Abstract
Objective:
To compare the anatomical and functional outcomes of intensive and relaxed intravitreal aflibercept regimen in the management of neovascular age-related macular degeneration.
Methods:
The medical records of 50 eyes of 50 patients with treatment-naive neovascular age-related macular degeneration, who underwent intravitreal aflibercept therapy in the Retinal Department of University of Health Sciences Okmeydanı Training and Research Hospital (Istanbul, Turkey), were retrospectively reviewed. The demographic features and best-corrected visual acuities at baseline and at 1st-, 2nd-, 3rd-, 4th-, 6th-, 10th- and 12th- months, optical coherence tomography measurements, presence of intraretinal fluid, subretinal fluid and intraocular pressure values were studied. The patients were divided into two groups: intensive (subretinal fluid intolerant) and relaxed (<200 microns subretinal fluid tolerated) intravitreal aflibercept regimens.
Results:
The change in the mean best-corrected visual acuities from baseline to 12th month was 13.72 ± 33.97 letters in the intensive treatment group and 8.68 ± 27.22 in the relaxed group. There was no statistically significant difference between the groups (p = 0.566). Similar proportions of both groups achieved ⩾10 letters gain (%48 vs. %40, respectively; p = 0.755) and ⩾10 letters loss (%20 vs. %24, respectively; p = 0.755). The mean central subfield thickness values decreased statistically significantly in both groups (p < 0.001). There was no statistically significant difference between the groups (442.40 ± 161.32 to 318.0 ± 63.10 vs. 431.64 ± 151.52 to 303.08 ± 140.91, respectively; p = 0.724). The mean total injection numbers were significantly lower in the relaxed group (6.4 ± 1.65 vs. 4.8 ± 1.45, respectively; p = 0.01).
Conclusion:
Relaxed intravitreal aflibercept regimen has comparable efficiency and safety outcomes at 12 months with intensive intravitreal aflibercept regimen with fewer injections in the management of neovascular age-related macular degeneration.
Keywords
Introduction
Age-related macular degeneration (AMD) is the most common cause of legal blindness in individuals above the age of 65, particularly in developed countries. 1 The risk factors of AMD, whose prevalence increases with prolongation of human life are age, smoking, ultraviolet exposure, race, genetic factors, nutritional characteristics, systemic hypertension and certain drugs.2,3
AMD is classified into early and advanced or non-neovascular (dry and atrophic) and neovascular (wet and exudative) types. Non-neovascular AMD accounts for 85-90% and is the main clinical manifestation of drusen and retina pigment epithelium (RPE) atrophy. Neovascular AMD (nAMD) constitutes 10-15% of AMD cases and is the major cause of severe vision loss. 4 In the late stage of the disease, geographic atrophy or disciform scarring occurs. 5
Many therapies have been attempted in the treatment of nAMD. The indications of argon laser photocoagulation, photodynamic therapy, transpupillary thermotherapy, radiation therapy and surgical treatments such as submacular surgery, macular translocation and pneumatic displacement, have been largely diminished by intravitreal anti-vascular endothelial growth factor (VEGF) agents, which are considered the gold standard treatment. 6
Aflibercept is a humanized recombinant protein produced from human VEGF receptor fragments that inhibits VEGF-B and placental growth factor (PIGF) in addition to VEGF-A. It binds to VEGF receptors with higher affinity and has a longer half-life than other anti-VEGF agents. 7
Currently, three different regimens are used in anti-VEGF treatments for nAMD: monthly, pro-re-nata (PRN) and treat and extend (T&E). 8 In the PRN regimen, the patient is monitored monthly for disease activity after the administration of a loading dosage. If best-corrected visual acuity (BCVA) decreases, subretinal (SRF) and/or intraretinal fluids (IRF) are present in the optical coherence tomography (OCT), or newly formed choroidal neovascularization (CNV) or newly developed macular haemorrhage occurs, intravitreal anti-VEGF injections are readministered. In the T&E regimen, patients receive intravitreal injections on a monthly basis until there is no IRF or SRF. After the macula is completely dry, the follow-up intervals for the patients are extended for 2 weeks. If a new macular haemorrhage or fluid is detected during patient follow-up, intravitreal injection is performed, and the follow-up interval is shortened to 2 weeks. 8
To date, no studies had examined the re-treatment criteria for anti-VEGF therapy in nAMD prior to the FLUID (SRF Tolerance in Ranibizumab-Treated AMD Eyes) study. According to the 24-month results of this study, published at the end of 2018, it was observed that naive nAMD patients treated with intravitreal ranibizumab had reached similar BCVA changes by tolerating 200 µm or less of SRF. 9
Since there has been no similar research on intravitreal aflibercept (IVA), in this study, we aimed to compare the efficacy and safety of the 1-year results of two treatment groups labelled as intensive (SRF intolerant) and relaxed (<200 µm SRF tolerated) using an IVA PRN regimen in our case series of naive patients with nAMD.
Materials and methods
We retrospectively studied the medical records of the patients who were diagnosed with nAMD by clinical examination, FFA (Visucam 524®; Carl Zeiss Meditec, Jena, Germany) and OCT (Spectralis (SD) OCT®, Heidelberg Engineering, Heidelberg, Germany) and treated with IVA at the Retina Department of the University of Health Sciences (UHS), Okmeydanı Training and Research Hospital (Istanbul, Turkey) between January 2017 and December 2019.
Fifty eyes from 50 patients were included in the study based on the criteria given below.
An age of 50 years or older;
No previous intravitreal injection therapy with a different agent;
Type-1 and/or type-2 CNV due to nAMD developed and followed up regularly for at least 1 year with IVA treatment.
In our study, patients with the criteria given below were excluded:
Those with other concomitant eye pathologies (other retinal diseases, corneal pathologies, amblyopia, etc.);
Patients with secondary CNV (degenerative myopia, angioid streaks, polypoidal choroidal vasculopathy, retinal angiomatous proliferation, etc.);
Patients with a history of uncontrolled hypertension, thromboembolic disease;
Patients who had not followed up regularly for at least 1 year after treatment.
The medical records of patients with treatment-naive nAMD who underwent IVA therapy were retrospectively reviewed. The demographic features, BCVAs, OCT measurements, the presence of IRF, SRF and intraocular pressure (IOP) values were studied at baseline and at first, second, third, fourth, sixth, eighth, 10th and 12th months. The patients were divided into two groups according to the regimen: intensive (SRF intolerant) and relaxed (<200 microns SRF tolerated). Some of the clinicians in our clinic did not retreat the patients with mild SRF following to the release of FLUID study at November 2018. The patients who were included in the relaxed treatment group were selected from those.
All patients received three consecutive IVA monthly loading doses. At the monthly controls, after the third injection, the intensive treatment group was retreated with IVA following indication of the presence and/or an increase of any IRF or SRF, visual loss or newly developed retinal haemorrhage, while the relaxed group was retreated for similar indications except that the treatment was not given when SRF ⩽ 200 µm.
BCVA was determined according to the Early Treatment Diabetic Retinopathy Study (ETDRS) chart. IOP was measured by an applanation tonometer. OCT scans were performed with the same device in tracking mode by the same experienced operator and measurements were done by callipers on SD OCT.
Intravitreal injections were administered under sterile conditions. For topical anaesthesia, 0.5% propacaine was applied, and then the eye and its surroundings were wiped from the centre to the periphery with 10% povidone iodine twice; then, 5% povidone iodine was dropped into the eye and eyelashes and left for at least 2 min. Then, 2 mg/0.05 ml aflibercept was applied with a 27-gauge needle in the lower or upper temporal region at a distance of 4 mm in phakic eyes and 3.5 mm in pseudophakic eyes (pars plana). To prevent vitreous incarceration, the injection site was pressed with a cotton applicator. After injection, topical moxifloxacin was administered to patients four times a day during a 1-week period. Patients were monitored on the first day after the injection for side effects.
The IBM SPSS 25.0 (SPSS Inc., Chicago, IL, USA) programme was used for statistical analysis. Descriptive statistics were given in the form of the mean and standard deviation for variables showing a normal distribution and as the median and percentile (percent) for non-normally distributed variables. Parametric and/or non-parametric tests were used for comparisons between the groups and were based on a normal distribution. The comparison of gain and loss ratios of ⩾10 letters and other categorical variables was performed using Fisher’s exact test. The significance of the changes in BCVA, central subfield thickness (CST) and IOP values over time and the effects of treatment regimen were investigated by using repeated measures of analysis of variance (ANOVA). A Greenhouse-Geisser correction was preferred in cases where a sphericity assumption was not provided. P < 0.05 was considered statistically significant in all analyses.
Results
Baseline characteristics
Fifty eyes of 50 patients (25 males (M)/25 females (F)) were included in this retrospective comparative study. Twenty-five patients (14 M/11 F) were included in the intensive PRN regimen group and 25 patients (11 M/14 F) in the relaxed group. The ages of the subjects ranged from 50 to 87 with a mean of 70.32 ± 10.77 years, which was similar in the two groups (p = 0.796).
Baseline clinical and descriptive characteristics are shown in Table 1. There was no statistically significant difference in the baseline BCVA, CST and IOP values between the groups (p > 0.05). The baseline mean BCVA was 35.80 ± 31.64 letters and 31.64 ± 29.47 letters, respectively. The mean CST values were 442.4 ± 161.32 µm and 431.6 ± 151.52 µm, respectively, at baseline. Before the treatment, the proportions of participants with IRF were 56% and 68% and with SRF were 88% and 84%, respectively.
Baseline clinical and descriptive characteristics of the participants in intensive and relaxed treatment groups.

OCT, optical coherence tomography.
BCVA changes
The change in BCVA from baseline to 12 months was 13.72 ± 33.97 letters in the intensive arm and 8.68 ± 27.22 letters in the relaxed arm, and the difference was not statistically significant (p = 0.566). Similar proportions of both groups gained ⩾10 letters (48% vs. 40%, respectively; p = 0.755) and lost ⩾10 letters (20% vs. 24%, respectively; p = 0.755).
In all patients, the mean BCVA increase was statistically significant (p = 0.014, respectively), as shown in Figure 1; however, there was no significant difference between the two groups (p = 0.595, respectively).

The change of mean best-corrected visual acuity from baseline to month 12 in intensive and relax treatment groups.
CST changes
At the end of 12 months, the mean CST values decreased from 437.02 ± 154.98 to 310.54 ± 108.31 µm in all patients (p = 0). The mean CST value in the intensive arm decreased from 442.40 ± 161.32 to 318.0 ± 63.10 µm, whereas in the relaxed arm, it decreased from 431.64 ± 151.52 to 303.08 ± 140.91 µm. As shown in Figure 2, the mean changes in CST were similar in both groups (p = 0.724).
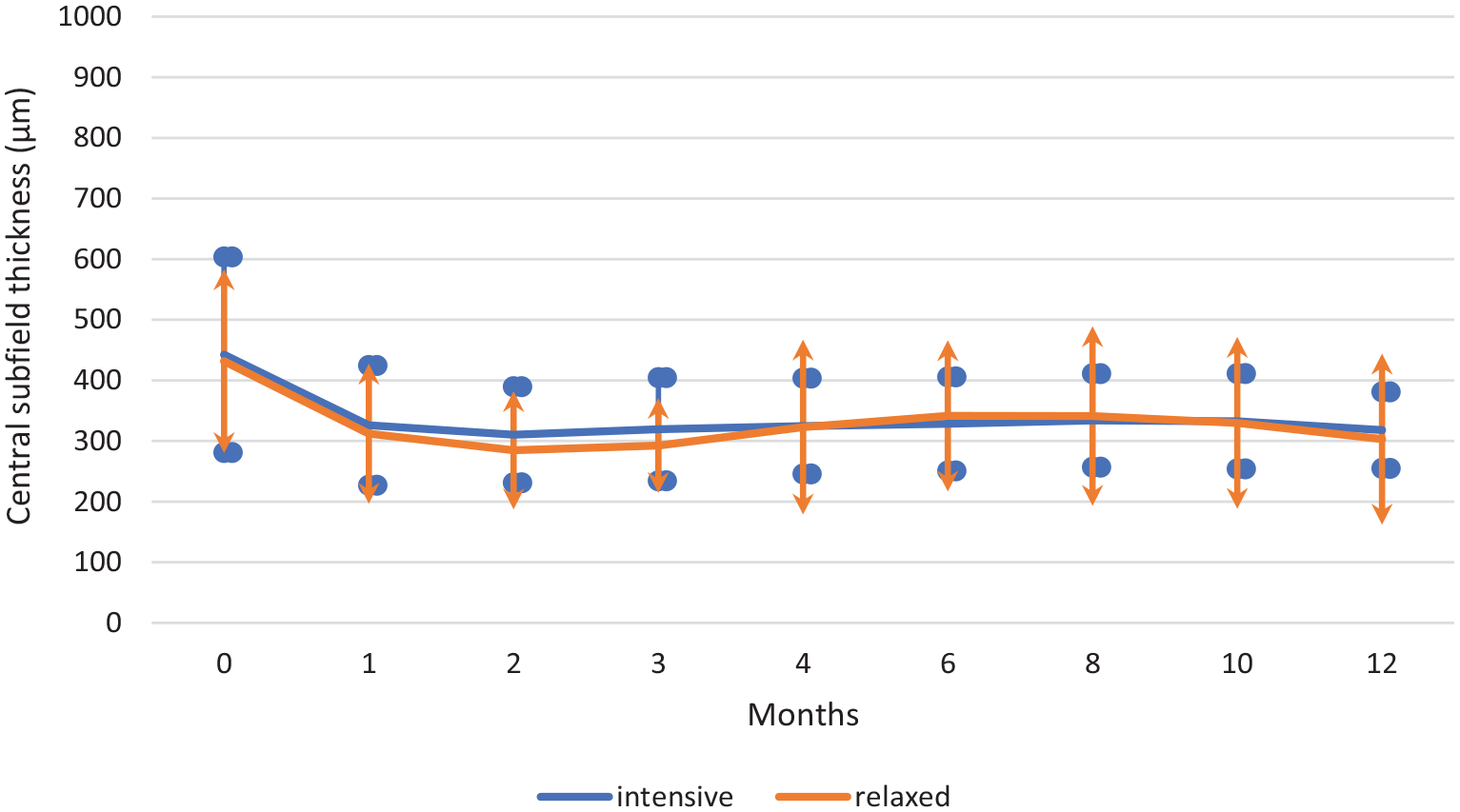
The change of mean central subfield thickness from baseline to month 12 in intensive and relaxed treatment groups.
Intraretinal and SRF changes
In all patients, the proportion of patients with IRF at 12 months significantly decreased from 62% to 8% (p = 0). However, the change in the proportion of patients with IRF was similar among the groups (p = 0.712).
Similar results were observed in the SRF changes. In all patients, the proportion of patients with SRF at 12 months reduced from 86% to 24% (p = 0). SRF proportions were similar between the groups (p = 0.751; Table 2)
Subretinal and intraretinal fluid proportions in intensive and relaxed treatment groups.

IRF, intraretinal fluid; SRF, subretinal fluid.
Total numbers of injections
The mean number of IVA injections at 12 months was significantly lower in the relaxed group (4.8 ± 1.45) than in the intensive treatment group (6.4 ± 1.65; p = 0.01).
Safety
There was no difference in the number of adverse events and serious adverse events for either all body systems or the ocular system among the intensive and relaxed groups. No new safety signals were observed for aflibercept.
In our study, there was no significant change in IOP values at 12 months (p = 0.96). Again, IOP changes were similar (p = 0.296).
Discussion
This pilot study investigated whether minimal SRF is a re-treatment criterion in the management of nAMD with the IVA PRN regimen. We found that similar anatomical and functional results may be achieved by tolerating some SRF in patients with nAMD.
The FLUID study, which published 24-month results in November 2018, is a multicentre, randomized, phase-IV trial involving naive nAMD patients. In this study, 349 patients were divided into two groups: intensive and relaxed. Intravitreal ranibizumab (0.5 mg) with the T&E regimen was administered monthly to the intensive group until the entire IRF and SRF disappeared, while the other group was injected only in the presence of IRF by tolerating SRF less than 200 µm. In the study, the 2-year BCVA results were similar in both groups. The mean baseline BCVA was 62.3 ± 15.2 letters in the intensive group and 64.2 ± 12.6 letters in the relaxed group. The mean letter gains were 3.0 ± 16.3 in the intensive arm and 2.6 ± 16.3 in the relaxed arm at the end of 2 years. The proportions of patients with 20/40 and better BCVA were 53.5% and 56.6% and of patients with 20/200 and worse BCVA were 8.7% and 8.1%, respectively, at 24 months. 9
In our study, 50 eyes of 50 patients with nAMD who were treated with the IVA PRN regimen were examined. Similar to the FLUID study, the patients were divided into two equal groups as either intensive or relaxed treatment, and their 12-month outcomes were compared. The mean baseline BCVA, which is lower than that of the FLUID study, was 35.80 ± 31.64 letters in the intensive arm and 31.64 ± 29.47 in the relaxed arm. The mean letter gains, which are higher compared to the FLUID study, were 13.72 ± 33.97 in the intensive arm and 8.68 ± 27.22 in the relaxed arm at 12 months. We attributed this higher letter gain to the fact that the patients in our study had lower initial BCVA. As is known, patients with lower BCVA gain more letters than patients with higher BCVA due to the ceiling effect.
In our study, the mean letter gain in the intensive group was not better than that in the relaxed group, akin to the results of the FLUID study. This finding suggests that a comparable BCVA gain may be achieved by tolerating some SRF in the treatment of nAMD with IVA.
In the FLUID study, although the mean CST changes were similar between the two groups at 12 months, the decrease in the value in the intensive treatment group was significantly higher than in the relaxed group at 24 months. 9 In our study, the mean CST was significantly reduced at the end of 12 months, but there was no notable difference between the groups.
In the FLUID study, the incidence of SRF decreased significantly at 2, 12, and 24 months, but there was no difference between the two groups. While the incidence of IRF showed a significant decrease in total, there was no difference between the groups at 24 months. However, the rate of IRF was significantly higher in the relaxed group at 2 and 12 months. In our study, the incidence of IRF and SRF was significantly reduced in all patients, but no significant difference was found between the groups at 12 months. However, the intensive group had more SRF than the relaxed group throughout the study period, which was not an expected finding.
The phase-III studies of aflibercept, VIEW-1 and VIEW-2 (VEGF Trap-Eye: Investigation of Efficacy and Safety in Wet AMD) are prospective, randomized, double-blind studies. The VIEW-1 and VIEW-2 studies have shown that in eyes with IRF at baseline, there is a lower BCVA outcome, while SRF does not affect BCVA. 10 In addition, the resolution of IRF occurs rapidly in the first week after treatment, while SRF can persist until the 12th week. 11 The VIEW-1 and VIEW-2 studies concluded that there is a need for further detailed studies to understand whether aggressive SRF treatment is necessary.
In the CATT study, eyes with IRF were associated with worse BCVA results at 24 months, while eyes with SRF had significantly better results. 12 Therefore, the CATT study has also indicated the need for studies about tolerating a certain amount of SRF and less-aggressive anti-VEGF therapy. 13
In randomized controlled trials, the mean number of injections per year was 7.7 in the HARBOUR study, 6.9 in the CATT study, 7.6 in the VIEW study, and 9.5 in the intensive arm and 8.9 in the relaxed arm of the FLUID study.9,11,13,14 In our retrospective study, the mean number of injections per year was 6.4 in the intensive arm and 4.8 in the relaxed arm, which is similar to the meta-analysis of Kim and colleagues 15 in which the real-life data were analysed; the mean numbers of injections per year were 3.6 in Germany, 2.8 in France, and 4.5 in the United Kingdom.
The FLUID study showed that SRF in nAMD may be the result of a non-exudative process. SRF caused by active CNV may not always be indicative of the need for treatment. 9 In addition, SRF may also be useful in reducing the development of atrophy, as indicated in the CATT and HARBOUR studies.13,14 Moreover, the SEVEN-UP (Seven-Year Observational update of macular degeneration patients post-MARINA/ANCHOR and HORIZON trials) and CATT studies have shown that more injections for completely dry retinas increase the risk of atrophy.13–16 Furthermore, the most important outcome of our study is achieving similar BCVA and CST results with significantly less intravitreal injections by tolerating some SRF within the 12-month period.
Our study was the first that tolerated some SRF in the treatment of IVA in nAMD. It should be noted that the re-treatment criteria of our clinic have changed based on the results of the FLUID study (November 2018), and SRF ⩽ 200 µm was tolerated in nAMD. Namely, the patients who were included in the intensive treatment group were selected from those followed in our clinic between January 2017 and November 2018, whereas those followed in November 2018 and December 2019 were included in the relaxed treatment group.
We believe that the main limitation of this study is its low sample size; unfortunately we could only enrol such a small number of participants who met the inclusion criteria. Another limitation is including the patients with very low BCVA leading to high standard deviations. Since our study had a retrospective nature and involved a low number of patients, our results may not be applicable to the general population. Therefore, further prospective and comprehensive studies are needed on this subject.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethicals Statement
This study conformed to the tenets of Helsinki Declaration. The study was approved by the UHS Okmeydani Training and Research Hospital Clinical Research Ethics Committee (approval no. 48670771). Written informed consent was obtained from all participants before the study.
