Abstract
Purpose:
To evaluate the relationship between measurements and various morphometric parameters of primary pterygium on anterior segment optical coherence tomography(AS-OCT) and refractive and keratometric astigmatism.
Methods:
In this cross-sectional study, patients with primary pterygium were included. The AS-OCT parameters of pterygium consisting of epithelial thickness near apex(µm), apical or head thickness(µm), its thickness at limbus(µm), horizontal length of pterygium(mm), central corneal thickness (µm) and maximum pterygium thickness(mm) were evaluated.
Results:
Sixty three patients comprising of 63 eyes with a primary pterygium were studied. The mean keratometric and refractive astigmatism were 1.69 ± 3.15 Diopter(D) and0.5 ± 1.4D, respectively. There was a significant association between pterygium grade and stocker’s line with keratometric astigmatism (p = 0.02, 0.00) respectively. Vertical and horizontal length on slit lamp and horizontal length on AS-OCT showed significant association with keratometric astigmatism (p = 0.05, 0.00,0.00), respectively. Limbal thickness on AS-OCT showed significant positive correlation with refractive astigmatism (r = 0.29, p = 0.02).
Conclusion:
The pterygium grade influences the keratometric astigmatism. Increased limbal thickness of pterygium on AS-OCT leads to more refractive astigmatism.
Keywords
Introduction
A pterygium is a fibroelastic degeneration which is triangular, fibrovascular connective tissue overgrowth of bulbar conjunctiva encroaching onto the cornea. It may have an effect on corneal curvature and cause irregular astigmatism or dellen formation. Therefore, sometimes surgical removal becomes necessary before the lesion advance close to the visual axis. High-grade corneal astigmatism is considered as an indication for pterygium surgery.1,2 Pterygium causes corneal distortion and induces significant amount of astigmatism.3,4 This astigmatism may occur either due to traction generated by the pterygium, pulling on and distorting the cornea, or by tears pooling near apex of the pterygium or both. 5
The effect of pterygium on the corneal refractive status has been measured by refraction, keratometry, corneal topography and computerized video keratoscopy.3,6,7 Pterygium leads to astigmatism and exhibit signs of limbal stem cell deficiency, chronic inflammation, basement membrane destruction and fibrous ingrowth. 8
It is mandatory to maintain the proper documentation of the dimensions of pterygium in all directions like length, width and depth in order to detect its growth and recurrence. As compared to slit lamp bio-microscopy, anterior segment optical coherence tomography (AS-OCT) provides more precise and reproducible measurements of pterygium. 9 On AS-OCT the pterygium is seen as a highly hyper-reflective demarcated fibrillary layer separating the corneal epithelium and stroma, which represents Bowman’s membrane (BM) disruption with scarring of anterior stroma. 10 On the other hand, an intact BM with overlying normal epithelium can also be noticed. 11 The overlying epithelium may be normal, thinned out or thickened.12,13
As the pterygium causes with the rule astigmatism which depends upon the pterygium size both length and width. Furthermore, the astigmatism is directly proportional to its corneal encroachment with respect to its measurements. 14 AS-OCT gives significantly more reproducible results than the slit lamp beam for measurements of the distance of a pterygium’s apex from the limbus. 15 AS-OCT can quantify the depth of anterior stromal scarring at the apical area of pterygium as well as at corneo-scleral junction (limbus). This precise measurements and quantification helps in assessment, monitoring and planning the surgery. This study was done to evaluate accurate dimensions of extension of pterygia onto the cornea and the relationship between various morphometric parameters of primary pterygium on AS-OCT and refractive and keratometric astigmatism.
Methods
In this cross-sectional study, 63 eyes of 63 individuals with primary pterygium with age > 18 years were included in the study. Exclusion criteria consisted of patients with pseudo-pterygium which is conjunctival adhesion to cornea due to limbal or corneal inflammation or trauma. A probe can be passed underneath this which can’t be passed under true pterygium due to its adherence to underlying epi-sclera and the sclera throughout the extent of the growth. Other exclusion consisted of acute ocular surface inflammation or disease, any history of previous ocular surgery, ocular trauma, ocular infection, severe dry eye and known cases of limbal stem cell deficiency as these conditions can alter the outcome of the study. The patients who were not willing were also excluded. In addition, various parameters were noted which included the age, sex, pterygium grade, its type(progressive/regressive), associated stocker line, best corrected visual acuity (BCVA), subjective refraction, central corneal thickness (CCT), nasal limbal corneal thickness, astigmatism by auto-refractometer and manual keratometry for refractive and keratometric astigmatism with axis. Corneal astigmatism was measured with either an optical keratometer (Javal-Schiotz) or auto-keratometer as in some cases manual keratometry was not possible, auto-keratometer was used in such cases.
On slit lamp examination, standard magnification of 16X was used to measure various parameters of the pterygium from the limbus to the apex. Size of pterygium with respect to both length and width at limbus was measured on slit lamp examination. Readings were taken from the slit lamp scale and were recorded to the nearest 10th of a millimeter. Various measurements of pterygium were taken by using Fourier-Domain Optical Coherence Tomography (FD-OCT) system RTVue (Optovue, Inc, Fremont, CA,USA.) by using a Corneal Adaptor Module (CAM) consisting of wide-angle lens which provides a scan of width up to 6 mm, and a transverse resolution of 15 µm. CAM helps in telecentric scanning of anterior segment. All tests were performed in a dim lit room between 21–250C with regulated humidity to avoid reflex tearing. Patients were asked to look straight ahead at the fixating target within the OCT system. The patient’s head was stabilized by a chin/forehead rest and patient’s gaze was fixed by an internal target. Two gooseneck lights were used to illuminate the anterior segment for concurrent video imaging and fixation of the contra-lateral eye. Any contact of the scanning module with the patient’s eye was avoided. The AS-OCT parameters of pterygium measured were epithelial thickness near apex(µm), apical or head thickness(µm), its thickness at limbus(µm), horizontal length of pterygium(mm), CCT (µm) and maximum pterygium thickness(mm) were measured by using pachymetry scan, CL- Crossline(6 mm) and CL-Raster (6 mm X 4 mm) in horizontal scan. BM disrupted or not disrupted was also recorded. In CL- raster four high-resolution optical sections separated by 450 showing seventeen images within 6 mm were seen. The images captured were analyzed for computer calipers measurements. Correct meridian to image the lesion was at 180 degrees and 90° clockwise canvas rotation was done to obtain a vertical visualization of the anterior segment section. The cross-sectional AS-OCT images were processed and exported for computer calipers measurements and calculated with integrated analysis custom software in µm which were further calculated in unit of mm.
Each pterygium was independently measured and recorded on slit lamp and AS-OCT by single observer to avoid measurement bias. Grading of pterygium was done by TAN’S classification on slit lamp examination as follows:
Grade I: Atrophic pterygium, which extends till 2 mm onto the cornea along with clearly visible epi-scleral blood vessels under the pterygium.
Grade II: Intermediate pterygium, which involves 4 mm of the cornea with partially visible episcleral blood vessels under the pterygium.
Grade III: Progressive pterygium, which involves more than 4 mm of the corneal epithelium, which can eventually hamper the visual axis and the epi-scleral blood vessels are obscured under the pterygium. 16
On AS-OCT the length of the pterygia was measured using the inbuilt software with calipers as the distance from limbus to the apical part of pterygium. Apical thickness was the areas of hyper-reflectivity just at apex or head of pterygium. Epithelial thickness was measured just ahead of apical part of pterygium before sloping down to the cornea. Limbal thickness was defined as the distance between the scleral and the ocular surface at the corneo-scleral junction. The status of the BM was observed as Intact or non-intact depending upon the hyper-reflectivity if it was observed beneath the BM then it was considered to be disrupted otherwise intact. CCT was measured by pachymetry scan. Maximum pterygium thickness or depth was measured as maximum depth of anterior stromal hyper-reflectivity underneath the body of pterygium. BCVA was noted on Snellen’s chart and was converted using a decimal scale which is Logarithm of Minimum Angle of resolution (LogMAR). The refractive astigmatism was obtained by the refraction, retinoscopy or autorefractometer. The keratometric astigmatism was obtained by manual and auto-keratometer. An average three readings were taken to confirm the final reading on automated keratometry. Refractive and kerato-metric astigmatism were further divided in 3 groups as ⩽ 1, 1–3,>3 in diopters (D)
Statistical analysis
Initially data obtained were entered into an excel spreadsheet and then transferred to SPSS software (Statistical Package for Social Sciences, version 22, SPSS Inc, Chicago, IL) for analysis. Statistical data were expressed in terms of means ± standard deviations (mean ± SD).
The descriptive statistics was used to express data in terms of frequency and percentage. A two tailed test was used for all measurements. Chi-square test was used to find out the association between continuous variables like apical epithelial thickness, apical or head thickness(µm), limbal thickness, horizontal length, CCT and maximum pterygium thickness on AS-OCT with the refractive and keratometric astigmatism.
The differences among three groups between the means of (Refractive astigmatism ⩽ 1, 1–3,>3 and keratometric astigmatism ⩽ 1, 1–3,>3) in diopters(D) and various pterygium variables were compared by one-way analysis of variance (ANOVA). p value < 0.05 was considered statistically significant.
Results
Sixty three patients comprising 63 eyes with pterygia, maximum 20 (68.3%) were males and 43 (31.7%) were females with a mean age of 50.35 ± 14.68 years (range of 20–71 years). Mean BCVA was 0.2 ± 0.8 (decimal scale). Predominantly grade 2 pterygium was seen in 40 (63.5%) and 45 (71.4%) had progressive pterygium. Morphologically two types of pterygia were seen as flat and nodular in 43 (68.25%) and 20 (31.74%) cases, respectively. Stocker’s line was absent in 46 (73%) patients. BM was not intact in maximum 38 (60.3%) patients. Maximum cases showed vertical length between 3 and 4 mm in 33 (52.4%). Maximum cases showed horizontal length between < 3 mm in 47 (74.6%). With the rule astigmatism was seen in maximum 43 (68.3%) cases. Predominantly, 34 (54%) and 35 (55.6%) cases showed refractive and keratometric astigmatism, respectively, of ⩽ 1 D each (Table 1). The mean difference between horizontal and vertical keratometry was 1.69 ± 3.15 (D) with a 47.37 ± 32.70° deviation from the horizontal meridian. On AS-OCT in the horizontal OCT scans parallel to the axis of the pterygium growth, primary pterygia revealed wedge-shaped mass separating the cornea epithelium from the underlying BM with an elevated corneal epithelium which became irregular, wavy and interrupted (Figure 1). Progressive and regressive pterygia on AS-OCT were seen as in Figure 2(a) and (b). On AS-OCT the mean epithelial thickness, apical or head thickness and limbal thickness were 57.59 ± 7.65 µm, 124.06 ± 53.42 µm and 455.78 ± 188.34 µm, respectively. The mean keratometric and refractive astigmatism were 1.69 ± 3D, 150.5 ± 1.4D, respectively. Mean CCT and maximum pterygium thickness were 511.56 ± 31.21 µm and 1.07 ± 0.12 mm, respectively (Table 2). On AS-OCT two distinct morphological appearances of pterygium were seen either flat corneoscleral transition, bridged this zone or the nodular appearance with bulged ocular surface contour.
Depicting baseline pterygium characteristics and demographics.

AS-OCT, anterior segment optical coherence tomography.
Various pterygium parameters on AS-OCT.

AS-OCT, anterior segment optical coherence tomography; BCVA, best corrected visual acuity.

Anterior segment optical coherence tomography showing morphological features showing epithelium, stroma, Bowman’s membrane and wedge shaped hyperreflective area of pterygium.

(a) Showing regressive pterygium on slit-lamp with flat corneoscleral junction on AS-OCT. (b) Showing progressive pterygium on slit-lamp and nodular morphology of pterygium at limbus on AS-OCT.
Relationship of various pterygium parameters with keratometric and refractive astigmatism
There was a significant association between pterygium grade and stocker’s line with keratometric astigmatism with p value of 0.02 and 0.00, respectively. Vertical and horizontal length on slit lamp and horizontal length on AS-OCT showed significant association with keratometric astigmatism (p value 0.05, 0.00 and 0.00), respectively (Table 3). The horizontal length on slit lamp and on AS-OCT showed significant association with refractive astigmatism (p = 0.00) each (Table 4).
Relationship of various pterygium parameters on AS-OCT with keratometric astigmatism.

AS-OCT, anterior segment optical coherence tomography.
p* value calculated by chi-square test.
Depicting the relationship between various pterygium parameters on AS-OCT with refractive astigmatism.

AS-OCT, anterior segment optical coherence tomography.
p* value calculated by chi-square test.
Association of various qualitative pterygium parameters on AS-OCT with keratometric astigmatism and refractive astigmatism
The ANOVA was non-significant for various pterygium variables like epithelial thickness, pterygium head thickness, limbal thickness and maximum thickness with the difference between the three different groups of keratometric astigmatism with p value of 0.35, 0.64, 0.88 and 0.46, respectively. This shows the existing difference between the pterygium variables and keratometric astigmatism groups ⩽ 1, 1–3 and > 3D (Table 5). These results allowed to reject the null hypothesis. Pterygium length had not shown any difference between the three different groups of keratometric astigmatism and refractive astigmatism ⩽ 1, 1–3 and > 3D each with significant p value 0.00 (Tables 5 and 6). Difference in between the three different groups of refractive astigmatism did not show any difference with respect to BCVA with p value of 0.05 (Table 6).
Association of various qualitative pterygium parameters on AS-OCT with keratometric astigmatism.

AS-OCT, anterior segment optical coherence tomography; BCVA, best corrected visual acuity; SD, standard deviation.
p# value calculated by one way ANOVA.
Association of various qualitative pterygium parameters on AS-OCT with refractive astigmatism.
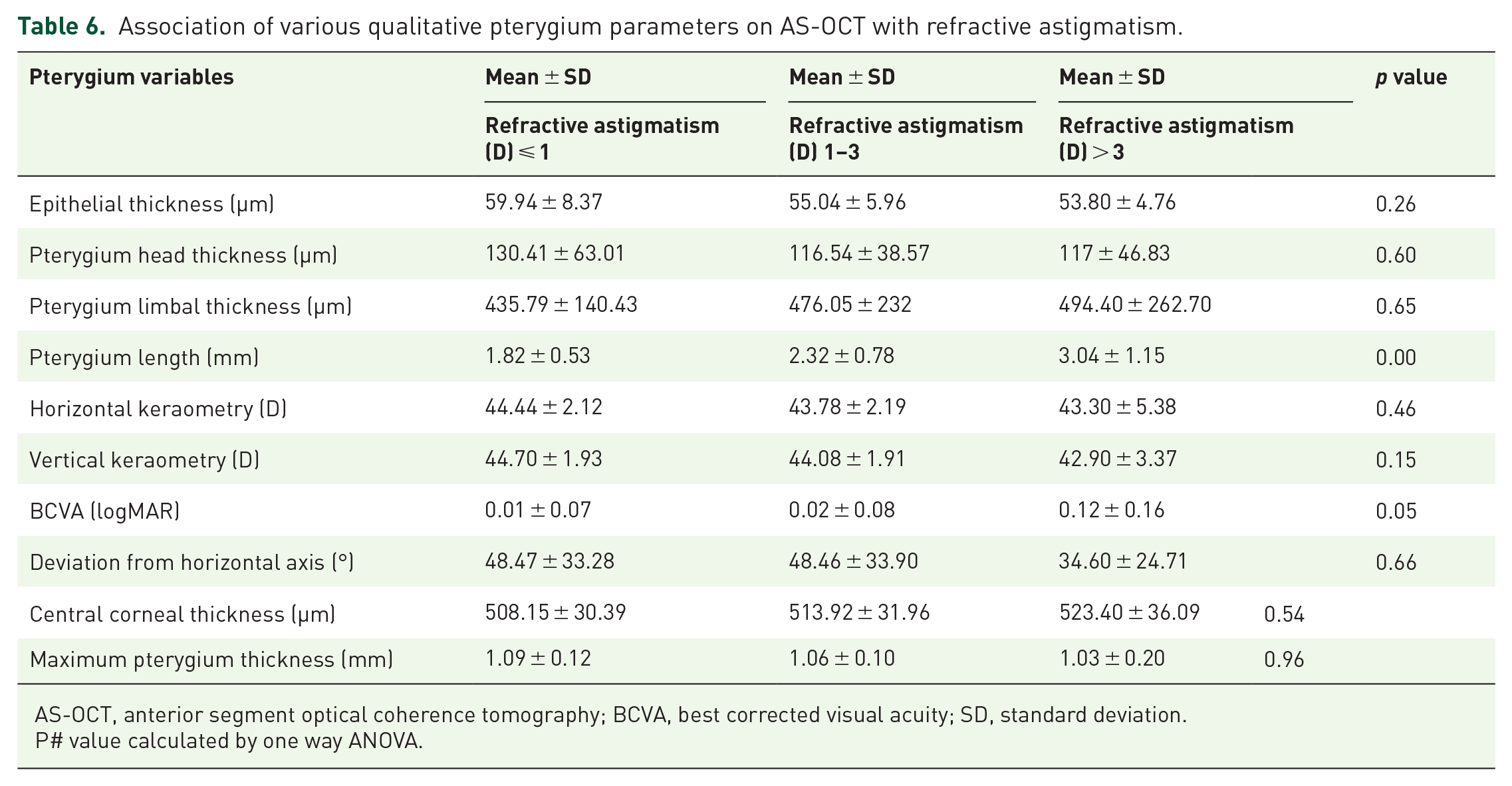
AS-OCT, anterior segment optical coherence tomography; BCVA, best corrected visual acuity; SD, standard deviation.
P# value calculated by one way ANOVA.
There was a significant positive correlation between anterior apical thickness and keratometric astigmatism (r = 0.25, p = 0.04). Limbal thickness on AS-OCT showed significant positive correlation with refractive astigmatism (r = 0.29, p = 0.02). Epithelial thickness showed significant positive correlation with horizontal and vertical keratometry (r = 0.32, p = 0.01) each.
Discussion
In the present study, AS-OCT has been used to evaluate the morphological parameters of pterygium and their relationship with both keratometric and refractive astigmatism. The mean refractive astigmatism and keratometric astigmatism were 0.5 ± 1.4 D and 1.69 ± 3.15 D, respectively, which shows concordance with results of Soriano and colleagues 1 who found that mean preoperative astigmatism in eyes with pterygium was 2.41 D in the horizontal meridian. Astigmatism was with-the-rule in maximum (68.3%) cases which shows consistency with results of study by Fong and colleagues 17 which showed that astigmatism was with-the-rule in 85.7% of pterygium cases. Oner and colleagues 14 reported significant corneal flattening in the horizontal plane compared with the mean corneal power. They found the mean magnitude of refractive astigmatism was 1.66 ± 0.84 D with-the rule in 88.4% of the eyes which was 0.5 ± 1.4 D in the present study. There was a significant association between keratometric astigmatism and horizontal and vertical length of pterygium on slit lamp and horizontal measurements on AS-OCT. Horizontal length on slit lamp and AS-OCT showed association with refractive astigmatism which was proved by a study done by Lin and Stern 18 which showed that pterygia extending to > 45% of the corneal radius or within 3.2 mm of the visual axis produce increasing degrees of astigmatism. In the current study on AS-OCT, two different morphological appearances as flat and nodular were found at limbus and same were reported by Gasser and colleagues 19 also. Loss of integrity of the BM was seen in 60.3% with mean anterior stromal thickness of 124.06 ± 53.42 µm. Similarly Gasser and colleagues 19 reported non-intact BM in 79.3% of cases on AS-OCT which shows concordance with the results of present study.
In 2003, Buchwald and colleagues used both the AS-OCT and the ultrasonic bio-microscopy (UBM) to describe ocular surface lesions including patients with pterygia. They reported that the pterygium appeared as a hyper-reflective lesion above the cornea. The thickness measurements are only possible with OCT or UBM images and not with photographs or slit lamp. 20
Payman demonstrated that pterygium extension and pterygium area had the most influence on corneal astigmatism. 21 This holds true for this study also which showed that the vertical and horizontal dimensions of pterygium on AS-OCT showed evidence of its effect on the keratometric astigmatism. Refractive astigmatism was influenced by the horizontal length measurements both by slit lamp and AS-OCT.
Our findings suggest that the pterygium grade influenced the keratometric astigmatism. The length of pterygium showed its effect on the BCVA. In the current study, it was observed that more the limbal thickness, more will be the refractive astigmatism. This observation was not consistent with Gasser and colleagues who reported that the presence of less limbal thickness was significantly associated with increasing astigmatism. The horizontal and vertical keratometry increased with the increased epithelial thickness just ahead of head of pterygium in the current study.
There are few limitations of the current study as all measurements were done with AS-OCT in horizontal scan only. We have studied the primary pterygium only to rule out various other confounding factors in recurrent or pseudo-pterygium. Corneal topography was not done in these cases. In cases of deep scarring the artifacts in form of shadowing of the corneal light reflex can be seen which leads to problem in qualitative analysis.
In conclusion, AS-OCT is a useful tool for the assessment of various morphometric parameters which helps in monitoring of pterygium and its surgical planning. The pterygium grade influence the keratometric astigmatism. The BCVA shows changes due to pterygium length. Increased limbal thickness of pterygium leads to more refractive astigmatism. Significant associations are seen in between keratometric astigmatism and horizontal measurements on AS-OCT. Various parameters of pterygium like epithelial thickness, pterygium head thickness, limbal thickness and length of pterygium shows effect on both keratometric and refractive astigmatism on AS-OCT which otherwise cannot be elucidated on slit- lamp examination.
Footnotes
Acknowledgements
Mr. Shubham Pandey, Assistant Professor and head biostatistics department, Mr Ankit, Lecturer department biostatistics for Statistical analysis, Mr. Surendra Singh Bhandari, office Assistant for technical support and photographic documentation.
Ethics statement
All procedures performed were in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was approved by the research and ethics committee of the Himalayan Institute of Medical Sciences (approval number HIMS/RC/2018/236). Written informed consent was obtained from all participants prior to their enrollment in this study.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
