Abstract
Background:
Some patients with neovascular age-related macular degeneration (nAMD) have persistent intraretinal/subretinal fluid (IRF/SRF) despite being treated with anti-VEGF agents. There is limited data on efficacy of switching to intravitreal brolucizumab (IVBr) in these patients.
Purpose:
To determine anatomic and visual outcomes of eyes with nAMD treated with for persistent IRF/SRF.
Methods:
Retrospective series of eyes with nAMD treated initially with aflibercept (IVA, n = 48) and bevacizumab (IVBe, n = 10), then switched to IVBr for persistent IRF/SRF.
Results:
In the IVA-IVBr group, a mean of 42 days after one IVBr, mean logMAR changed from 0.50 to 0.49 (p = 0.73) and mean CSFT changed from 340 to 305 µm (p < 0.001); 31% of eyes had no fluid, 42% had persistent but reduced fluid, 25% had stable fluid, and 2% had increased fluid. For a subgroup of 25 eyes that completed a series of 3 IVBr, mean logMAR changed from 0.44 to 0.40 (p = 0.35) and mean CSFT changed from 325 to 277 µm (p = 0.001); 24% of eyes had no fluid at last follow-up, a mean of 54 days after last IVBr. In the IVBe-IVBr group, a mean of 44 days after one IVBr, mean logMAR changed from 0.46 to 0.40 (p = 0.114) and mean CSFT from 401 to 325 µm (p = 0.009); 30% of eyes had no fluid and 70% had persistent but reduced fluid. For a subgroup of four eyes that completed a series of three IVBr, mean logMAR changed from 0.33 to 0.35 (p = 0.391) and mean CSFT improved from 375 to 275 µm (p = 0.001); 50% of eyes had no fluid at last follow-up, a mean of 65 days after last IVBr.
Conclusion:
In nAMD eyes previously treated with IVA and IVBe, switching to IVBr significantly reduced persistent IRF/SRF but did not significantly affect visual outcomes.
Keywords
Introduction
Age-related macular degeneration (AMD) is the leading cause of irreversible legal blindness in people ⩾65 years of age in the Western world and affected approximately 3 million people in the United States in 2020. 1 Neovascular age-related macular degeneration (nAMD), although less prevalent than dry AMD, commonly causes acute and substantial central vision loss due to growth of pathologic choroidal neovascularization (CNV), which causes exudation of blood and/or fluid into the macula.2,3
The aberrant vascular growth and exudation in nAMD are driven by cascade of many cytokines involved in angiogenesis, primarily vascular endothelial growth factor-A (VEGF). 4 The introduction of intravitreally administered anti-VEGF agents has led to notably improved outcomes for patients with nAMD and includes ranibizumab, aflibercept, brolucizumab, and bevacizumab (off-label for intraocular use). 5 Brolucizumab (Beovu®, Novartis, Basel, Switzerland) is a 26-kDa, humanized monoclonal single-chain antibody fragment that binds VEGF and is the most recently approved therapy for nAMD in the United States, as of October 2019. Brolucizumab is the first single-chain antibody to be developed for nAMD and is smaller than ranibizumab (48 kDa) and aflibercept (115 kDa). The smaller molecule size facilitates higher molar concentration, 11 to 13 times greater than that of aflibercept. 6
The efficacy of intravitreal brolucizumab (IVBr) in the treatment of nAMD was evaluated in HAWK and HARRIER, two prospective, randomized control trials. 7 IVBr met the primary efficacy objective of non-inferiority in best corrected visual acuity (BCVA) from baseline to week 48 compared with intravitreal aflibercept (IVA). In addition, IVBr demonstrated superiority in the secondary endpoints of the degree of central subfield thickness (CSFT) and presence of retinal fluid. At 48 weeks, 56% of HAWK patients and 51% of HARRIER patients in the 6 mg IVBr group were maintained on a 12-week treatment interval after the initial loading phase. 7 Two-year data demonstrated that fewer patients with nAMD had retinal fluid with IVBr 6 mg compared with IVA in HAWK (24% versus 37%) and HARRIER (24% versus 39%). Reduction in CSFT was also greater at 96 weeks in the 6 mg IVBr group compared with IVA in both HAWK (−175 µm versus −149 µm) and HARRIER (−198 µm versus −155 µm). 8 While this trial compared IVBr against IVA in treatment-naïve nAMD patients, there are limited data available about efficacy of switching to IVBr in eyes with refractory nAMD. The purpose of this study was to examine the outcomes of patients who were switched to IVBr due to persistence of intraretinal fluid (IRF) or subretinal fluid (SRF) despite treatment with other anti-VEGF agents.
Methods
This retrospective chart review studied consecutive patients diagnosed with refractory nAMD who were treated with their first IVBr between 1 November 2019 and 10 January 2020. The project was reviewed by Advarra institutional review board (IRB) and they determined that this study was exempt from the IRB approval process. The research adhered to the tenets of the Declaration of Helsinki and all patients signed an informed consent form to proceed with treatment. While a consensus definition of refractory nAMD has not been established by the literature, for the purpose of this study it was diagnosed if the patients showed persistent IRF or SRF despite at least three prior anti-VEGF treatments, including any combination of intravitreal bevacizumab (IVBe), intravitreal ranibizumab (IVR), or IVA. Patients were stratified into groups based on their most recent anti-VEGF agent. Four retina specialists (R.H., S.P.B., S.H., and K.H.P.) were the treating providers, all of whom typically treat nAMD using a ‘treat and extend’ protocol, in which the interval between injections is extended gradually when there is a dry macula.
Exclusion criteria included other causes of macular exudation, such as diabetic macular edema or retinal vein occlusion. Patients were also excluded if they had fewer than three previous anti-VEGF treatments and fewer than two IVBr injections. Figure 1 is a flow chart that shows inclusion criteria and number of patients in each group (classified by last agent injected prior to IVBr and number of IVBr injections per group). Patient charts were reviewed for eligibility, and data were extracted regarding the patient’s age, gender, race, lens status, presence of geographic atrophy (based on the physicians’ most recent fundus examination and OCT scan interpretation), previous interventions and treatment intervals, CSFT, and presence of IRF and SRF on OCT. Fluorescein angiography was performed on each patient at initial presentation to the clinic. The BCVA, CSFT, treatment dates from the initial visit, and follow-up visits were recorded. Snellen visual acuity was converted to the logarithm of the minimal angle of resolution equivalents (logMAR visual acuity) before statistical analysis. Mean values for logMAR and CSFT, as well as standard deviations and range, were calculated at each follow-up visit. A paired t test was performed on the CSFT and logMAR at various intervals using SPSS version 25, with p values representing change from baseline. A cutoff of p < 0.05 was used for statistical significance.

Flow chart that details the inclusion criteria and breakdown of number of patients in each treatment group (classified by last anti-VEGF agent prior to switch and number of brolucizumab injections received after switch). Anti-VEGF, anti-vascular endothelial growth factor; IVA, intravitreal aflibercept; IVBe, intravitreal bevacizumab; IVR, intravitreal ranibizumab.
On all visits, response to treatment was evaluated subjectively by Snellen BCVA, and objectively by either Zeiss Cirrus SD-OCT (Carl Zeiss Meditec, Jena, Germany) or Spectralis SD-OCT (Heidelberg Engineering, Heidelberg, Germany). In all cases, the use of IVBr and its potential risks and benefits were discussed with the patients before they signed an informed consent. IVBr 6 mg was injected 3.5 to 4 mm posterior to the limbus under aseptic conditions. Patients were assessed at individualized treatment intervals using a ‘treat and extend’ approach with extension of intervals by 1 week if the treating physician determined the macula was dry. After the first IVBr, the follow-up interval remained unchanged from the previous visit.
On 23 February 2020, the American Society of Retina Specialists (ASRS) issued a safety warning about intraocular inflammation and occlusive vasculitis associated with IVBr use (personal email communication). This unexpected news resulted in the eventual discontinuation of IVBr in all the patients in this study.
Results
A total of 92 eyes in 80 patients were identified as having been treated with IVBr for nAMD during the course of the study. Included in the analysis were 59 eyes of 56 patients that had persistent fluid despite at least three previous anti-VEGF injections with a minimum of two visits of follow-up after switching to IVBr. Of those 59 eyes, 48 were last treated with IVA (the IVA-IVBr cohort), 10 were last treated with IVBe (the IVBe-IVBr cohort), and one was last treated with IVR. The patient demographic and baseline characteristics for each cohort are summarized in Table 1. Table 2 describes the compartments of persistent fluid (IRF, SRF, or both) for patients prior to switch to IVBr.
Baseline characteristics and demographics of patients switched to intravitreal brolucizumab.
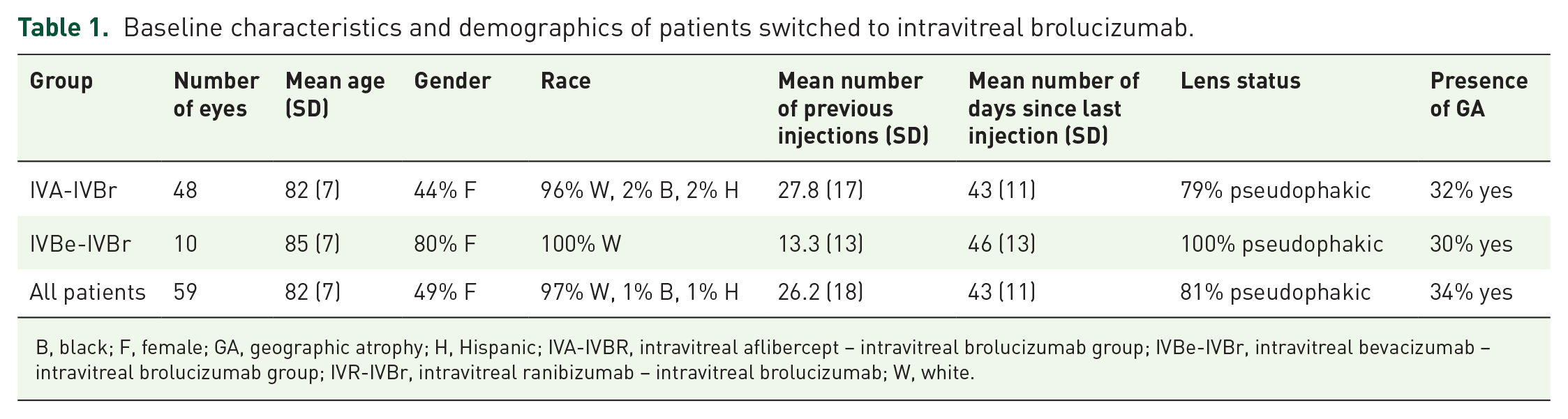
B, black; F, female; GA, geographic atrophy; H, Hispanic; IVA-IVBR, intravitreal aflibercept – intravitreal brolucizumab group; IVBe-IVBr, intravitreal bevacizumab – intravitreal brolucizumab group; IVR-IVBr, intravitreal ranibizumab – intravitreal brolucizumab; W, white.
Baseline fluid compartments of patients with persistent fluid prior to switch to IVBr.

IVA-IVBR, intravitreal aflibercept – intravitreal brolucizumab group; IVBe-IVBr, intravitreal bevacizumab – intravitreal brolucizumab group; IRF, intraretinal fluid; SRF, subretinal fluid.
Results for IVA-IVBr cohort
Patients in the IVA-IVBr cohort had nAMD for a mean of 3.5 ± 2.2 years and received a mean of 28 ± 17 (range: 4–77) prior anti-VEGF treatments prior to switching to IVBr. Specifically, these 48 eyes received a mean of 6 IVBe, 0.6 IVR, and 21 IVA. Geographic atrophy was documented in 17/52 (33%) of eyes. The mean time period between the last IVA and the first IVBr injection was 43 ± 11 (range: 28–79) days. The mean CSFT and logMAR BCVA results are summarized in Tables 3 and 4 and Figures 2 and 3, respectively.
Visual acuity change after switching to brolucizumab.

IVA-IVBR, intravitreal aflibercept – intravitreal brolucizumab group; IVBe-IVBr, intravitreal bevacizumab – intravitreal brolucizumab group; IVR-IVBr, intravitreal ranibizumab – intravitreal brolucizumab group; LogMAR BCVA, logarithm of the minimal angle of resolution best corrected visual acuity; SD, standard deviation.
Macular thickness change after switching to brolucizumab.

CSFT, central subfield thickness; IVA-IVBR, intravitreal aflibercept – intravitreal brolucizumab group; IVBe-IVBr, intravitreal bevacizumab – intravitreal brolucizumab group; IVR – IVBr, intravitreal ranibizumab – intravitreal brolucizumab group; SD, standard deviation.

Scatterplot showing the mean best corrected visual acuity (logMAR) for each visit after switching to brolucizumab. There was a slight trend for improved mean logMAR for the IVBe-IVBr group. The IVBe-IVBr subgroup that completed a series of three IVBr initially had improved logMAR until the final visit, when logMAR became worse than baseline. For the IVA-IVBr group, the mean logMAR remained stable throughout the study, but for the subgroup that completed a series of three IVBr there was a slight decrease in mean logMAR every visit. There was no statistically significant change in mean logMAR for any of the groups in this study.
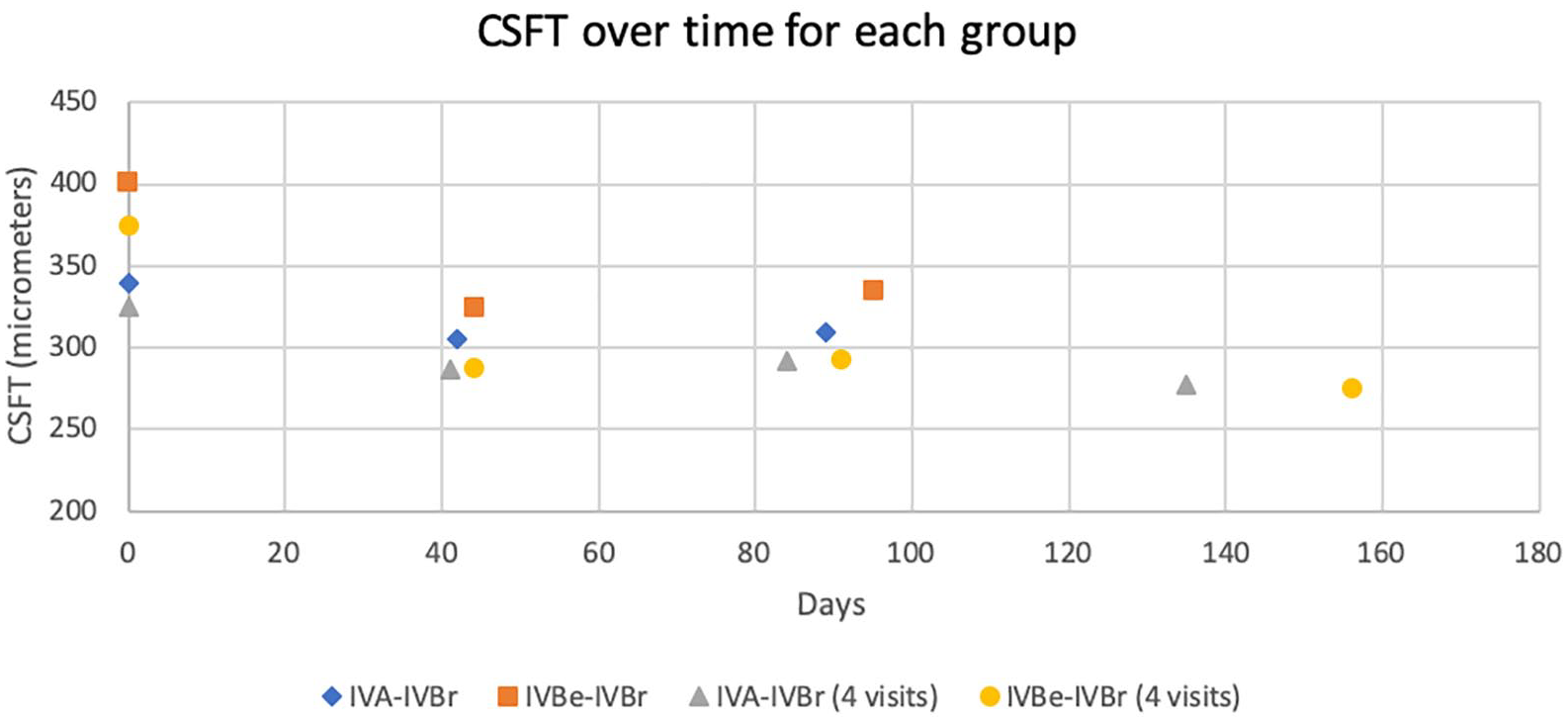
Scatterplot showing the mean central subfield thickness (CSFT) for each visit after switching to brolucizumab. For both the IVA-IVBr and IVBe-IVBr groups, there was a statistically significant reduction in CSFT after the first and second IVBr. For both subgroups that completed a series of three IVBr, there was further reduction of CSFT after the third IVBr.
The mean BCVA before the first IVBr was 20/63 (logMAR visual acuity 0.50 ± 0.31, range: 0.10–1.6) and the mean CSFT was 340 ± 67 (range: 252–561) µm. There was baseline persistent SRF alone in 46%, IRF alone in 38%, and both in 17% of eyes. After one IVBr, when checked a mean of 42 ± 10 (range: 28–70) days later, mean BCVA was 20/50 (logMAR 0.49 ± 0.39, range: 0–1.6, p = 0.73). The mean CSFT improved by 35 µm to 305 ± 76 (range: 204–556) µm (p < 0.001). Complete resolution of fluid was achieved in 15/48 (31%) of eyes, reduced but persistent fluid was achieved in 20/48 (42%) eyes, 12/48 (25%) eyes had no meaningful change in amount of fluid, and 1/48 (2%) eyes had increased fluid (see Figure 4).

Bar graph showing the change in the subjective grading of fluid at each visit after switching from intravitreal aflibercept to intravitreal brolucizumab. The greatest reductions in fluid occurred after the first brolucizumab injection, and there was an increasing proportion of patients with worsening fluid with subsequent treatments. This was likely related to the extended time period between injections as a result of the treat and extend protocol.
After the second IVBr, when checked at the third visit a mean of 47 ± 12 (range: 28–72) days later, the mean BCVA was 20/63 (logMAR 0.50 ± 0.40, range: 0–1.82, p = 0.87). The mean CSFT increased to 310 ± 72 µm (range: 191–573 µm, p < 0.001). Complete resolution of fluid was achieved in 12/48 (25%) of eyes, reduced but persistent fluid was achieved in 15/48 (31%) eyes, 12/48 (25%) eyes had no change in amount of fluid, and 9/48 (19%) eyes had increased fluid compared with the previous visit (see Figure 4).
In total, 25 eyes received a third IVBr treatment and had adequate follow-up and OCT data for inclusion in the data set. This cohort was analyzed as a separate data set due to the large drop off in sample size. At the first, second, and third visits, this cohort’s mean logMAR BCVA was 0.44 ± 0.23, 0.42 ± 0.31 (p = 0.36), and 0.41 ± 0.27 (p = 0.14), respectively. The mean CSFT was 325 ± 62 µm, 287 ± 75 µm (p < 0.001), and 292 ± 66 µm (p = 0.003), respectively, during these visits. When checked at the fourth visit a mean of 54 ± 15 (range: 28–87) days later, the mean BCVA was 20/50 (logMAR 0.40 ± 0.26, range: 0.0–1.0, p = 0.35). The mean CSFT was 277 ± 65 (range: 180–421) µm (p = 0.001). Complete resolution of fluid was achieved in 6/25 (24%) of eyes, reduced but persistent fluid was achieved in 6/25 (24%) eyes, 6/25 (24%) eyes had no change in amount of fluid, and 7/25 (28%) eyes had increased fluid compared with the previous visit (see Figure 4).
Results for IVBe-IVBr cohort
Patients in the IVBe-IVBr cohort had nAMD for a mean of 2.8 years and received a mean of 13 (range: 3–37) prior anti-VEGF treatments prior to switching to IVBr. Specifically, these 10 eyes received a mean of 11.3 IVBe, 0 IVR, and 2 IVA. Geographic atrophy was documented in 3/10 (30%) of eyes. The mean time period between the last IVA and the first IVBr injection was 46 ± 13 (range: 28–67) days.
The mean BCVA before the first IVBr was 20/58 (logMAR 0.46 ± 0.24, range: 0.3–1.1) and the mean CSFT was 401 ± 89 (range: 296–538) µm. There was baseline persistent SRF alone in 60%, IRF alone in 30%, and both in 10% of eyes. After one IVBr, when checked a mean of 44 ± 12 (range: 28–58) days later, mean BCVA improved to 20/50 (logMAR 0.40 ± 0.26, range: 0.1–1.0, p = 0.114). The mean CSFT improved by 76 µm to 325 ± 89 (range: 217–517) µm (p = 0.009). Complete resolution of fluid was achieved in 3/10 (30%) of eyes, reduced but persistent fluid was achieved in 7/10 (70%) eyes, and no eyes had unchanged or worsening fluid compared with the previous visit (see Figure 5).

Bar graph showing the change in the subjective grading of fluid at each visit after switching from intravitreal bevacizumab to intravitreal brolucizumab. All the patients had either improved or completely resolved fluid after the first brolucizumab injection. With subsequent brolucizumab injections, there was an increasing proportion of patients with worsening fluid as the treatment intervals were extended.
All 10 eyes received a second IVBr treatment. At the follow-up, a mean of 51 ± 13 (range: 28–77) days later, the mean BCVA was 20/51 (logMAR 0.40 ± 0.27, range: 0.1–1.0, p = 0.169). The mean CSFT was 335 ± 109 (range: 214–571) µm (p = 0.025). Complete resolution of fluid was achieved in 3/10 (30%) of eyes, reduced but persistent fluid was achieved in 4/10 (40%) eyes, 2/10 (20%) eyes had stable fluid, and 1/10 (10%) had worsening fluid (see Figure 5).
Four eyes received a third IVBr and had adequate follow-up for inclusion in the data set. These remaining four eyes were analyzed as a separate data set due to the large drop off in sample size. At the first, second, and third visits, this cohort’s logMAR BCVA was 0.33 ± 0.05 (Snellen 20/42), 0.27 ± 0.11 (Snellen 20/37, p = 0.182), and 0.22 ± 0.10 (Snellen 20/33, p = 0.084), respectively. The CSFT was 375 ± 53 µm, 288 ± 32 µm (p = 0.046), and 293 ± 52 µm (p = 0.006) on these visits, respectively.
For the remaining four eyes, at follow-up a mean of 65 ± 21 (range: 35–86) days later, the mean BCVA was 20/45 (logMAR 0.35 ± 0.06, range: 0.3–0.4, p = 0.391). The mean CSFT was 275 ± 29 (range: 244 – 311) µm (p = 0.088). Complete resolution of fluid was achieved in 2/4 (50%) of eyes, reduced but persistent fluid was achieved in 1/4 (25%) eyes, and 1/4 (25%) eyes had increased fluid compared with the previous visit (see Figure 5).
Results for IVR-IVBr cohort
As ranibizumab is not used frequently in our clinics, only one patient was switched from IVR to IVBr. The patient had baseline 20/200 BCVA, with persistent SRF and CSFT of 626 µm when checked 42 days after his last IVR. The BCVA remained stable at 20/200 after two IVBr injections. The SRF was reduced with each injection, and CSFT improved to 524 and 537 µm, when checked 42 and 49 days later, respectively.
Safety profile
Of the 92 eyes treated with IVBr, one patient developed mild anterior uveitis that resolved with topical steroids. As it was uncertain whether the inflammation was related to IVBr, the patient was treated with another IVBr 1 month later, and there was recurrence of the anterior uveitis. After successful treatment with topical steroids a second time, IVBr was discontinued. This patient was not included in the final analysis as they did not meet the inclusion criteria for follow-up. No patients were found to have evidence of vitritis or vasculitis after treatment with IVBr, though patients did not have a dilated exam on every visit unless there were new symptoms or a drop in vision.
Discussion
The October 2019 US FDA approval of IVBr provided another anti-VEGF treatment option for ophthalmologists treating patients with nAMD. The phase 3 HAWK and HARRIER trials demonstrated that IVBr was noninferior to IVA in terms of visual gains at 48 weeks, with over half of the IVBr patients maintaining a dry retina at 12-week dosing intervals.
At the time of this writing there are limited published data on the efficacy of switching to IVBr in patients previously treated with anti-VEGF agents. One case series of six patients with recalcitrant nAMD showed that after a single IVBr, there was a reduction of SRF/IRF and reduced CSFT in all six patients. Visual acuities remained stable and there were no adverse events in any patients. 9 Another multi-center retrospective real-world case series included a subgroup of 109 eyes that were switched from another anti-VEGF agent to IVBr due to persistence of fluid on OCT. These eyes had baseline BCVA of 64.1 ETDRS letters and received a mean of 1.46 IVBr injections. Like our study, there was no significant change in BCVA (final BCVA 63.3 ETDRS letters; p = 0.65). The mean CSFT in all eyes prior to starting IVBr was 296.7 µm and was 269.8 µm at the last study examination (mean difference, 26.9 µm; p = 0.003). Intraocular inflammation was reported in 8.1% of eyes, with half of the cases self-resolved, though one patient developed occlusive retinal vasculitis with severe loss of vision. 10 The REBA study was an international retrospective observational real-world IVBr study with a subgroup of 55 switch-therapy patients (80 eyes), which showed statistically significant improvement in VA (mean gain of 10.4 ± 4.8 letters, p = 0.014) and CSFT (−185.7 µm, p = 0.013). Out of the 80 eyes included in this group, 27 (33.7%) were considered to have no disease activity after the first injection, 38 (47.5%) after the second injection, and 15 (18.8%) after the third injection. 11
In this study, there was a statistically significant reduction in CSFT in both the IVA-IVBr and IVBe-IVBr groups after just one IVBr injection. The IVBe-IVBr group had a higher mean baseline CSFT than the IVA-IVBr group (401 ± 89 versus 336 ± 68 µm), so there was a larger improvement in mean CSFT between the first and second visits (66 versus 26 µm). For both groups, there was a trend for slightly increased CSFT between the second and third injection visits, though the CSFT improvement was still statistically significant compared with baseline at the third visit. For the IVA-IVBr and IVBe-IVBr subgroups that had four visits of uninterrupted follow-up, the CSFT improvement maintained statistical significance at last follow-up for the IVA-IVBr group but not for the IVBe-IVBr group (the sample size was much smaller for the IVA-IVBr group). There was no statistically significant change in visual acuity for any of the groups at any point in this study. It is possible that pre-existing photoreceptor damage from chronic fluid and geographic atrophy limited the visual acuity gains despite anatomic improvements after switching to IVBr. Previous studies have demonstrated that some SRF, but not IRF, can be tolerated in nAMD eyes without compromising visual outcomes.12,13 In our study, 46% and 60% of patients in the IVA-IVBr and IVBe-IVBr groups had SRF alone at baseline, respectively; 54% and 40% had IRF either alone or in combination with SRF, which may predispose those patients to worse visual acuity.
As IVBr has the smallest molecular size (26 kDa) compared with IVA (115 kDa) and IVBe (149 kDa), improvements in anatomic outcomes could be due to increased tissue penetrance and higher molar dosing per injection. However, tachyphylaxis to the previous agent may also account for the improved response in patients switched to IVBr. Gale et al. 14 demonstrated that in nAMD patients who were suboptimal responders to IVA, switching to IVR led to a significant improvement in CSFT (−31 microns by day 90) with approximately 60% of patients experiencing stabilized/improved BCVA. In another retrospective study, when non-responders or poor responders to three monthly IVR were then treated with three monthly IVA, there was a significant anatomic improvement in CSFT, but no significant change in visual acuity, comparable to the results in our study. 15 Despreaux et al. 16 showed that in nAMD patients previously switched from IVR to IVA, a ‘switchback’ of IVA to IVR resulted in a short-term benefit in terms of both CSFT and BCVA.
In our study, there was a trend for increased mean CSFT after the second treatment. This is best explained by the treat and extend protocol employed in this study, which is the most commonly employed treatment regimen by retina specialists 17 and mirrors the real-world approach more than the fixed dosing intervals utilized in large registration trials such as HAWK and HARRIER. Patients were extended by increments of approximately 1 week if the treating physician determined the macula was dry, which was reflected in the mean follow-up of approximately 6 weeks after first IVBr, 7 weeks after second IVBr, and 8 weeks after third IVBr. Complete resolution of fluid was achieved after the first IVBr in 31% and 30% of the eyes in the IVA-IVBr and IVBe-IVBr groups, respectively. After the second and third injections for the IVA-IVBr group, only 25% and 24% of eyes had a dry macula, respectively, which was likely a result of the extended time interval between visits. For the IVBe-IVBr group, the percentage of eyes with a dry macula increased to 30% and 50% after the second and third injections, respectively, despite the extended time interval between injections.
Rare cases of visually devastating occlusive retinal vasculitis have been reported in patients treated with IVBr. 18 As a result, IVBr was discontinued in all patients as a safety precaution. As of 1 April 2020, there were 26 reported cases of retinal vasculitis in 25 patients, after injection of 70,000 vials in 37,000 patients. In the HAWK and HARRIER phase 3 trials, IVBr-associated inflammation was noted at a rate of 4% (32 of 730 patients) and retinal artery occlusion at a rate of ~1% (6 of 730 patients). 7 There were no serious adverse events observed in any of the patients in this study, though one patient developed anterior uveitis that resolved with topical steroids.
Limitations of this study include its retrospective nature, modest sample size, lack of control group, interruptions in care related to the COVID-19 pandemic, and the unexpected interruption in IVBr treatment following the ASRS safety warning. To avoid confounded data caused by using ‘last observation carried forward’ for missing data points, a separate analysis was performed for the smaller subgroups groups that completed four visits of follow-up. Prior to switching to IVBr, the mean interval of 6 weeks since last anti-VEGF injection is longer than the suggested treatment interval of 4 weeks for refractory nAMD; however, patients kept the same follow-up interval after their first IVBr injection, so the comparison between the old and new drug was not confounded by shorter follow-up interval.
In summary, intravitreal brolucizumab utilized in a treat and extend protocol may reduce persistent IRF/SRF in patients with nAMD who have been previously treated with aflibercept or bevacizumab. For both groups, there was a statistically significant reduction in CSFT after one IVBr, with approximately 30% of patients free of IRF/SRF. Visual acuity did not significantly change for either group during the course of the study.
Footnotes
Author contributions
RMH conceptualized and designed this study. RMH and AN participated in data collection. RMH and NAY performed statistical analysis. RMH created the initial draft of this manuscript. NAY, SH, SPB, KHP, and SMH. contributed to critical revision of the manuscript. All authors approved the final version of this manuscript.
Conflict of interest statement
R.M.H. has served on an advisory board for Alimera Sciences. N.A.Y. has served on advisory boards for Novartis, Genentech, and Alimera Sciences. S.M.H. is a consultant or on the speaker’s bureau for Allergan, Graybug, Novartis, OD-OS, Clearside Biomedical, EyePoint Pharmaceuticals, Alimera Sciences, Spark Therapeutics and Regeneron. None of the remaining authors have any financial disclosures to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
