Abstract
Objective:
The purpose of our study was to determine the 4-year visual and anatomic outcomes of intravitreal aflibercept treatment for neovascular age-related macular degeneration (AMD) using a treat-and-extend (TAE) regimen.
Methods:
We retrospectively reviewed the medical records of 39 patients with neovascular AMD who were treated continuously with intravitreal aflibercept injections using the TAE regimen for at least 4 years. The outcome measures were the best-corrected visual acuity (BCVA) and central macular thickness (CMT) on spectral-domain optical coherence tomography. The BCVAs were measured as decimal values and converted to the corresponding Early Treatment Diabetic Retinopathy Study (ETDRS) letter scores for statistical analysis. The Wilcoxon signed-rank test was used to compare the differences in BCVAs and CMTs.
Results:
The mean ETDRS letter scores improved significantly from 63.9 at baseline to 70.4, 67.8, 67.2, and 67.3 at 1, 2, 3, and 4 years, respectively. The mean baseline CMT was 380 µm, which decreased significantly to 229, 231, 221, and 210 µm at 1, 2, 3, and 4 years, respectively. The mean numbers of injections were 7.9, 6.0, 5.5, and 5.4 at 1, 2, 3, and 4 years, respectively. The percentages of patients with a treatment interval of 12 weeks or more were 46.2%, 46.2%, 43.6%, and 46.2% at 1, 2, 3, and 4 years, respectively. At year 4, 30.8% of the patients had a treatment interval of 7 weeks or less, whereas 25.6% had 16 weeks or more.
Conclusion:
Intravitreal aflibercept TAE treatment may be an effective and efficient method for treating patients with neovascular AMD up to 4 years of follow-up. The TAE regimen is a potential tool to optimize appropriate treatment intervals, avoiding both undertreatment and overtreatment of neovascular AMD.
Keywords
Introduction
Age-related macular degeneration (AMD) is the leading cause of irreversible blindness in patients 50 years of age or older in developed countries. Anti-vascular endothelial growth factor (VEGF) therapy currently is considered the gold standard for treating neovascular AMD. Although pivotal randomized controlled trials have reported that anti-VEGF therapy is effective for neovascular AMD, most of these studies were not designed to follow patients for more than 2 years.1–3 In addition, a fixed monthly regimen in these trials generated a high treatment burden in real-world clinical practice. To reduce the treatment burden with satisfactory visual and anatomic outcomes, variable treatment regimens, which are based mainly on the activity of neovascular AMD being treated, have evolved.
A treat-and-extend (TAE) regimen is a proactive strategy that continues treatment to avoid recurrences that may result in visual loss but gradually individualizes the treatment intervals based on the ongoing neovascular AMD activity.4–9 Compared with the fixed monthly regimen, the TAE regimen may reduce the numbers of both monitoring visits and injection treatments while still achieving satisfactory visual and anatomic outcomes. The purpose of our study was to determine the 4-year real-world outcomes of an intravitreal aflibercept (Eylea, Regeneron, Tarrytown, NY, USA) TAE regimen for neovascular AMD.
Methods
We retrospectively reviewed the medical records of patients with neovascular AMD who presented to the macular degeneration unit of Kurume University Hospital from February 2013 to September 2018. Patients were included in the study if they had been continuously treated with intravitreal aflibercept injections using the TAE regimen for at least 4 years. We included only one eye of each patient as no patient had a 4-year treatment period in both eyes. Patients who underwent cataract surgery in the study eyes during the treatment period were excluded from analysis of the best-corrected visual acuity (BCVA) postoperatively. This study adhered to the tenets of the Declaration of Helsinki. The Ethical Committee of Kurume University approved this study and waived the need for individual patient consent (No. 17107).
Patients received at least three monthly injections until anatomic improvement of the macula became apparent, as judged by the absence of intraretinal and subretinal fluid on spectral-domain optical coherence tomography (SD-OCT; RS-3000, NIDEK, Gamagori, Japan). Subsequent treatment intervals were determined based on the disease activity. The exudative change was defined as new retinal hemorrhage or presence of intraretinal and/or subretinal fluid on SD-OCT. The injection interval was extended by 2 weeks if there was no exudative change, whereas the interval was shortened by 2 weeks if any exudative change was seen. A loss of BCVA by itself did not prohibit extension. The scheduled treatment interval was shortened to a minimum of 4 weeks and extended to a maximum of 20 weeks in this study.
The diagnosis of neovascular AMD was confirmed using color fundus photography, SD-OCT, fluorescein angiography, and indocyanine green angiography. At each clinical visit, all patients underwent complete ocular examinations, including measurement of the BCVA and intraocular pressure, slit-lamp biomicroscopy, dilated fundus examinations, and SD-OCT. The BCVAs were measured as decimal values and converted to the corresponding Early Treatment Diabetic Retinopathy Study (ETDRS) letter scores for statistical analysis. 10 The data are presented as the mean ± standard deviation. The Wilcoxon signed-rank test was used to compare the differences in BCVAs and central macular thicknesses (CMTs).
Results
Thirty-nine eyes of 39 patients (25 men, 14 women) with neovascular AMD were classified at baseline into three subtypes: 10 eyes with typical AMD, 26 eyes with polypoidal choroidal vasculopathy, and 3 eyes with retinal angiomatous proliferation. Twenty-one eyes were treatment-naive. Eighteen eyes had been treated previously with either intravitreal ranibizumab (Lucentis; Genentech Inc., South San Francisco, CA, USA) injections (5 eyes), photodynamic therapy (1 eye), or photodynamic therapy with intravitreal ranibizumab and/or pegaptanib sodium (Macugen; OSI Pharmaceuticals, Melville, NY, USA) injections (12 eyes). Six patients underwent cataract surgeries in the study eyes during the treatment period. The mean age of the included patients was 75.5 ± 6.7 years.
The BCVA improved significantly at 1, 2, 3, and 4 years compared with baseline. The ETDRS letter scores (mean ± standard deviation) improved from 63.9 ± 15.0 at baseline to 70.4 ± 13.5 (p < 0.0001), 67.8 ± 16.9 (p < 0.01), 67.2 ± 17.3 (p < 0.01), and 67.3 ± 14.0 (p < 0.01) at 1, 2, 3, and 4 years, respectively (Figure 1). After 4 years of treatment, eight eyes (24%) improved by 15 letters or more, whereas two eyes (6%) worsened by 15 letter scores or more compared with baseline. Subgroup analyses showed that ETDRS letter scores (mean ± standard deviation) in the previously untreated group were 69.4 ± 13.3 at baseline and 76.2 ± 10.5 (p < 0.0001), 74.3 ± 13.9 (p < 0.0001), 76.9 ± 11.4 (p < 0.0001), and 74.6 ± 11.4 (p < 0.0001) at 1, 2, 3, 4 years, respectively. On the other hand, ETDRS letter scores (mean ± standard deviation) in the previously treated group were 57.5 ± 14.4 at baseline and 63.5 ± 13.4 (p < 0.01), 60.1 ± 16.8, 56.2 ± 16.2, and 58.6 ± 11.7 at 1, 2, 3, 4 years, respectively.
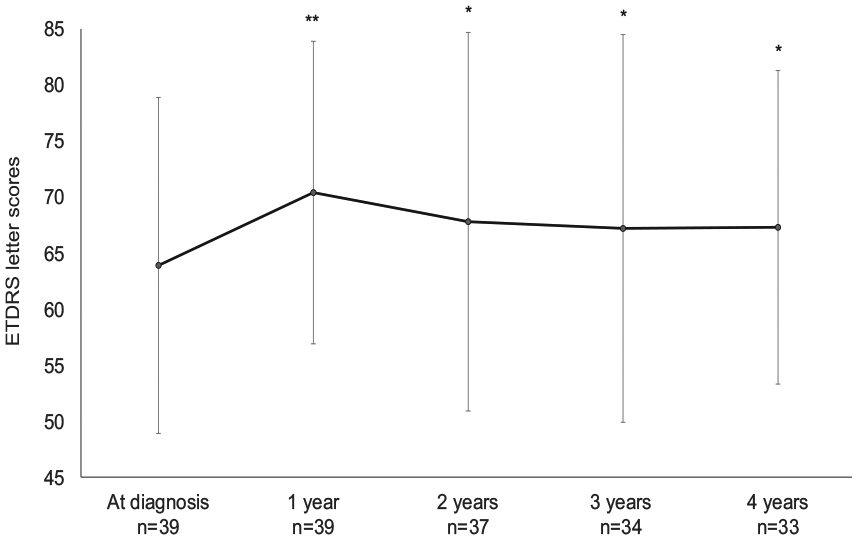
The best-corrected visual acuity (mean ± standard deviation). Patients who underwent cataract surgery in the study eyes during the treatment period were excluded from analysis of the best-corrected visual acuity postoperatively.
The CMT measured by SD-OCT decreased significantly at 1, 2, 3, and 4 years compared with baseline. Patients had a baseline CMT (mean ± standard deviation) of 380 ± 117 µm, which decreased to 229 ± 97 (p < 0.0001), 231 ± 143 (p < 0.0001), 221 ± 86 (p < 0.0001), and 210 ± 84 (p < 0.0001) µm at 1, 2, 3, and 4 years, respectively (Figure 2).
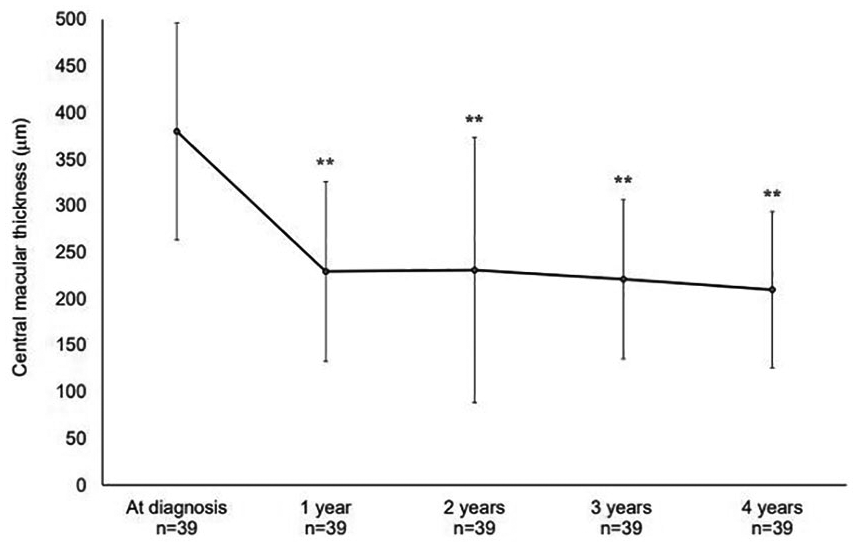
The central macular thickness (mean ± standard deviation).
The numbers of injections (mean ± standard deviation) were 7.9 ± 1.4, 6.0 ± 2.7, 5.5 ± 2.5, and 5.4 ± 2.3 at 1, 2, 3, and 4 years, respectively (Figure 3). The percentages of patients with a treatment interval of 12 weeks or more were 46.2%, 46.2%, 43.6%, and 46.2% at 1, 2, 3, and 4 years, respectively (Figure 4). At year 4, 30.8% of the patients had a treatment interval of 7 weeks or less, whereas 25.6% had a treatment interval of 16 weeks or more.

The numbers of injections (mean ± standard deviation) administered each year of the study.

Proportion of treatment intervals over the course of the study.
Discussion
Pivotal clinical trials have shown that anti-VEGF therapy in patients with neovascular AMD leads to excellent visual improvements with a fixed monthly regimen. However, the extended follow-up studies showed that visual improvements were difficult to maintain. The SEVEN-UP study reported an overall mean reduction in letter scores from a baseline of 8.6 after 7.3 years of treatment. 11 This group had received 2 years of monthly ranibizumab treatment, followed by an additional 2 years of as-needed ranibizumab treatment in the HORIZON study. However, the patients in the SEVEN-UP study seemed to be undertreated after release from the HORIZON study, given the facts that the macula had intraretinal or subretinal fluid in two-thirds of patients and that an average of only 1.6 annual injections was given during the last 3.3 years. In addition, macular atrophy was detected in 98% of the study eyes and may have contributed to the poorer visual outcomes. The CATT follow-up study reported similar results, with a mean reduction in letter scores from baseline of 3 after 5 years of treatment. 12
Long-term follow-up of anti-VEGF therapy using the TAE regimen [with bevacizumab (Avastin; Genentech Inc., South San Francisco, CA, USA.), ranibizumab, or aflibercept] showed that the BCVA was maintained for an extended period in patients with neovascular AMD. Berg and colleagues reported that the BCVA improved significantly from baseline during the first 6 years of treatment in patients who completed the 8-year follow-up. 13 In this study, we evaluated the visual and anatomic outcomes of intravitreal aflibercept TAE treatment in patients with neovascular AMD who completed the 4-year follow-up. The current results showed that the BCVA improved significantly from baseline up to 4 years of treatment. The CMT also decreased significantly from baseline during the 4 years of treatment. Our study confirmed that intravitreal aflibercept TAE treatment may be appropriate for long-term treatment of neovascular AMD.
Traine and colleagues reported the 4-year outcomes of intravitreal aflibercept TAE treatment for neovascular AMD. 9 In their study, the BCVA improved significantly during the first 3 years of treatment and remained stable at year 4 compared with baseline. Patients received an average of 7.7 injections during the first year and an average of 4.4 annual injections during the last 3 years. Their visual and anatomic outcomes were similar to ours; however, their study showed decreased BCVA from year 3 to year 4 compared with this study. Gayadine-Harricham and colleagues also reported the 4-year outcomes of intravitreal aflibercept modified TAE treatment for neovascular AMD. 14 In their study, the BCVA improved significantly during the first 2 years of treatment and remained stable at the last 2 years compared with baseline. Patients received an average of 5.7 injections during the first year and an average of 3.5 annual injections during the last 3 years. The difference between their regimen and ours was that they used modified TAE, in which patients were treated as needed from week 12 to week 32 to determine the recurrence interval.
In the VIEW 1/2 studies, intravitreal aflibercept injected every 2 months after three initial monthly injections was noninferior to monthly ranibizumab injections for treating neovascular AMD. 3 The aflibercept TAE regimen can be compared with the every-2-month regimen used in the VIEW 1/2 studies. In the current TAE study, the mean number of injections from baseline to year 4 was 24.8, whereas the theoretical number of injections would be 25.0 if treated with the every-2-month regimen for a period of 4 years. Although the total number of injections seemed to be comparable between these two regimens, we believe that the TAE regimen has some advantages over the every-2-month regimen. In the current TAE study, 30.8% of patients were being treated at 7 weeks or less at year 4. These patients would be undertreated with an every-2-month regimen, potentially leading to inferior visual and anatomic outcomes. Conversely, 25.6% of patients being treated at 16 weeks or more at year 4 would be overtreated with the every-2-month regimen, potentially increasing the risk of developing macular atrophy. 15 Although patients with neovascular AMD can have divergent responses to anti-VEGF therapy, the TAE regimen seems to have potential for optimizing the appropriate treatment intervals by avoiding both undertreatment and overtreatment of neovascular AMD.
The current TAE study showed that the percentages of patients with a treatment interval of 12 weeks or more were stable between 43.6% and 46.2% during the 4 years of treatment. These results suggested that each patient has an individually optimized treatment interval, which can be determined by the TAE regimen. In addition, of the 17 patients treated at an interval of 12 weeks or more at the end of year 1, 13 patients (76.5%) had the same interval at the end of year 4. Conversely, of the seven patients treated at an interval of 7 weeks or less at the end of year 1, five patients (71.4%) had the same interval at the end of year 4. These results indicated that the treatment interval at the end of year 1 may predict the treatment interval at the end of year 4.
The limitations of our study included its retrospective nature, the small number of cases, and the heterogeneous patients with typical AMD, polypoidal choroidal vasculopathy, and retinal angiomatous proliferation. Polypoidal choroidal vasculopathy is highly prevalent in Japanese individuals and accounts for 55% of patients with neovascular AMD in Japan. 16 The entire population of this study was Japanese, indicating that the results may not be applicable to a larger AMD population. In addition, 18 of 39 eyes had a previous history of neovascular AMD treatment. However, our study reported real-world outcomes of intravitreal aflibercept TAE treatment for neovascular AMD for a period of up to 4 years.
In conclusion, this study showed that intravitreal aflibercept TAE treatment may be an effective and efficient method for treating patients with neovascular AMD up to 4 years of follow-up. The TAE regimen is a potential tool for optimizing appropriate treatment intervals by avoiding undertreatment and overtreatment of neovascular AMD.
Footnotes
Acknowledgements
The authors thank Lynda Charters for English editing.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
