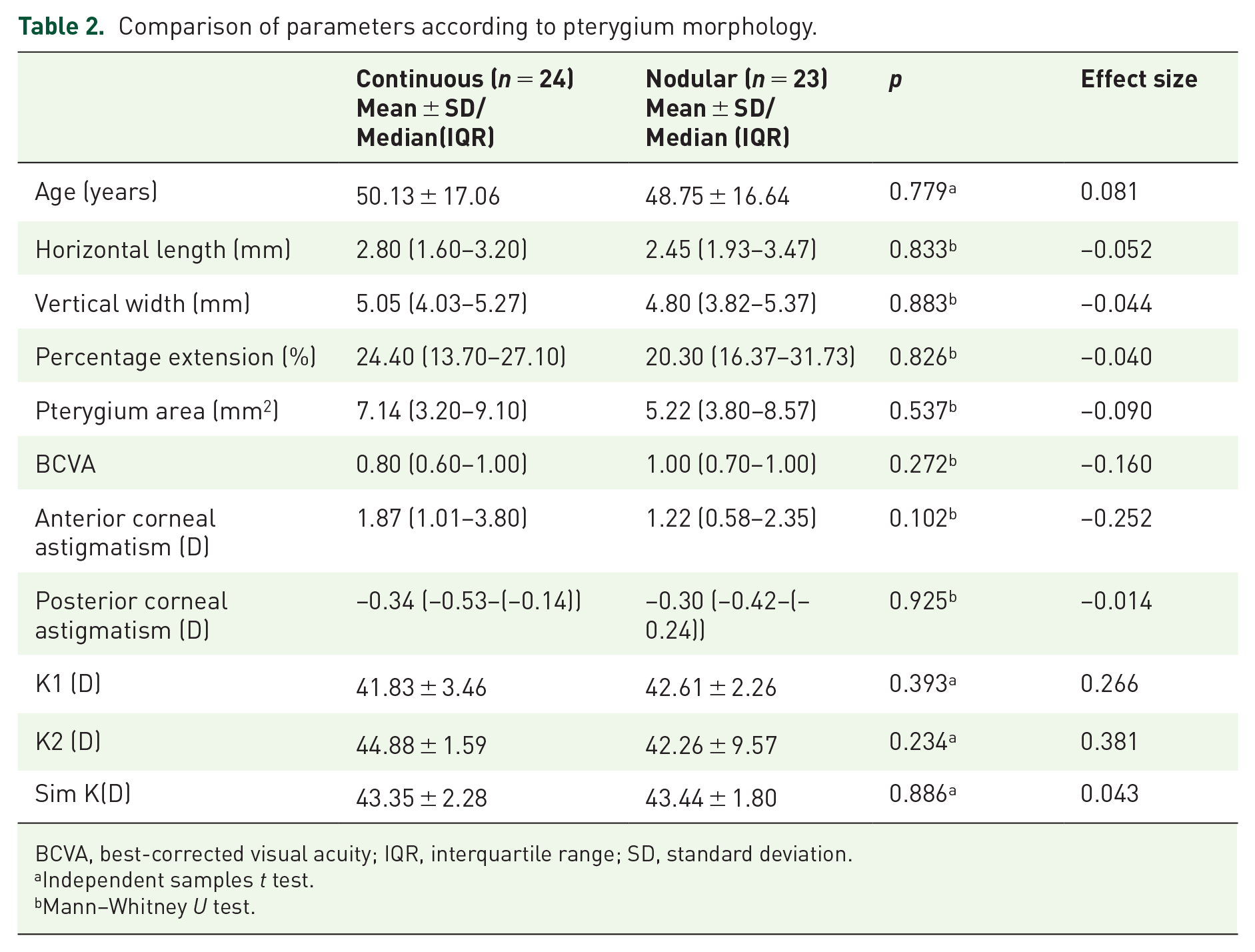
BCVA, best-corrected visual acuity; IQR, interquartile range; SD, standard deviation.
Independent samples t test.
Mann–Whitney U test.
Spearman correlation analysis was used to study the correlation between pterygium sizes and anterior corneal astigmatism. There was a positive moderate linear correlation between anterior corneal astigmatism and pterygium horizontal length (p < 0.001 r = 0.549), vertical width (p = 0.013, r = 0.382), percentage extension of the pterygium (p < 0.001 r = 0.573), pterygium area (p < 0.001, r = 0.582). Among them, percentage extension, pterygium area, and horizontal length of pterygium have the most significant correlation (Figure 4).

Correlation analysis between pterygium parameters and corneal astigmatism.
Analyzing the correlations in groups separately, a positive moderate statistically significant correlation was present between vertical width, percentage extension, pterygium area and anterior corneal astigmatism in continuous group (p1 = 0.032, r1 = 0.508; p2 = 0.048, r2 = 0.451; p3 = 0.025, r3 = 0.501, respectively). Similarly there was a positive moderate statistically significant correlation between horizontal length, vertical width, percentage extension, pterygium area, and anterior corneal astigmatism in nodular group (p1 = 0.004; r1 = 0.597; p2 = 0.041, r2 = 0.449; p3 = 0.006, r3 = 0.581; p4 = 0.007, r4 = 0.569, respectively) (Table 3).
Correlation between corneal astigmatism and pterygium size and morphology.

Discussion
The results of this study demonstrated that while increased pterygium sizes are related to higher anterior corneal astigmatism, the morphology of pterygium does not have a significant effect. A majority of the studies in the literature have focused on visual and topographic changes induced by pterygium.3–7
Pterygium leads to focal flattening of the central cornea due to mechanical traction of the pterygium or pooling of tears at the apex of the pterygium. 12 Numerous studies have determined a significant association between corneal refractive status and the width, length, and total area of pterygium. Similar to our results, previous studies have demonstrated that larger sizes of pterygium are associated with pterygium-induced corneal astigmatisms.3,5,7 Han and colleagues 3 reported that pterygium-induced corneal astigmatism have a stronger correlation with the horizontal length of pterygium than the width and total area. Mohammad-Salih and colleagues reported that extension of pterygium had the strongest significant correlation with corneal astigmatism, followed by the total area of pterygium. They also reported that pterygium can lead to ⩾2 D corneal astigmatism when its width is ⩾5 mm, its length is ⩾2.2 mm, or its total area is ⩾6.2 mm2. 7 In the present study, percentage extension, pterygium area, and horizontal length of pterygium had the most significant correlation.
To our knowledge, a limited number of studies have used AS-OCT to analyze how pterygium morphology affects corneal topography. The primarily histopathological studies that have been performed to determine pterygium morphology have demonstrated fibrovascular proliferation of the conjunctival connective tissue with elastotic degeneration of collagen situated below the corneal epithelium.8,13 Some of these studies determined a correlation between the histopathologic changes and clinical findings, reporting that larger pterygium, which has increased fibrovascular tissue, were associated with increased redness and fleshiness.13,14 Safi and colleagues 14 reported that the horizontal length and area of pterygium had a significantly positive correlation with vascular density and a negative correlation with stromal elastosis.
AS-OCT is an important diagnostic tool that can enable in vivo histological evaluations of the anterior segment and provide detailed images of the ocular surface, especially the cornea. 15 Although studies using AS-OCT have identified the morphological patterns of pterygium, pinguecula, and pseudopterygium,10,11 there is currently no systematic classification about the anatomical relationship between pterygium and the cornea. Soliman and colleagues defined primary pterygium as well-delineated, wedge-shaped masses that coming from the conjunctival side, creeping under the corneal epithelium and separating it from underlying wavy and irregular Bowman’s membrane. They reported various types of pterygium morphology, such as scarring at the superficial corneal stroma and the presence of satellite masses that extend under the epithelium beyond the pterygium margins. They also determined that the apex of pterygium in recurrent pterygium cases is more advanced and creeping. 10 However, in this study there was no data about the effects of pterygium morphology on clinical findings.
In another study, Gasser and colleagues evaluated the AS-OCT parameters of pterygium that may be associated with clinical findings. They defined pterygium morphology according to the corneoscleral transition zone. The appearances of a nodular mass bulging the ocular surface contour or flat bridging corneoscleral transition zone were not altered ocular surface contour. They also assessed histological sections of the pterygium tissue and suggested that limbal nodularity was caused by the accumulation of elastotic degenerative changes at the limbus during the early stages of pterygium and bridging into the peripheral cornea during the advanced stages of pterygium. Finally, they reported that the flat corneoscleral transitional zone was associated with higher astigmatism and increased corneal scarring. 11
In the present study, we identified two pterygium patterns according to the extension of the tip of the pterygium apex below the corneal epithelium. We think that this continuous morphology may be indicative of pterygium’s advanced stages. The median posterior corneal astigmatism was similar between groups and it was not affected by the pterygium morphology. The median anterior corneal astigmatism was 1.87 D in the continuous group and 1.22 D in the nodular group. Although anterior corneal astigmatism was higher in the continuous group, this finding was not statistically significant. This discrepancy may have been caused by the increased force of horizontal traction that was exerted onto the cornea by the contractile elements within the pterygium tissue. Anterior corneal stromal scarring with partially destroyed Bowman’s membrane may have also caused this issue. This morphological analysis can be used to make surgical decisions. Surgeries can be performed earlier in cases with continuous morphology where higher astigmatism is expected.
This study has some limitations. Although measurements were performed by the same examiner repeatedly under the same conditions, objective measurements may not have been achieved when the examiner performed their measurements manually. Another limitation of this study was the small number of patients. More precise results can be attained with groups involving more patients. In addition to the morphological analysis, measurement of the invasion depth of the pterygium at the limbus or pterygium apex could be performed to provide additional information. Moreover, future studies could investigate pterygium morphology that has been provided by AS-OCT to evaluate recurrent pterygium cases retrospectively.
In conclusion, the current study’s results revealed that increased pterygium sizes are associated with higher anterior corneal astigmatism. Although the continuous group had higher astigmatism than the nodular group, this finding was not statistically significant. Using AS-OCT to evaluate the morphology of pterygium can provide important information. Studies with larger sample sizes are needed to assess the relationship between pterygium and the cornea with AS-OCT.
Footnotes
Compliance with ethical standards
This study was performed with the prior approval of the Institutional Review Board (71522473/050.01.04/81).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
